-
Name
Sasapyrine
- EINECS 209-027-4
- CAS No. 552-94-3
- Article Data15
- CAS DataBase
- Density 1.404 g/cm3
- Solubility Hydrolysis in water
- Melting Point 148-151°C
- Formula C14H10O5
- Boiling Point 482.9 °C at 760 mmHg
- Molecular Weight 258.23
- Flash Point 188 °C
- Transport Information
- Appearance white crystalline powder
- Safety 37/39-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Sasapyrinum;o-Salicylsalicylic acid;Salicylicacid, bimol. ester (7CI);Salicylic acid, salicylate (6CI);2-Hydroxybenzoicacid 2-carboxyphenyl ester;Diacesal;Diplosal;Disalcid;Disalgesic;Disalicylic acid;Sasapyrine;Disalyl;Mono-Gesic;NSC 49171;Nobacid;Salflex;Salical;Salicyl salicylate;Salicyloxysalicylic acid;Salicyloylsalicylic acid;Salicylsalicylic acid;Salina;Salsalate;Salysal;Sasapirin;
- PSA 83.83000
- LogP 2.30960
Synthetic route

| Conditions | Yield |
|---|---|
| With diethylamine In water; 1,2-dichloro-ethane at 0 - 25℃; for 12h; Temperature; Large scale; | 85.08% |
| With dimethyl fluorenylidenetributylphosphoranylidenesuccinate In benzene at 80℃; for 7h; | 79% |
| Multi-step reaction with 2 steps 1: pyridine / -5 °C 2: aluminium bromide; benzene View Scheme |
-
-
123-91-1
1,4-dioxane

-
-
623-11-0
1-methyl-4-nitrosobenzene

-
-
486-58-8
disalicylide

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
75-44-5
phosgene

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
75-44-5
phosgene

-
-
69-72-7
salicylic acid

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester


| Conditions | Yield |
|---|---|
| With diluted alkali | |
| With 10 percent human plasma In acetonitrile at 37℃; Rate constant; phosphate buffer (pH 7.4); or in undiluted plasma; | |
| With diluted alkali |

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With acid | |
| With acid |

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With diluted alkali | |
| With aluminum tri-bromide; benzene | |
| With diluted alkali |
-
-
75-44-5
phosgene

-
-
121-69-7
N,N-dimethyl-aniline

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
75-44-5
phosgene

-
-
121-69-7
N,N-dimethyl-aniline

-
-
69-72-7
salicylic acid

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
530-75-6
acetylsalicylsalicylic acid

-
A
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
B
-
50-78-2
aspirin

| Conditions | Yield |
|---|---|
| In water at 40℃; Rate constant; ionic strength 0.5; effect of pH; |
-
-
50-78-2
aspirin

-
A
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
B
-
530-75-6
acetylsalicylsalicylic acid

-
C
-
69-72-7
salicylic acid

| Conditions | Yield |
|---|---|
| With magnesium stearate at 60℃; Product distribution; |

-
-
50-78-2
aspirin

-
A
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
B
-
530-75-6
acetylsalicylsalicylic acid

-
C
-
85539-30-6
2-{2-[2-(2-acetoxy-benzoyloxy)-benzoyloxy]-benzoyloxy}-benzoic acid

-
D
-
85531-17-5
tri-salicylic acid

-
F
-
69-72-7
salicylic acid

| Conditions | Yield |
|---|---|
| With magnesium carbonate at 85℃; for 2h; Product distribution; also in benzene or toluene with or without magnesium carbonate; |

-
-
7664-93-9
sulfuric acid

-
-
108-24-7
acetic anhydride

-
-
64-19-7
acetic acid

-
-
6543-57-3
tetrasalicylide

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
7719-09-7
thionyl chloride

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
7719-09-7
thionyl chloride

-
-
121-69-7
N,N-dimethyl-aniline

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
121-69-7
N,N-dimethyl-aniline

-
-
108-88-3
toluene

-
-
69-72-7
salicylic acid

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
7719-09-7
thionyl chloride

-
-
69-72-7
salicylic acid

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
7719-09-7
thionyl chloride

-
-
121-69-7
N,N-dimethyl-aniline

-
-
69-72-7
salicylic acid

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
110-86-1
pyridine

-
-
69-72-7
salicylic acid

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
121-69-7
N,N-dimethyl-aniline

-
-
69-72-7
salicylic acid

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
71-43-2
benzene

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester


-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With acetic acid |

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; sodium hydroxide | |
| With sodium hydroxide; acetic acid |

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester
-
-
7647-01-0
hydrogenchloride

-
-
64-19-7
acetic acid

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester



| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aqueous acetone 2: diluted alkali View Scheme | |
| Multi-step reaction with 2 steps 1: pyridine 2: diluted alkali View Scheme | |
| Multi-step reaction with 2 steps 1: dimethylaniline 2: diluted alkali View Scheme |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
657-24-9
dimethylbiguanide

-
-
1411413-55-2
{[amino(imino)methyl]amino}(dimethylamino)methaniminium 2-[(2-hydroxybenzoyl)oxy]benzoate

| Conditions | Yield |
|---|---|
| In acetonitrile at 0 - 20℃; for 2h; | 96% |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
108-24-7
acetic anhydride

-
-
530-75-6
acetylsalicylsalicylic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid 1.) 20 min; 2.) 40 min, 80 deg C; | 84% |
| With sulfuric acid at 20℃; | 5.5 g |
| With sulfuric acid at 20℃; | 5.5 g |
-
-
111-19-3
sebacoyl chloride

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
537048-81-0
1,10-bis-salicylsalicyl-sebacate

| Conditions | Yield |
|---|---|
| Stage #1: sebacoyl chloride; 2-hydroxy-benzoic acid, 2-carboxyphenyl ester With sodium hydride In tetrahydrofuran at 0℃; for 2h; Stage #2: With hydrogenchloride In tetrahydrofuran; water pH=2; | 83% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 1h; | 80% |

| Conditions | Yield |
|---|---|
| With tributylphosphine In tetrahydrofuran at -15℃; for 3h; Inert atmosphere; | 80% |
| With tributylphosphine In tetrahydrofuran at -15℃; for 3h; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 25 - 30℃; | A 80% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-benzoic acid, 2-carboxyphenyl ester With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: C33H55N3O5S2 In N,N-dimethyl-formamide for 2h; | 75% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-benzoic acid, 2-carboxyphenyl ester With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: C19H39NO5Si In N,N-dimethyl-formamide at 20℃; for 2h; | 75% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 1h; | 63% |
-
-
38780-40-4, 52691-24-4, 61848-70-2, 61848-66-6, 61848-62-2
{(1R,2R)-cyclohexane-1,2-diamine}dichloridoplatinum(II)

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With sodium carbonate; silver nitrate In N,N-dimethyl-formamide at 20℃; for 24h; Darkness; | 52% |
-
-
67-56-1
methanol

-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
85531-25-5
2-salicyloyloxy-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic anhydride | |
| With acetic anhydride |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
90510-23-9
2-salicyloyloxy-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With chloroform; chlorine |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With chloroform; bromine |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
56529-74-9
2-(2-hydroxy-5-iodo-benzoyloxy)-benzoic acid

| Conditions | Yield |
|---|---|
| With chloroform; Iodine monochloride | |
| With chloroform; iodine trichloride |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

| Conditions | Yield |
|---|---|
| With chloroform; nitric acid |
-
-
552-94-3
2-hydroxy-benzoic acid, 2-carboxyphenyl ester

-
-
5981-18-0
1,9,17-trioxa-[2.2.2]orthocyclophane-2,10,18-trione

| Conditions | Yield |
|---|---|
| With xylene; trichlorophosphate |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
Sasapyrine Consensus Reports
Sasapyrine Specification
The Salsalate, with the CAS registry number 552-94-3, is also known as Diacesal. It belongs to the product categories of Drug Bulk; Functional Materials; Liquid Crystals & Related Compounds; Phenyl Esters (Liquid Crystals); API's. Its EINECS registry number is 209-027-4. This chemical's molecular formula is C14H10O5 and molecular weight is 258.2262. Its IUPAC name is called 2-(2-hydroxybenzoyl)oxybenzoic acid.
Physical properties of Salsalate: (1)ACD/LogP: 3.05; (2)ACD/LogD (pH 5.5): 0.55; (3)ACD/LogD (pH 7.4): -0.19; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 3.42; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 5; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 5; (11)Index of Refraction: 1.645; (12)Molar Refractivity: 66.7 cm3; (13)Molar Volume: 183.8 cm3; (14)Surface Tension: 64.9 dyne/cm; (15)Density: 1.404 g/cm3; (16)Flash Point: 188 °C; (17)Enthalpy of Vaporization: 78.78 kJ/mol; (18)Boiling Point: 482.9 °C at 760 mmHg; (19)Vapour Pressure: 3.88E-10 mmHg at 25°C.
Preparation of Salsalate: this chemical can be prepared by 2-hydroxy-benzoic acid. This reaction will need reagent phosphorus chloride.
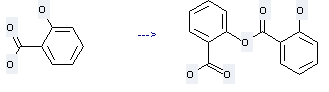
Uses of Salsalate: it can be used to produce 2-(2-acetoxy-benzoyloxy)-benzoic acid at temperature of 80 °C. This reaction will need reagent cc. H2SO4 with reaction time of 40 min. The yield is about %.

Salsalate is used to reduce pain and inflammation caused by conditions such as rheumatoid arthritis, osteoarthritis, and related rheumatic conditions. It has also been used as an alternative to narcotic pain medicine for people with spinal disc protrusion.
When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C1=CC=C(C(=C1)C(=O)OC2=CC=CC=C2C(=O)O)O
(2)InChI: InChI=1S/C14H10O5/c15-11-7-3-1-5-9(11)14(18)19-12-8-4-2-6-10(12)13(16)17/h1-8,15H,(H,16,17)
(3)InChIKey: WVYADZUPLLSGPU-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LDLo | unreported | 1200mg/kg (1200mg/kg) | "Abdernalden's Handbuch der Biologischen Arbeitsmethoden." Vol. 4, Pg. 1289, 1935. | |
| guinea pig | LDLo | unreported | 2gm/kg (2000mg/kg) | "Abdernalden's Handbuch der Biologischen Arbeitsmethoden." Vol. 4, Pg. 1289, 1935. | |
| man | TDLo | oral | 2229mg/kg/2Y- (2229mg/kg) | KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" | Annals of Internal Medicine. Vol. 107, Pg. 116, 1987. |
| mouse | LD50 | subcutaneous | 1020mg/kg (1020mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 22, Pg. 1101, 1980. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View