-
Name
TETRAETHYLLEAD
- EINECS 201-075-4
- CAS No. 78-00-2
- Article Data109
- CAS DataBase
- Density 1.653 g/mL at 25 ºC(lit.)
- Solubility Insoluble
-
Hazard Symbols

- Synonyms Lead,tetraethyl-; NSC 22314; TEL; Tetraethyllead; Tetraethylplumbane
- PSA 0.00000
- LogP 3.51480
Synthetic route
-
-
79110-50-2
(η5-cyclopentadienyl)W(CO)3PbEt3

-
A
-
79123-28-7
{(η5-cyclopentadienyl)W(CO)3}2PbEt2

-
B
-
78-00-2
tetraethyllead(IV)

| Conditions | Yield |
|---|---|
| heated in vac. to 60°C for 8 h, under nitrogen, yellow-orange liquid became solid and darker orange in colour; dissolved in CH2Cl2/hexane solution, placed upon alumina column, elutedwith hexane, then hexane/CH2Cl2 solution, concentrated and placed refrigerator; | A 58% B n/a |
-
-
79110-53-5
(η5-cyclopentadienyl)Mo(CO)3PbEt3

-
B
-
78-00-2
tetraethyllead(IV)

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); 12 h; | A 50% B n/a |

| Conditions | Yield |
|---|---|
| With pyridine; lead sodium | |
| With pyridine; tetrasodium-monoplumbide; water at 38℃; | |
| With lead; sodium at 70℃; im geschlossenen Gefaess; |

| Conditions | Yield |
|---|---|
| With lead sodium; water; toluene; potassium iodide |

| Conditions | Yield |
|---|---|
| With lead sodium; hydrogen at 100℃; under 7355.08 Torr; |

| Conditions | Yield |
|---|---|
| With lead; lead(II) iodide at 100 - 130℃; | |
| With lead sodium | |
| With hydrogenchloride; lead sodium |

| Conditions | Yield |
|---|---|
| With lead(IV) chloride; Petroleum ether anfangs unter Kuehlung; |

| Conditions | Yield |
|---|---|
| With PbS | |
| With lead(II) chloride | |
| With lead(II) oxide |

| Conditions | Yield |
|---|---|
| Electrolysis.an einer Bleianode; |

| Conditions | Yield |
|---|---|
| With lead(II) chloride | |
| With lead(II) chloride |

| Conditions | Yield |
|---|---|
| With benzene |

| Conditions | Yield |
|---|---|
| With sodium fluoride Electrolysis.an Blei-Anoden; |

| Conditions | Yield |
|---|---|
| With lead(II) chloride | |
| With diethyl ether; lead(II) chloride | |
| With lead(II) chloride |

| Conditions | Yield |
|---|---|
| With diethyl ether; lead(II) chloride anschliessend Einleiten Luft oder Sauerstoff nach Zusatz von verd.Salzsaeure; |

| Conditions | Yield |
|---|---|
| With ethyl iodide | |
| With silicatene |

| Conditions | Yield |
|---|---|
| at 170℃; | |
| With hydrogenchloride; oxygen In neat (no solvent) | |
| With hydrogenchloride; air In neat (no solvent) | |
| With air; HCl In neat (no solvent) | |
| With O2; HCl In neat (no solvent) |

| Conditions | Yield |
|---|---|
| With lead(II) chloride |

| Conditions | Yield |
|---|---|
| With tetrasodium-monoplumbide at 72℃; | |
| With lead sodium |
-
-
925-90-6
ethylmagnesium bromide

-
-
41141-89-3
triethyllead bromide

-
-
141-78-6
ethyl acetate

-
-
78-00-2
tetraethyllead(IV)


| Conditions | Yield |
|---|---|
| at 35 - 70℃; Kinetics; | |
| at 35 - 70℃; |

| Conditions | Yield |
|---|---|
| at 75℃; Kinetics; | |
| at 75℃; |

| Conditions | Yield |
|---|---|
| at 30℃; Kinetics; | |
| at 30℃; |

-
-
78-00-2
tetraethyllead(IV)

| Conditions | Yield |
|---|---|
| With lead(II) oxide | |
| With PbS |

| Conditions | Yield |
|---|---|
| With diethyl ether; copper |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - -30℃; Product distribution; further aldehydes and tetraalkylleads; reaction with mixed tetraalkylleads; other conditions; | 98% |
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 98% |
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 96% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 98% |
-
-
650-52-2
bis(trifluoromethyl)chlorophosphine

-
-
78-00-2
tetraethyllead(IV)

-
-
25196-23-0
Ethylbis-trifluormethyl-phosphin

| Conditions | Yield |
|---|---|
| 110°C (24 h); | 95% |
| 110°C (24 h); | 95% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 94% |
-
-
40848-81-5
N-(ethoxycarbonyl)-N-methyl-1-ethoxybenzylamine

-
-
78-00-2
tetraethyllead(IV)

-
-
127202-36-2
Methyl-(1-phenyl-propyl)-carbamic acid ethyl ester

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - 25℃; | 88% |

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 65℃; for 3h; | 84% |
-
-
78-00-2
tetraethyllead(IV)

-
-
127202-34-0
(1-Ethoxy-octyl)-methyl-carbamic acid ethyl ester

-
-
127202-43-1
(1-Ethyl-octyl)-methyl-carbamic acid ethyl ester

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - 25℃; | 82% |
-
-
20738-34-5
(p-chlorophenyl)arsonous dichloride

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
83627-00-3
(p-chlorophenyl)ethylarsinous chloride

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 82% C n/a |

-
-
81445-44-5
(S)-2-(benzyloxy)propanal

-
-
78-00-2
tetraethyllead(IV)

-
-
114418-33-6
(2S,3S)-2-benzyloxy-3-pentanol

| Conditions | Yield |
|---|---|
| With titanium tetrachloride at -78 - -60℃; | 81% |

| Conditions | Yield |
|---|---|
| In further solvent(s) in boiling solvent, 2 h; crystn. from C6H6/hexane, CS2/hexane or CH2Cl2; | 77% |
| In benzene in boiling benzene, 2 h; crystn. from C6H6/hexane, CS2/hexane or CH2Cl2; | 77% |
-
-
16248-71-8
2-(dichloroamino)-2-methylpropionitrile

-
-
78-00-2
tetraethyllead(IV)

-
-
1529-35-7
triethyl lead (1+); hydroxide

| Conditions | Yield |
|---|---|
| With NaOH; HCl In tetrachloromethane byproducts: azobisbutyronitrile; stirring (25-30°C), heating (50°C, 1 h), solvent removal (vac.), addn. of aq. NaOH, stirring (pptn. of org. byproduct), neutrlization (aq. HCl); collection of ppt. (filtration); | 75% |
-
-
63294-40-6
Dichlor(o-methoxyphenyl)arsin

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
61130-93-6
ethyl-o-anisylarsine chloride

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 74% C n/a |

-
-
20738-32-3
(p-chlorophenyl)arsonous dichloride

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
83627-06-9
(o-chlorophenyl)ethylarsinous chloride

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 73% C n/a |
| at 80 - 90℃; for 1h; | A n/a B 73% C n/a |


| Conditions | Yield |
|---|---|
| With mercury dichloride In ethanol 4h heated at 65°C;; precipitation at cooling;; | A n/a B 72% |
| With HgCl2 In ethanol 4h heated at 65°C;; precipitation at cooling;; | A n/a B 72% |
-
-
698-64-6
dichloro(4-methylphenyl)arsine

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
51525-20-3
ethyl-p-tolylarsine chloride

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 71% C n/a |


| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 71% C n/a |


| Conditions | Yield |
|---|---|
| In benzene refluxing in ratio of SbCl3:Pb(C2H5)4=3:1 for 8-10 h; distn.; | 71% |
| In benzene refluxing in ratio of SbCl3:Pb(C2H5)4=3:1 for 8-10 h; distn.; | 71% |
| in ratio of SbCl3:Pb(C2H5)4=3:1 in bulk at 100°C in N2 atmosphere for 7 h; distn.; | |
| in ratio of SbCl3:Pb(C2H5)4=3:1 in bulk at 100°C in N2 atmosphere for 7 h; distn.; |

| Conditions | Yield |
|---|---|
| In toluene byproducts: PbEt3Br; (argon); -60°C to room temp., stirring 2 h; solvent removal (vac.), dissoln. (hexane), filtration; | 71% |

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 70% C n/a |

-
-
65452-83-7
p-anisylarsine dichloride

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
61130-95-8
chloroethyl(p-methoxyphenyl)arsine

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 69% C n/a |

-
-
57170-65-7
o-Bromphenylarsindichlorid

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
83627-07-0
C8H9AsBrCl

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 68% C n/a |


| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 68% C n/a |


| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 67% C n/a |

-
-
73013-32-8
p-bromophenylarsine dichloride

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
83627-01-4
C8H9AsBrCl

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 64% C n/a |


| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 64% C n/a |


| Conditions | Yield |
|---|---|
| In xylene for 20h; Heating; | 63.8% |
-
-
696-28-6
dichlorophenylarsine

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
698-13-5
ethylphenylarsine chloride

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 63% C n/a |

-
-
697-97-2
o-tolylarsonous dichloride

-
-
78-00-2
tetraethyllead(IV)

-
A
-
75-00-3
chloroethane

-
B
-
53980-31-7
Aethyl-o-tolylarsinchlorid

| Conditions | Yield |
|---|---|
| at 90 - 110℃; | A n/a B 62% C n/a |

-
-
78-00-2
tetraethyllead(IV)

-
-
135441-05-3
C11H19N3O3

-
A
-
135441-06-4
N'-(Piperidine-1-carbonyl)-hydrazinecarboxylic acid tert-butyl ester

-
B
-
135441-08-6
N'-Ethyl-N'-(piperidine-1-carbonyl)-hydrazinecarboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In hexane; dichloromethane at -78℃; for 0.7h; | A 6% B 61% |


| Conditions | Yield |
|---|---|
| In chlorobenzene | 61% |

TETRAETHYL LEAD Chemical Properties
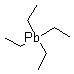
IUPAC Name: tetraethylplumbane
Molecular Formula:C8H20Pb
Molecular Weight:323.4444
Appearance:colorless, viscous liquid
Melting Point:136 °C(lit.)
Boiling Point:84-85 °C15 mm Hg(lit.)
density:1.653 g/mL at 25 °C(lit.)
refractive index:n20/D 1.519(lit.)
Fp:84 °F
Solubility:slightly soluble in ethanol;
soluble in benzene, petroleum ether, toluene, gasoline
Insolubility:Water
Synonyms of TETRAETHYL LEAD(78-00-2):TEL;TETRAETHYLLEAD;
(C2H5)4Pb;Czteroetylek olowiu;czteroetylekolowiu;czteroetylekolowiu(polish);Lead tetraethyl;lead,tetraethyl-
Categories of TETRAETHYL LEAD(78-00-2): Organometallics;Organolead;Organometallic Reagents;Others
Merck 13,9277
TETRAETHYL LEAD Uses
TETRAETHYL LEAD Production
4 NaPb + 4 CH3CH2Cl → (CH3CH2)4Pb + 4 NaCl + 3 Pb
TETRAETHYL LEAD Toxicity Data With Reference
| 1. | otr-ham:emb 12,500 µg/L | AETODY Advances in Modern Environmental Toxicology. 1 (1980),241. | ||
| 2. | ihl-hmn TCLo:1749 g/m3/30M:NOSE,EYE,GIT | SAIGBL Sangyo Igaku. Japanese Journal of Industrial Health. 17 (1975),223. | ||
| 3. | unr-man LDLo:1470 µg/kg | 85DCAI Poisoning; Toxicology, Symptoms, Treatments. 2 (1970),73. | ||
| 4. | orl-rat LD50:12,300 µg/kg | EXPEAM Experientia. 28 (1972),923. | ||
| 5. | ihl-rat LC50:850 mg/m3/60M | BJIMAG British Journal of Industrial Medicine. 18 (1961),277. | ||
| 6. | ipr-rat LD50:15 mg/kg | MELAAD Medicina del Lavoro. Industrial Medicine. 54 (1963),486. | ||
| 7. | ivn-rat LD50:14,400 µg/kg | MELAAD Medicina del Lavoro. Industrial Medicine. 54 (1963),486. | ||
| 8. | par-rat LD50:15 mg/kg | AOHYA3 Annals of Occupational Hygiene. 3 (1961),226. | ||
| 9. | ihl-mus LCLo:650 mg/m3/7H | SAIGBL Sangyo Igaku. Japanese Journal of Industrial Health. 15 (1973),3. | ||
| 10. | skn-dog LDLo:547 mg/kg | SAIGBL Sangyo Igaku. Japanese Journal of Industrial Health. 15 (1973),3. |
TETRAETHYL LEAD Consensus Reports
TETRAETHYL LEAD Safety Profile
Human poison by an unspecified route. Experimental poison by ingestion, intraperitoneal, intravenous, subcutaneous, and parenteral routes. Moderately toxic by inhalation and skin contact. Experimental teratogenic and reproductive effects. Questionable carcinogen with experimental carcinogenic data. Mutation data reported. Lead compounds are particularly toxic to the central nervous system. It is a solvent for fatty materials and has some solvent action on rubber as well. The fact that it is a lipoid solvent makes it an industrial hazard because it can cause intoxication not only by inhalation but also by absorption through the skin. Decomposes when exposed to sunlight or allowed to evaporate; forms triethyl lead, which is also a poisonous compound, as one of its decomposition products. May cause elemental lead intoxication by coming in contact with the skin.
A combustible liquid when exposed to heat, flame, or oxidizers. Can react vigorously with oxidizing materials. Exposure to air for several days may cause explosive decomposition. To fight fire, use dry chemical, CO2, mist, foam. When heated to decomposition it emits toxic fumes of Pb. See also LEAD COMPOUNDS.
Hazard Codes T+,N
Risk Statements 61-26/27/28-33-50/53-62-38-10
Safety Statements 53-45-60-61-36/37-26
RIDADR UN 1649 6.1/PG 1
WGK Germany 3
RTECS TP4550000
TSCA Yes
HazardClass 6.1(a)
PackingGroup I
TETRAETHYL LEAD Standards and Recommendations
ACGIH TLV: TWA 0.1 mg(Pb)/m3 (skin); Not Classifiable as a Human Carcinogen
DFG MAK: 0.05 mg/m3
DOT Classification: 6.1; Label: Poison, Flammable Liquid
TETRAETHYL LEAD Analytical Methods
Related Products
- TETRAETHYL GERMANE
- TETRAETHYL LEAD
- Tetraethyl methylenediphosphonate
- Tetraethyl ranelate
- Tetraethylammonium
- Tetraethylammonium hydrogensulfate
- Tetraethylammonium hydroxide
- Tetraethylammonium iodide
- Tetraethylammonium nitrate
- Tetraethylene glycol monophenyl ether
- 78003-71-1
- 78005-04-6
- 78008-15-8
- 78008-36-3
- 7800-91-1
- 780-11-0
- 78016-98-5
- 7801-80-1
- 78028-01-0
- 78031-18-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View