-
Name
TRIPHENYLPHOSPHINE SULFIDE
- EINECS 223-407-7
- CAS No. 3878-45-3
- Article Data240
- CAS DataBase
- Density 1.2 g/cm3
- Solubility Insoluble in water.
- Melting Point 161-163 °C(lit.)
- Formula C18H15 P S
- Boiling Point 428.7 °C at 760 mmHg
- Molecular Weight 294.357
- Flash Point 213.1 °C
- Transport Information
- Appearance WHITE CRYSTALLINE SOLID
- Safety A poison by intravenous route. When heated to decomposition it emits toxic vapors of POx and SOx.
- Risk Codes 21/22-36/37/38
-
Molecular Structure
- Hazard Symbols A poison.
- Synonyms NSC 62216;Triphenylphosphine monosulfide; Triphenylphosphine sulfide
- PSA 41.90000
- LogP 4.09300
Synthetic route

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-N'-(5-sulfanylidene-1,2,4-dithiazol-3-yl)methanimidamide In perdeuteriopyridine for 0.0833333h; | 99.9% |
| With sulfur In chloroform-d1 at 15 - 25℃; for 12h; Inert atmosphere; Sealed tube; | 99% |
| With (Et4N)2[MoVIO(S)(S2C2(COOMe)2)2] In acetonitrile at -40 - -25℃; for 5h; Kinetics; Activation energy; Equilibrium constant; Temperature; | 96% |
-
-
61775-35-7
phenylsulfenyl methyl thiocarbonate

-
A
-
108-98-5
thiophenol

-
B
-
3878-45-3
triphenylphosphine sulfide

-
C
-
791-28-6
Triphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With triphenylphosphine In benzene Mechanism; Ambient temperature; reaction of var. sulfenyl thiocarbonates with triphenylphosphine; | A 99% B 1% C 99% |


| Conditions | Yield |
|---|---|
| In acetonitrile a suspension of (PPh3Et)2Mo3S7Br6 and SePPh3 in MeCN was boiled for 1 h under Ar;; toluene was added to the filtrate, filtn., washed with toluene, dried in vac.;; | A n/a B 99% |

-
-
591-50-4
iodobenzene

-
-
96620-51-8
(hydroxymethyl)diphenylphosphine sulfide

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; 1,1'-bis-(diphenylphosphino)ferrocene; 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene at 40℃; for 3h; | 99% |
-
-
591-50-4
iodobenzene

-
-
1420040-32-9
(benzoyloxymethyl)diphenylphosphine sulfide

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; 1,1'-bis-(diphenylphosphino)ferrocene; sodium ethanolate In tetrahydrofuran at 70℃; for 2h; Inert atmosphere; | 99% |
-
-
591-50-4
iodobenzene

-
-
1420040-31-8
(t-butyldimethylsilyloxymethyl)diphenylphosphine sulfide

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; 1,1'-bis-(diphenylphosphino)ferrocene; cesium fluoride In tetrahydrofuran at 70℃; for 12h; Inert atmosphere; | 99% |
-
-
64-17-5
ethanol

-
-
603-35-0
triphenylphosphine

-
-
343783-49-3
3-tert-butyl-5-phenyl-3H-[1,2,4]dithiazole

-
A
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| at 20℃; for 1h; | A 72% B 98% |


| Conditions | Yield |
|---|---|
| With triphenylphosphine In xylene for 2h; Heating; | 97% |

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With PPh3 In tetrahydrofuran (N2), stirred for 0.5 h; evapd. in high vac., triturated with diethyl ether, NMR; | A 96% B 60% |
-
-
152291-64-0
5-(2,4,6-tris(bis(trimethylsilyl)methyl)phenyl)-5-(2,4,6-triisopropylphenyl)-1,2,3,4,5-tetrathiagermolane

-
-
603-35-0
triphenylphosphine

-
A
-
152291-63-9
[2,4,6-tris[bis(trimethylsilyl)methyl]phenyl](2,4,6-triisopropylphenyl)germanethione

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| In hexane refluxing (2 h),; filtration, concentration (Ar); | A 96% B 4% |

-
-
108-86-1
bromobenzene

-
-
96620-51-8
(hydroxymethyl)diphenylphosphine sulfide

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With 1,1'-bis(diisopropylphosphino)ferrocene; palladium diacetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene at 60℃; for 4h; Catalytic behavior; Reagent/catalyst; Temperature; Inert atmosphere; | 96% |


-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With PPh3 In toluene addn. of PPh3 to suspn. of Cu-complex , dissolution, pptn.; treatment with light petroleum (b.p. 30-50°C), washing of PPH3S off (diethyl ether), recrystn. (CS2, ether); elem. anal.; | A 95% B n/a |
-
-
84369-18-6
{(η5-C5H5)Re(NO)(PPh3)(η2-H2C=S)}(+)PF6(-)

-
A
-
71763-25-2
{(η5-C5H5)Re(NO)(PPh3)(CH2PPh3)}(+)PF6(-)

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With triphenylphosphine In acetone stirred for 2 h under N2 atmosphere; CH2Cl2 was added, ether was slowly introduced by vapor diffusion, crystals of the Re-complex were collected after 2 d by filtration, PPh3S was isolated from the filtrate; | A 95% B 88% |
-
-
72050-06-7
p-Fluorophenylsulfenyl methyl thiocarbonate

-
A
-
371-42-6
4-Fluorothiophenol

-
B
-
3878-45-3
triphenylphosphine sulfide

-
C
-
791-28-6
Triphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With triphenylphosphine In benzene Mechanism; Ambient temperature; | A 90% B 6% C 94% |

-
-
10544-50-0
sulfur

-
-
18421-48-2
trans-{PtCOPh(Cl)(PPh3)2}

-
-
201230-82-2
carbon monoxide

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| In chlorobenzene; benzene Kinetics; N2 atmosphere; addn. of solvents to mixt. of Pt-complex and S8 (room temp.), introduction of CO (10 atm, (31)P((1)H)-NMR monitoring), standing (room temp., 10 days); evapn. to dryness (vac.), mixt. of three complex isomers (60:27:13) not sepd.; | A n/a B 94% |

-
-
17763-67-6
Phenyl triflate

-
-
96620-51-8
(hydroxymethyl)diphenylphosphine sulfide

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With 1,1'-bis(diisopropylphosphino)ferrocene; tetrabutylammomium bromide; palladium diacetate; 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene at 60℃; for 3h; Reagent/catalyst; Inert atmosphere; | 94% |
-
-
123317-05-5
tris(2,6-dimethoxyphenyl)phosphibe sulfide

-
-
603-35-0
triphenylphosphine

-
A
-
123317-07-7
Tris-(2,6-dimethoxy-phenyl)-phosphane; hydrochloride

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With hydrogenchloride In acetone at 50℃; for 3h; | A n/a B 92% |

-
-
827-42-9
5-phenyl-3H-1,2-dithiol-3-one

-
-
64-17-5
ethanol

-
A
-
36367-20-1
ethyl ester of β-(mercapto)cinnamic acid

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With triphenylphosphine In chloroform for 2h; Heating; | A 50% B 91% |

-
-
603-35-0
triphenylphosphine

-
A
-
21612-82-8
P,P,P-triphenylphosphine imide hydrochloride

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With S4N3Cl In acetonitrile at 30℃; for 24h; Product distribution; Mechanism; other cyclic sulphur-nitrogen chlorides; also in the presence of Et3N, pyridine or Na2CO3; var. solvents; | A 91% B 1.09 g |
| With S4N3Cl In acetonitrile for 24h; Ambient temperature; | A 91% B 0.35 g |
-
-
110-91-8
morpholine

-
-
21204-67-1
methyl (triphenylphosphoranylidene)acetate

-
A
-
117081-21-7
methyl 2-morpholino-2-thioxoacetate

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With sulfur In toluene for 12h; Heating; | A 75% B 90% |

-
-
76289-06-0
diisopropoxyphosphinothionyl tert-butylamino trisulfide

-
-
603-35-0
triphenylphosphine

-
A
-
76276-34-1
diisopropoxyphosphinothionyl tert-butylamino disulfide

-
B
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; for 0.25h; | A 81.5% B 90% |


| Conditions | Yield |
|---|---|
| With perchloric acid; (s-tetrathiane-3,6-diylidene)bis(dimethylammonium) diperchlorate In acetonitrile at 60℃; | A 40% B 90% |

| Conditions | Yield |
|---|---|
| With oct-1-ene; sulfur In tetrahydrofuran for 24h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| In acetonitrile a suspension of (NEt4)2Mo3S7Br6 and SePPh3 in MeCN was boiled for 1 h under Ar;; toluene was added dropwise to the filtrate, soln. was allowed to stay at -5°C for 12 h, filtn., washed with toluene, dried in vac., elem. anal.;; | A 90% B >99 |


| Conditions | Yield |
|---|---|
| With perchloric acid In acetonitrile for 0.0166667h; Heating; | A 0.35 g B 89% |


| Conditions | Yield |
|---|---|
| With perchloric acid; (s-tetrathiane-3,6-diylidene)bis(diethylammonium) diperchlorate In acetonitrile for 0.0166667h; Heating; | A 0.35 g B 89% |
-
-
54129-78-1
Dithiooxalsaeure-O,O'-dimethylester

-
-
1439-36-7
1-triphenylphosphoranylidene-2-propanone

-
A
-
85477-45-8
(2E)-3-Acetyl-2-methoxythioacrylsaeure-O-methylester

-
B
-
85477-44-7
(2Z)-3-Acetyl-2-methoxythioacrylsaeure-O-methylester

-
C
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| In benzene for 130h; Heating; | A 23% B 23% C 89% |


-
C
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With triphenylphosphane In dichloromethane (Ar), refluxed (40°C) for 40 min; column chromatography on silica gel (pentane/CH2Cl2 (2:1), pentane/CH2Cl2 (1:2), CH2Cl2), recrystallization from CHCl3/pentane at -25°C; | A 9.4% B 88.5% C n/a |

-
-
113270-33-0
methylthiotriphenylphosphonium methylsulfate

-
A
-
74-93-1
methylthiol

-
B
-
75-18-3
dimethylsulfide

-
C
-
603-35-0
triphenylphosphine

-
D
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| With lithium chloride In methanol electrolysis; Further byproducts given; | A n/a B n/a C 86% D n/a |

-
-
16640-68-9
cyanomethylene triphenylphosphorane

-
A
-
3878-45-3
triphenylphosphine sulfide

-
B
-
764-42-1
1,2-Dicyanoethylene

| Conditions | Yield |
|---|---|
| With sulfur In toluene for 3h; Heating; | A 86% B 60% |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane for 20h; Mechanism; other thiophosphoramides are also investigated; | 100% |
| With 3,3-dimethyldioxirane In dichloromethane; acetone for 0.0833333h; Ambient temperature; | 100% |
| With pyridine; trifluoroacetic anhydride for 3h; | 100% |

-
-
3878-45-3
triphenylphosphine sulfide

-
-
460095-62-9
[Rh(1,5-cyclooctadiene)Cl(triphenylphosphinesulfide)]

| Conditions | Yield |
|---|---|
| In dichloromethane under refluxing condition for 30 min; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 3h; | 97% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tris-(trimethylsilyl)silane In toluene at 80℃; for 1h; Product distribution; Further Variations:; Reaction partners; reaction times; | 96% |
| With n In tetrahydrofuran at 60℃; for 3h; | 95% |
| Stage #1: triphenylphosphine sulfide With oxalyl dichloride In toluene at 20℃; for 0.166667h; Inert atmosphere; Stage #2: With lithium aluminium tetrahydride In tetrahydrofuran; toluene at 20℃; for 0.5h; Inert atmosphere; | 88% |
-
-
3878-45-3
triphenylphosphine sulfide

-
-
55344-99-5
triphenylphosphine sulfide diiodine

| Conditions | Yield |
|---|---|
| With iodine In dichloromethane for 4h; | 96% |
| With iodine In carbon disulfide at 20.3℃; Equilibrium constant; |

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane PPh3S in CH2Cl2 added to (Ru(CO)2Cl2)n dissolved in CH3OH (1:1), reaction mixture stirred at room temp. for 2 h; solvent evapd. under vacuum, washed with Et2O, recrystallized from CH2Cl2, elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(acetonitrile)copper(I)tetrafluoroborate; 2,2′‐(1,4‐butanediyl)bis‐1,3‐benzoxazole In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #2: triphenylphosphine sulfide In dichloromethane at 20℃; for 4h; Inert atmosphere; | 94% |

-
-
29892-37-3
chloro(dimethylsulfide) gold(I)

-
-
3878-45-3
triphenylphosphine sulfide

-
-
15279-72-8
chloro(triphenylphosphine sulfide)gold(I)

| Conditions | Yield |
|---|---|
| In dichloromethane excess Ph3PS; concn. (vac.), Et2O addn., crystn. on cooling (refrigerator, overnight),filtration, washing (Et2O), drying (vac.); second crop from filtrate; e lem. anal.; | 93% |

-
-
3878-45-3
triphenylphosphine sulfide

| Conditions | Yield |
|---|---|
| In toluene byproducts: (C5Me5)2, PPh3; (Ar or N2); 3 days, solid PPh3 filtn. off; solvent removing (vac.), product washing (hexane), recrystn. (THF), solvent evapn. gave (C5Me5)2; | 93% |
-
-
102349-56-4
MoI2(CO)3(MeCN)2

-
-
3878-45-3
triphenylphosphine sulfide

-
-
131054-48-3
iodo(tricarbonyl)tris(triphenylphosphinesulfide)molybdenum(II) iodide

| Conditions | Yield |
|---|---|
| In dichloromethane addn. of the ligand to Mo compd. in CH2Cl2 with stirring under N2, stirring for 2 h, addn. of further ligand and stirring for 18 h; filtn., removing of solvent (vac.) and recrystn. from CH2Cl2; elem. anal.; | 91% |
-
-
26042-63-7
silver(I) hexafluorophosphate

-
-
64704-71-8
chloro(dicyclohexylamine)gold(I)

-
-
3878-45-3
triphenylphosphine sulfide

-
-
64736-29-4
Au(P(C6H5)3S)2(1+)*PF6(1-)=[Au(P(C6H5)3S)2]PF6

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: AgCl; stoich. amts. of Au-complex and ligand, 10 % excess of AgPF6; filtration off of AgCl, concn. (vac.), crystn. on Et2O addn. (refrigerator); elem. anal.; | 91% |

Triphenylphosphine sulfide Chemical Properties
IUPAC Name: Triphenyl(sulfanylidene)-$l^{5}-phosphane
The MF of Triphenylphosphine sulfide (CAS NO.3878-45-3) is C18H15PS.
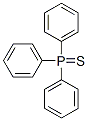
The MW of Triphenylphosphine sulfide (CAS NO.3878-45-3) is 294.35.
Synonyms of Triphenylphosphine sulfide (CAS NO.3878-45-3): Phosphine sulfide, triphenyl- ; Triphenylphosphine sulfide ; Phosphorane, triphenyl-, sulfide
Product Categories: Organic Building Blocks;Phosphorus Compounds;Synthetic Reagents
Apperance: white crystalline solid
Index of Refraction: 1.657
EINECS: 223-407-7
Density: 1.2 g/ml
Flash Point: 213.1 °C
Boiling Point: 428.7 °C
Melting Point: 162-164 °C
BRN: 645872
Triphenylphosphine sulfide Uses
Triphenylphosphine sulfide (CAS NO.3878-45-3) is used as chemical reagent, fine chemicals, organic synthesis reagents, pharmaceutical intermediates.
Triphenylphosphine sulfide Toxicity Data With Reference
| 1. | ivn-mus LD50:180 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#05727 . |
Triphenylphosphine sulfide Safety Profile
A poison by intravenous route. When heated to decomposition it emits toxic vapors of POx and SOx.Safety information of Triphenylphosphine sulfide (CAS NO.3878-45-3):
Hazard Codes  Xn
Xn
Risk Statements
21/22 Harmful in contact with skin and if swallowed
36/37/38 Irritating to eyes, respiratory system and skin
Safety Statements
26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37 Wear suitable protective clothing and gloves
RIDADR UN3464
WGK Germany 3
RTECS SZ2820000
HazardClass 6.1
PackingGroup III
Related Products
- Triphenylphosphine
- Triphenylphosphine hydrobromide
- Triphenylphosphine oxide
- Triphenylphosphine sulfide
- Triphenylphosphine-3,3',3''-trisulfonic acid trisodium salt
- 3878-55-5
- 38786-67-3
- 387867-13-2
- 387868-07-7
- 38787-41-6
- 38789-75-2
- 3879-08-1
- 38791-62-7
- 38792-89-1
- 38795-19-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View