-
Name
TRANS-1,3-PENTADIENE
- EINECS 217-909-5
- CAS No. 504-60-9
- Article Data122
- CAS DataBase
- Density 0.683 g/mL at 25 °C(lit.)
- Solubility
- Melting Point -87 °C(lit.)
- Formula C5H8
- Boiling Point 44.1 °C at 760 mmHg
- Molecular Weight 68.1185
- Flash Point <−30 °F
- Transport Information UN 3295 3
- Appearance colourless liquid
- Safety 16-23-26-36-62-24/25
- Risk Codes 11-65
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xn
Xn
- Synonyms 1-Methyl-1,3-butadiene;1-Methylbutadiene;Piperylene;
- PSA 0.00000
- LogP 1.74850
Synthetic route

| Conditions | Yield |
|---|---|
| at 800℃; under 0.1 Torr; for 2.77778E-06h; Product distribution; Mechanism; var. temp., deuterium substituted 3,5-dimethylpyrazole; | 100% |
| at 456.9℃; under 0.2 - 0.5 Torr; for 2.77778E-05h; Kinetics; Mechanism; Thermodynamic data; Irradiation; various temp., ΔH(excit.); |
-
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

-
-
504-60-9
penta-1,3-diene

| Conditions | Yield |
|---|---|
| With hydrogen bromide; tetrabutyl phosphonium bromide at 200℃; for 0.25h; Menshutkin Reaction; Inert atmosphere; | 99% |
| With monoaluminum phosphate at 450℃; under 35 Torr; | |
| With hydrogen bromide |

| Conditions | Yield |
|---|---|
| With oxygen; Bi-Mo oxide (1/1) at 400℃; Rate constant; Kinetics; also without O2; other temperature; | 99% |
| With multi-component bismuth molybdate at 320℃; |

| Conditions | Yield |
|---|---|
| With borosilicate zeolite B-MWW at 384.84℃; for 48h; Catalytic behavior; Kinetics; Reagent/catalyst; Temperature; Flow reactor; Green chemistry; | 86% |
| With monoaluminum phosphate at 350℃; | |
| With aluminium oxide#titanium oxide at 600℃; under 25 - 40 Torr; | |
| With sodium phosphate at 280℃; | |
| With kaolin at 400℃; under 70 Torr; |

| Conditions | Yield |
|---|---|
| With Ag/Pr/Zr-Si at 460 - 600℃; Reagent/catalyst; Temperature; Inert atmosphere; | 76% |
| With phosphorus at 400℃; Dehydratisierung.; | |
| Multi-step reaction with 2 steps 1: HBr 2: sodium; xylene View Scheme | |
| With VTi2P5.1O(x) at 350℃; for 3h; Catalytic behavior; |

| Conditions | Yield |
|---|---|
| With H3SiW12O40/SBA In dichloromethane at 300℃; Temperature; Reagent/catalyst; | 76% |

| Conditions | Yield |
|---|---|
| With borosilicate zeolite B-MFI at 384.84℃; for 10h; Catalytic behavior; Kinetics; Reagent/catalyst; Temperature; Flow reactor; Green chemistry; | A 76% B n/a |
| With 2-(diisopropylphosphonic acid)terephthalate exchanged zirconium terephthalate based metal-organic framework UiO66 at 280℃; under 4.9 Torr; Reagent/catalyst; Temperature; |

| Conditions | Yield |
|---|---|
| With tungsten trioxide on silica; hydrogen In water at 250℃; for 5h; Concentration; Temperature; Inert atmosphere; | A 19.9% B 72% C 8.6% |

-
-
40749-83-5
4-propenyl-1,3-dioxane

-
-
141-78-6
ethyl acetate

-
A
-
55230-25-6
6-methyl-3,6-dihydro-2H-pyran

-
B
-
504-60-9
penta-1,3-diene

-
C
-
2235-12-3
1,3,5-hexatriene

-
D
-
1516-17-2
2,4-hexadien-1-ol acetate

-
E
-
75338-23-7
3,5-hexadien-1-ol acetate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 90℃; for 9h; metal ampul; | A 17% B 3% C 5% D 20% E 50% |

-
-
78-78-4
methylbutane

-
A
-
513-35-9
2-methyl-but-2-ene

-
B
-
504-60-9
penta-1,3-diene

-
C
-
542-92-7
cyclopenta-1,3-diene

-
D
-
563-46-2
2-Methyl-1-butene

-
E
-
563-45-1
3-Methyl-1-butene

-
F
-
109-66-0
pentane

-
G
-
78-79-5
isoprene

| Conditions | Yield |
|---|---|
| With platinum-aluminum catalyst at 600℃; Gas phase; | A n/a B n/a C n/a D n/a E n/a F n/a G 28.72% |


| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; copper hydroxide In 1-methyl-pyrrolidin-2-one at 180℃; for 5h; Inert atmosphere; Green chemistry; | 14% |

| Conditions | Yield |
|---|---|
| With phosphoric acid at 380 - 400℃; | |
| Abbau durch erschoepfende Methylierung; | |
| Abbau durch erschoepfende Methylierung; dabei findet Verschiebung einer Doppelbildung statt; |

| Conditions | Yield |
|---|---|
| With Lebedew-catalyst |
-
-
71-23-8
propan-1-ol

-
-
64-17-5
ethanol

-
A
-
504-60-9
penta-1,3-diene

-
B
-
106-99-0
buta-1,3-diene

-
C
-
592-46-1
hexa-2,4-diene

| Conditions | Yield |
|---|---|
| ueber Lebedew-Katalysatoren; |


| Conditions | Yield |
|---|---|
| With aluminum oxide at 390℃; |

| Conditions | Yield |
|---|---|
| at 150 - 160℃; beim Leiten ueber einen Mischkatalysator aus Aluminium und Oxiden des Bors, Wolframs, Urans und Silbers; | |
| at 150 - 160℃; beim Leiten ueber einen Mischkatalysator aus Aluminium und Oxiden des Bors, Wolframs, Urans und Silbers; |


| Conditions | Yield |
|---|---|
| With acetic acid 1-methylbut-3-enyl ester at 550 - 570℃; under 70 - 80 Torr; ueber Glaswolle.; | |
| With borosilicate zeolite B-MWW at 334.84℃; Reagent/catalyst; Flow reactor; Green chemistry; |

| Conditions | Yield |
|---|---|
| With aluminium oxide-chromium oxide at 250℃; |

| Conditions | Yield |
|---|---|
| With aluminium oxide-chromium oxide at 250℃; |

| Conditions | Yield |
|---|---|
| With chromium corundum at 250℃; |

| Conditions | Yield |
|---|---|
| With water; magnesium chloride at 300℃; Kupfer aktivierte Katalysatore; |

| Conditions | Yield |
|---|---|
| With Lebedew-catalyst; acetone | |
| With trans-Crotonaldehyde; Lebedew-catalyst |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 100℃; Behandeln des entstandenen Pentens mit Brom in Hexan bei -20grad und Leiten des erhaltenen Dibromids ueber Natronalk bei 500grad.; |

| Conditions | Yield |
|---|---|
| With aluminum oxide; chromium(III) oxide at 600℃; under 130 Torr; |

| Conditions | Yield |
|---|---|
| With water; magnesium chloride at 300℃; Kupfer aktivierte Katalisator.; |

| Conditions | Yield |
|---|---|
| With soda lime; carbonic-acid at 600℃; |

| Conditions | Yield |
|---|---|
| man dampft das Reaktionsprodukt mit verd. Schwefelsaeure auf dem Wasserbad ein; |
-
-
504-60-9
penta-1,3-diene

-
-
870-50-8
di-tert-butyl-diazodicarboxylate

-
-
1072150-92-5
di-tert-butyl 3-methyl-1,2,3,6-tetrahydropyridazine-1,2-dicarboxylate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Diels-Alder reaction; | 100% |
-
-
504-60-9
penta-1,3-diene

-
-
1425040-20-5
tert-butyl 2-(4-chlorophenylcarbamoyl)diazenecarboxylate

| Conditions | Yield |
|---|---|
| With C50H56O4P(1-)*Ag(1+)*H2O In dichloromethane at -40℃; for 4h; Diels-Alder Cycloaddition; Darkness; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at -10 - -5℃; | 98% |
| With hydrogenchloride at -5 - 0℃; | 98% |
| With hydrogenchloride; N,N-dimethyl-formamide at -10 - -5℃; | 98% |
-
-
504-60-9
penta-1,3-diene

-
-
54730-18-6
bromomethanesulfonyl bromide

-
-
102683-77-2
4-bromo-2-pentenyl bromomethyl sulfone

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; | 98% |
-
-
10257-28-0
D-Galactose

-
-
504-60-9
penta-1,3-diene

-
-
81148-33-6, 112677-77-7, 125589-85-7, 125589-86-8
3-acetyl-2-methyl-5-(α,β-D-threofuranosyl)furan

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In ethanol; water for 48h; Heating; | 95% |
-
-
504-60-9
penta-1,3-diene

-
-
79060-54-1
2-phenyl-2,3-((4-methoxyphenyl)imino)-2,3-dihydro-1,4-naphthoquinone

-
-
87373-43-1
3,4-benzo-9-(4-methoxyphenyl)-1-phenyl-8-(1-propenyl)-9-azabicyclo<4.2.1>nonene-2,5-dione

| Conditions | Yield |
|---|---|
| In benzene for 80h; Irradiation; | 95% |
-
-
504-60-9
penta-1,3-diene

-
-
1130728-52-7
(3-methyl-1,4-dioxo-1,4-dihydronaphthalen-2-yl)boronic acid

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 2h; Diels-Alder reaction; | 95% |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
504-60-9
penta-1,3-diene

-
-
4141-59-7
1,2-bis(di-tert-butyl)phosphinoethane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran-d8 at 100℃; for 24h; Inert atmosphere; | 95% |


| Conditions | Yield |
|---|---|
| With C16H18Cl2CoN2O; sodium triethylborohydride In tetrahydrofuran at -30 - 25℃; for 12h; Inert atmosphere; enantioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With bismuth(lll) trifluoromethanesulfonate; tetrakis(actonitrile)copper(I) hexafluorophosphate; 1,2-bis-(diphenylphosphino)ethane In 1,4-dioxane at 50℃; for 18h; | 94% |
-
-
504-60-9
penta-1,3-diene

-
-
19656-74-7
N,N'-di-tert-butyldiaziridinone

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In benzene-d6 at 65℃; Inert atmosphere; | 94% |
-
-
504-60-9
penta-1,3-diene

-
-
1130728-55-0
2,4-dimethyl-3,6-dioxocyclohexa-1,4-dienylboronic acid

-
-
55756-89-3, 97521-93-2, 117661-49-1, 117661-66-2
(4aSR,5RS,8aSR)-2,5,8a-trimethyl-4a,5,8,8a-tetrahydronaphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 1h; Diels-Alder reaction; | 93% |
-
-
504-60-9
penta-1,3-diene

-
-
75927-49-0
pinacol vinylboronate

| Conditions | Yield |
|---|---|
| With (η6-naphthalene)(η4-1,5-cyclooctadiene)ruthenium(0) In benzene at 30℃; for 1h; Inert atmosphere; Schlenk technique; | 93% |

| Conditions | Yield |
|---|---|
| With dodecacarbonyl-triangulo-triruthenium; 2,2'-bis(diphenylphosphino)biphenyl In toluene at 130℃; for 48h; Inert atmosphere; diastereoselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| With bis(trifluoromethane)sulfonimide lithium In methyl cyclohexane at 20℃; Diels-Alder Cycloaddition; Electrochemical reaction; | 91% |

| Conditions | Yield |
|---|---|
| With hydroquinone at 140℃; for 12h; | 90% |
-
-
504-60-9
penta-1,3-diene

-
-
461677-71-4
1-benzyl-3-diazo-1,3-dihydro-2H-indol-2-one

-
-
864949-91-7
1'-(phenylmethyl)-2-[1-prop-1-en-1-yl]spiro[cyclopropane-1,3'-indol]-2'(1'H)-one

| Conditions | Yield |
|---|---|
| With dirhodium tetraacetate In benzene for 4h; Heating; | 90% |
-
-
504-60-9
penta-1,3-diene

-
-
35024-12-5
(4,4-dimethyl-2,6-dioxo-cyclohexyl)-phenyl-iodonium betaine

-
-
92898-20-9
2-(1-propenyl)-4-oxo-6,6-dimethyl-2,3,4,5,6,7-hexahydrobenzofuran

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 1h; Cycloaddition; Irradiation; | 89% |
-
-
504-60-9
penta-1,3-diene

-
-
503312-47-8
methyl 2-(diethoxyphosphoryl)imino-3,3,3-trifluoropropionate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 150h; aza Diels-Alder reaction; | 89% |

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride; triethylphosphine; bis(acetylacetonate)nickel(II) In toluene at 80℃; for 24h; | 89% |
-
-
504-60-9
penta-1,3-diene

-
-
13089-11-7
methyl 3,3,3-trifluoropyruvate

-
-
134224-64-9
6-trifluoromethyl-6-methoxycarbonyl-2-methyl-5,6-dihydro-2H-pyran

| Conditions | Yield |
|---|---|
| In hexane at 20℃; for 24h; | 88% |
-
-
504-60-9
penta-1,3-diene

-
-
139118-84-6
-1-(2-phenylpropylsulfinyl)-2-nitrocyclopentene

| Conditions | Yield |
|---|---|
| With hydroquinone In dichloromethane under 6000480 Torr; for 120h; Ambient temperature; | 88% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide Ambient temperature; | 88% |
-
-
504-60-9
penta-1,3-diene

-
-
84487-52-5
2-(cyclohex-2-en-1-yl)-6-methylaniline

| Conditions | Yield |
|---|---|
| With aluminium trichloride In benzene at 135℃; for 3h; | 88% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In pentane at 20℃; for 2h; | 87% |
-
-
504-60-9
penta-1,3-diene

-
-
418760-99-3
6-methyl-2-(1-cyclohexen-1-yl)aniline

| Conditions | Yield |
|---|---|
| With aluminium trichloride In benzene at 135℃; for 3h; | 87% |
trans-1,3-Pentadiene Consensus Reports
trans-1,3-Pentadiene Specification
The 1,3-Pentadiene, with the CAS registry number 504-60-9, is also known as 1-Methylbutadiene. Its EINECS registry number is 207-995-2. This chemical's molecular formula is C5H8 and molecular weight is 68.117. Its systematic name is called penta-1,3-diene. What's more, this chemical is a clear colorless liquid with an acrid odor. It is mainly used in the manufacture of polyisoprene resin and also can be used in organic synthesis.
Physical properties of 1,3-Pentadiene: (1)ACD/LogP: 2.38; (2)ACD/LogD (pH 5.5): 2.38; (3)ACD/LogD (pH 7.4): 2.38; (4)ACD/BCF (pH 5.5): 38.18; (5)ACD/BCF (pH 7.4): 38.18; (6)ACD/KOC (pH 5.5): 471.91; (7)ACD/KOC (pH 7.4): 471.91; (8)#Freely Rotating Bonds: 1; (9)Index of Refraction: 1.415; (10)Molar Refractivity: 24.99 cm3; (11)Molar Volume: 99.7 cm3; (12)Surface Tension: 18.6 dyne/cm; (13)Density: 0.682 g/cm3; (14)Enthalpy of Vaporization: 27.62 kJ/mol; (15)Boiling Point: 44.1 °C at 760 mmHg; (16)Vapour Pressure: 380 mmHg at 25°C.
Preparation of 1,3-Pentadiene: this chemical can be prepared by 3,5-dimethyl-1H-pyrazole. The reaction time is 0.01 s with reaction temperature of 800 °C. The yield is about 100%. This reaction can be used in product distribution//mechanism.
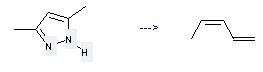
Uses of 1,3-Pentadiene: it can be used to produce pentane at temperature of 29 - 35 °C. This reaction is a kind of hydrogenation. It will need reagents raney nickel and ethanol.

When you are using this chemical, please be cautious about it as the following:
This chemical is highly flammable and harmful. It may cause lung damage if swallowed. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing. If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label.
You can still convert the following datas into molecular structure:
(1)SMILES: C=C\C=CC
(2)InChI: InChI=1/C5H8/c1-3-5-4-2/h3-5H,1H2,2H3
(3)InChIKey: PMJHHCWVYXUKFD-UHFFFAOYAV
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LC50 | inhalation | 1100mg/m3/2H (1100mg/m3) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 101, 1982. | |
| rat | LC50 | inhalation | 140gm/m3/2H (140000mg/m3) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 101, 1982. |
Related Products
- trans-1-(2-Chloroethyl)-3-(3-methyl cyclohexyl)-1-nitrosourea
- trans-1-(4-Butylcyclohexyl)-4-ethoxybenzene
- trans-1-(Bromomethyl)-4-propylcyclohexane
- trans-1-(Methylthio)-2-butene
- trans-1,1,2,3-Tetrachloro-2-propene
- trans-1,2,3,4,4a,10a-Hexahydrophenanthrene
- trans-1,2,5-Trimethylpiperazine
- trans-1,2,-Dihydroxy-1,2-dihydro-triphenylene
- trans-1,2-Bis(diphenylphosphino)ethylene
- trans-1,2-Bis(n-propylsulfonyl)ethylene
- 50461-74-0
- 50461-86-4
- 504-63-2
- 50463-25-7
- 50468-56-9
- 5047-01-8
- 50471-44-8
- 50473-81-9
- 50-47-5
- 504-78-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View