Hebei Kunsui Technology Co., Ltd.
1. A strong scientific research team . 2. Stable quality (with a complete scientific research center and testing center to ensure the quality stability of each batch of products). 3. Rich export experience (with practical experience in exporting to m
Cas:494-19-9
Min.Order:1 Kilogram
FOB Price: $8.2 / 8.8
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:494-19-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:494-19-9
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Cas:494-19-9
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:494-19-9
Min.Order:1
Negotiable
Type:Other
inquiryEnke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api, intermedi
Chemwill Asia Co., Ltd.
Iminodibenzyl CAS 494-19-9 10,11-Dihydro-5H-dibenz[b,f]azepine CAS no 494-19-9 5H-Dibenz (b,f)azepine, 10,11-dihydro- Iminodibenzyl 10,11-Dihydro-5H-dibenz[b,f]azepine 5H-Dibenz (b,f)azepine, 10,11-dihydro- Iminodibenzyl CAS 494-19-9 10,1
Cas:494-19-9
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:494-19-9
Min.Order:1 Kilogram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:494-19-9
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after the dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available &diam
Cas:494-19-9
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
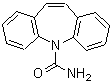
Chemical Name Iminodibenzyl CAS Number 494-19-9 Molecular Formula C14H13N Molecular Weight
Cas:494-19-9
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:494-19-9
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:494-19-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Iminodibenzyl CAS:494-19-9 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates
Cas:494-19-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:494-19-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:494-19-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Cas:494-19-9
Min.Order:1 Kilogram
FOB Price: $15.0 / 25.0
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
HANGZHOU YUNUO CHEMICAL CO.,LTD
superior quality Appearance:YELLOWISH TO BEIGE CRYSTALLINE POWDER Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:10,11-Dihydro-5H-dibenzo[b,f]aze
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Afine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Chemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and als
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:494-19-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Siwei Development Group Ltd.
Product name: Iminodibenzyl CAS No.:494-19-9 Molecule Formula:C14H13N Molecule Weight:195.26 Purity: 99.0% Package: 25kg/drum Description:Pale yellow crystalline powder Manufacture Standards:Enterprise Standard TES
Cas:494-19-9
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Synthetic route
-
-
13080-75-6
5-acetyliminodibenzyl

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With triethyl borane; sodium hydroxide In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; Sealed tube; | 99% |
| Stage #1: 5-acetyliminodibenzyl With Triethoxysilane; sodium triethylborohydride In tert-butyl methyl ether at 80℃; for 6h; Stage #2: With hydrogenchloride In tert-butyl methyl ether; water at 20℃; for 1h; chemoselective reaction; | 99% |
| Multi-step reaction with 2 steps 1: potassium hydroxide; triethyl borane / tetrahydrofuran / 24 h / 25 °C / Inert atmosphere; Schlenk technique; Sealed tube 2: sodium hydroxide; water / tetrahydrofuran / 1 h / 25 °C / Inert atmosphere; Schlenk technique; Sealed tube View Scheme |
-
-
40583-89-9
5-propionyl-10,11-dihydro-5H-dibenzo[b,f]azepine

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With triethyl borane; sodium hydroxide In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; Sealed tube; | 99% |
| Stage #1: 5-propionyl-10,11-dihydro-5H-dibenzo[b,f]azepine With Triethoxysilane; sodium triethylborohydride In tert-butyl methyl ether at 80℃; for 6h; Stage #2: With hydrogenchloride In tert-butyl methyl ether; water at 20℃; for 1h; chemoselective reaction; | 81% |
| Multi-step reaction with 2 steps 1: potassium hydroxide; triethyl borane / tetrahydrofuran / 24 h / 100 °C / Inert atmosphere; Schlenk technique; Sealed tube 2: sodium hydroxide; water / tetrahydrofuran / 1 h / 25 °C / Inert atmosphere; Schlenk technique; Sealed tube View Scheme |

| Conditions | Yield |
|---|---|
| With [Fe3(1,1,1,3,3,3-hexamethyldisilazan-2-ide)4]Fe(η6-toluene); hydrogen at 20℃; under 1425.14 Torr; for 3h; Inert atmosphere; Glovebox; chemoselective reaction; | 92% |
| With tetraethylammonium perchlorate; ammonium chloride In water; N,N-dimethyl-formamide cathodic (Hg, -2.0 V vs. SCE) reduction; | 82% |

| Conditions | Yield |
|---|---|
| With iodine; hypophosphorous acid In acetic acid for 24h; Heating; | A 92% B 8% |
-
-
33948-19-5
10,11-dihydro-dibenzo[b,f]azepine-5-carbonyl chloride

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| In acetonitrile electrolysis (Et4NClO4, Hg-dropelectrode); | 90% |

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With benzyltriethylammonium tetrathiomolybdate In acetonitrile at 25℃; for 0.75h; sonicated; | 88% |

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| Stage #1: C18H19NO With Triethoxysilane; sodium triethylborohydride In tert-butyl methyl ether at 80℃; for 6h; Stage #2: With hydrogenchloride In tert-butyl methyl ether; water at 20℃; for 1h; chemoselective reaction; | 87% |
| With triethyl borane; sodium hydroxide In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; Sealed tube; | 86% |
| Multi-step reaction with 2 steps 1: potassium hydroxide; triethyl borane / tetrahydrofuran / 24 h / 100 °C / Inert atmosphere; Schlenk technique; Sealed tube 2: sodium hydroxide; water / tetrahydrofuran / 1 h / 25 °C / Inert atmosphere; Schlenk technique; Sealed tube View Scheme |

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With C55H46N4O4W; potassium carbonate; N-ethyl-N,N-diisopropylamine In acetonitrile for 12h; Inert atmosphere; Irradiation; | 68% |

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With triethyl borane; sodium hydroxide In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; Sealed tube; | 67% |
| Stage #1: C21H25NO With Triethoxysilane; sodium triethylborohydride In tert-butyl methyl ether at 80℃; for 6h; Stage #2: With hydrogenchloride In tert-butyl methyl ether; water at 20℃; for 1h; chemoselective reaction; | 62% |
| Multi-step reaction with 2 steps 1: potassium hydroxide; triethyl borane / tetrahydrofuran / 24 h / 100 °C / Inert atmosphere; Schlenk technique; Sealed tube 2: sodium hydroxide; water / tetrahydrofuran / 1 h / 25 °C / Inert atmosphere; Schlenk technique; Sealed tube View Scheme |
-
-
13080-75-6
5-acetyliminodibenzyl

-
A
-
494-19-9
9,10-dihydrodibenzazepine

-
B
-
40583-88-8
11-ethyliminodibenzyl

-
C
-
64-17-5
ethanol

| Conditions | Yield |
|---|---|
| With Mo2((1)-H)2(OtBu)6; diphenylsilane In toluene for 25h; Reagent/catalyst; Solvent; Inert atmosphere; Schlenk technique; Reflux; | A 62% B 36% C n/a |


| Conditions | Yield |
|---|---|
| With [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; acetic acid In acetonitrile at 20℃; for 24h; Irradiation; Sealed tube; | A 50% B 22% C 23% |

-
-
256-96-2
dibenzoazepine

-
-
64-19-7
acetic acid

-
A
-
611-64-3
9-methyl-acridine

-
B
-
13080-75-6
5-acetyliminodibenzyl

-
C
-
4217-52-1
9-methyl-9,10-dihydroacridine

-
D
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With iodine; hypophosphorous acid for 96h; Heating; | A 18% B 9% C 20% D 47% |


| Conditions | Yield |
|---|---|
| Stage #1: aniline With copper(l) iodide; n-butyllithium; tetraethylammonium bromide; triethylamine In tetrahydrofuran at -50℃; for 4h; Inert atmosphere; Stage #2: 2-bromophenylacetyl chloride In tetrahydrofuran at 70℃; for 5h; Inert atmosphere; | 45.7% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid; zinc | 45% |

| Conditions | Yield |
|---|---|
| With 2-Picolinic acid; iron(III) chloride; tert-Butyl peroxybenzoate; 18-crown-6 ether In acetonitrile at 50℃; for 48h; | A 6% B 35% C 15% |

-
-
152700-41-9
5-[3-(3,5-Dichloro-2,4,6-trimethyl-phenyl)-4-methylene-4,5-dihydro-isoxazol-5-yl]-10,11-dihydro-5H-dibenzo[b,f]azepine

-
A
-
494-19-9
9,10-dihydrodibenzazepine


| Conditions | Yield |
|---|---|
| With zinc(II) chloride In toluene for 6h; Heating; Further byproducts given; | A 14% B 3% C 33% D 22% |

-
-
113-52-0
Imipramine hydrochloride

-
A
-
50-47-5
desipramine

-
B
-
494-19-9
9,10-dihydrodibenzazepine

-
C
-
796-28-1
10-Hydroxyimipramine

| Conditions | Yield |
|---|---|
| In water Ambient temperature; M. griseo-cyanus (ATCC 1207a); further microbial agents; | A 10 mg B n/a C n/a |

-
-
113-52-0
Imipramine hydrochloride

-
A
-
50-47-5
desipramine

-
B
-
494-19-9
9,10-dihydrodibenzazepine

-
C
-
796-28-1
10-Hydroxyimipramine

-
D
-
303-70-8
2-hydroxyimipramine

| Conditions | Yield |
|---|---|
| In water for 480h; Ambient temperature; C. blakesleeana (ATCC 8688a); further microbial agents; Further byproducts given; | A n/a B n/a C 30 mg D 88 mg |

-
-
113-52-0
Imipramine hydrochloride

-
A
-
494-19-9
9,10-dihydrodibenzazepine

-
B
-
796-28-1
10-Hydroxyimipramine

-
C
-
303-70-8
2-hydroxyimipramine

-
D
-
6829-98-7
imipramine N-oxide

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide for 480h; Ambient temperature; C. blakesleeana (ATCC 8688a); further microbial agents; Further byproducts given; | A n/a B 30 mg C 88 mg D n/a |

-
-
113-52-0
Imipramine hydrochloride

-
A
-
494-19-9
9,10-dihydrodibenzazepine

-
B
-
6829-98-7
imipramine N-oxide

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide for 360h; Ambient temperature; F. oxysporum f. sp. cepae (ATCC 11711); further microbial agents; | A 7 mg B n/a |
| In water for 312h; Ambient temperature; A. flavipes (ATCC 16795); further microbial agents; | A n/a B 112 mg |
-
-
152700-41-9
5-[3-(3,5-Dichloro-2,4,6-trimethyl-phenyl)-4-methylene-4,5-dihydro-isoxazol-5-yl]-10,11-dihydro-5H-dibenzo[b,f]azepine

-
A
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With zinc(II) iodide In toluene for 6h; Heating; |

-
A
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane for 4h; Heating; |

| Conditions | Yield |
|---|---|
| palladium on activated charcoal at 230℃; Rate constant; |
-
-
34124-14-6
2,2'-ethylenedianiline

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| at 265 - 275℃; |

| Conditions | Yield |
|---|---|
| at 280 - 326℃; Kinetics; |
-
-
152700-41-9
5-[3-(3,5-Dichloro-2,4,6-trimethyl-phenyl)-4-methylene-4,5-dihydro-isoxazol-5-yl]-10,11-dihydro-5H-dibenzo[b,f]azepine

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 33 percent / ZnCl2 / toluene / 6 h / Heating 2: aq. HCl / dioxane / 4 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: isoamyl alcohol; sodium 2: 265 - 275 °C View Scheme |

| Conditions | Yield |
|---|---|
| With peroxidase from horseradish type VI; dihydrogen peroxide at 37℃; for 1h; pH=7; aq. phosphate buffer; Enzymatic reaction; |
-
-
16968-19-7
2,2'-dinitrobibenzyl

-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogen 2: hydrogenchloride / Heating View Scheme |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
100527-68-2
2,4,6,8-tetrabromoiminodibenzyl

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; silica gel In dichloromethane at 18℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 100% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
91222-27-4
5-(4-methoxyphenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 100% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Reagent/catalyst; Inert atmosphere; | 91% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 91% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
106-95-6
allyl bromide

-
-
74074-21-8
5-allyl-10,11-dihydro-5H-dibenzoazepine

| Conditions | Yield |
|---|---|
| Stage #1: 9,10-dihydrodibenzazepine With sodium hydride In N,N-dimethyl-formamide for 0.333333h; Inert atmosphere; Stage #2: allyl bromide In N,N-dimethyl-formamide Inert atmosphere; | 99% |
| Stage #1: 9,10-dihydrodibenzazepine With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.333333h; Inert atmosphere; Stage #2: allyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 1h; Inert atmosphere; | 92% |
| 68% | |
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane; water for 72h; | 14% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
623-12-1
4-chloromethoxybenzene

-
-
91222-27-4
5-(4-methoxyphenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 1-methyl-2-(2-(dicyclohexylphosphino)phenyl)-1H-benzoimidazole; sodium t-butanolate In toluene at 20 - 135℃; for 20h; Buchwald-Hartwig reaction; Inert atmosphere; | 99% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Inert atmosphere; | 87% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 87% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
108-90-7
chlorobenzene

-
-
78943-60-9
10,11-dihydro-N-phenyldibenzazepine

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 99% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Inert atmosphere; | 80% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With 1-methyl-2-nitrobenzene; palladium on activated charcoal at 230℃; for 1.5h; Rate constant; Kinetics; Thermodynamic data; var. hydrogen acceptors (also without acceptor); ΔH(excit.), ΔS(excit.); var. temp. and time; | 98.2% |
| With oxygen; 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium nitrite In toluene at 120℃; under 9750.98 Torr; for 8h; | 24% |
| Stage #1: 9,10-dihydrodibenzazepine With N-Bromosuccinimide Stage #2: With pyridine |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
62005-48-5
(acetylamino)acetaldehyde dimethyl acetal

-
-
1262142-12-0
N-(2-(10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)ethyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylsilane; trifluoroacetic acid In dichloromethane at 20℃; for 2h; Inert atmosphere; chemoselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 98% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate In toluene at 100℃; for 20h; Inert atmosphere; Sealed tube; | 81% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 98% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Reagent/catalyst; Inert atmosphere; | 82% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 82% |
| With nickel(II) oxide; potassium tert-butylate; triphenylphosphine In tetrahydrofuran at 100℃; for 24h; Inert atmosphere; Sealed tube; Green chemistry; | 73% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 80℃; for 16h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 80℃; for 16h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 97% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate In toluene at 120℃; for 48h; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| Stage #1: hexanoic acid With sodium tetrahydroborate In toluene at 0 - 5℃; Stage #2: 9,10-dihydrodibenzazepine With sodium tetrahydroborate In toluene at 25 - 80℃; for 5h; Stage #3: With hydrogenchloride In water; isopropyl alcohol | 97% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
40583-96-8
2,8-dibromo-10,11-dihydro-5H-dibenzo[b,f]azepine

| Conditions | Yield |
|---|---|
| With phenyltrimethylammonium tribromide In acetonitrile at 20℃; for 4h; Inert atmosphere; | 96% |
| With N-Bromosuccinimide In chloroform at 0 - 20℃; for 1h; Darkness; | 78% |
| With N-Bromosuccinimide In dichloromethane; N,N-dimethyl-formamide at 20℃; for 1h; | 63% |
| With N-Bromosuccinimide In chloroform at 0 - 25℃; for 2h; Inert atmosphere; | 41% |
| With N-Bromosuccinimide; silica gel In dichloromethane at 18℃; for 0.2h; Yield given; |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 96% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 95% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 96% |
| With tris-(dibenzylideneacetone)dipalladium(0); lithium hexamethyldisilazane; ruphos In 1,4-dioxane at 100℃; for 24h; Inert atmosphere; Schlenk technique; Sealed tube; | 75% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 80℃; for 16h; Inert atmosphere; | 96% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
79-04-9
chloroacetyl chloride

-
-
3534-05-2
2-chloro-1-(10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)ethan-1-one

| Conditions | Yield |
|---|---|
| In toluene at 90℃; for 4h; Inert atmosphere; | 95% |
| In toluene for 5h; Heating; | 53% |
| In toluene Heating; | |
| With benzene |

| Conditions | Yield |
|---|---|
| for 3h; Reflux; | 95% |
| With acetic acid at 110℃; for 2h; | 84% |
-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
106-43-4
para-chlorotoluene

-
-
155162-78-0
5-(4-methylphenyl)-10,11-dihydro-5H-dibenzo[b,f]azepine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 1-methyl-2-(2-(dicyclohexylphosphino)phenyl)-1H-benzoimidazole; sodium t-butanolate In toluene at 20 - 135℃; for 20h; Buchwald-Hartwig reaction; Inert atmosphere; | 94% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Inert atmosphere; | 81% |
| With potassium tert-butylate; copper(II) oxide In dimethyl sulfoxide at 80℃; for 18h; Solvent; Sealed tube; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Reagent/catalyst; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With C30H43O2P*C13H12N(1-)*CH3O3S(1-)*Pd(2+); lithium hexamethyldisilazane In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 94% |
-
-
494-19-9
9,10-dihydrodibenzazepine

| Conditions | Yield |
|---|---|
| With caesium carbonate In dichloromethane at 20℃; for 8h; Inert atmosphere; | 94% |
-
-
110-91-8
morpholine

-
-
50-00-0
formaldehyd

-
-
494-19-9
9,10-dihydrodibenzazepine

-
-
64097-61-6
bis(morpholinomethyl)-2,8 dihydro-10,11 5H dibenzoazepine

| Conditions | Yield |
|---|---|
| With acetic acid for 30h; Heating; | 93% |

Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 Xi
Xi