Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
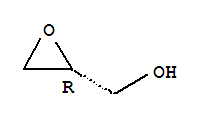
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:57044-25-4
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
Shanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
(R)-(+)-Glycidol Manufacturer Factory CAS 57044-25-4 (R)-(+)-Glycidol Manufacturer High quality Best price In stock factory CAS 57044-25-4 (R)-(+)-Glycidol COA TDS price MSDS Name (R)-(+)-Glycidol Synonyms (R)-3-Hydroxy-1,2-epoxyp
Cas:57044-25-4
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
best price high quality (R)-(+)-Glycidol Basic information Product Name: (R)-(+)-Glycidol Synonyms: (r)-oxiranemethano;(R)-(+)-2,3-EPOXY-1-PROPANOL;(R)-(+)-GLYCIDOL;(R)-GLYCIDOL;R(+)-O
Cas:57044-25-4
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:57044-25-4
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:57044-25-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $62.0 / 76.0
Type:Trading Company
inquiryChangchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram
Cas:57044-25-4
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional chemical raw materials and chemical reagents research and development production enterprises. We have several production line,So we can control the lowest price. We also have several
Cas:57044-25-4
Min.Order:25 Gram
FOB Price: $90.0 / 100.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:57044-25-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:57044-25-4
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:57044-25-4
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
(R)-(+)-Glycidol Chemical Properties Boiling point 56-57 °C11 mm Hg(lit.) density 1.116 g/mL at 20 °C(lit.) refractive index n20/D 1.43(lit.) Fp 178 °F storage temp. 2-8°C pka 14.62±0.10(Predicted) form
Cas:57044-25-4
Min.Order:1 Gram
FOB Price: $66.0
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:57044-25-4
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:57044-25-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Synthetic route
-
-
57090-45-6
(2R)-3-chloro-1,2-propanediol

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane for 18h; Ambient temperature; | 88% |
| With potassium carbonate In dichloromethane other reagent: Cs2CO3; | |
| With potassium phosphate In dichloromethane for 3h; Product distribution / selectivity; Heating / reflux; | |
| With potassium carbonate In dichloromethane at 20℃; for 24h; | |
| With potassium carbonate In dichloromethane at 20℃; for 20h; |
-
-
14437-88-8
(2R)-3-bromo-1,2-propanediol

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane for 18h; Ambient temperature; | 88% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; L-(+)-diisopropyl tartrate; tantalum pentaethoxide In dichloromethane at 0℃; for 48h; Product distribution / selectivity; Molecular sieve; | A 45% B n/a |
| With Cumene hydroperoxide; 3 A molecular sieve; titanium(IV) isopropylate; D-(-)-diisopropyl tartrate In dichloromethane at -5℃; for 6h; Title compound not separated from byproducts; | |
| With titanium(IV) isopropylate; L-(+)-diisopropyl tartrate; Cumene hydroperoxide 1.) CH2Cl2, -10 deg C, 20 min, 2.) CH2Cl2, -10 deg C, 5 h; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
2461-40-7
(+/-)-glycidyl butyrate

-
A
-
60456-26-0
(R)-glycidyl butyrate

-
B
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With sodium phosphate buffer; 25 kDa lipase-like enzyme immobilized on DEAE-Sepharose In 1,4-dioxane; water at 25℃; for 10h; pH=7.00; | A n/a B 45% |
| With thermomyces lanuginosa In 1,4-dioxane; aq. phosphate buffer at 30℃; for 2h; pH=7; Temperature; Resolution of racemate; Enzymatic reaction; | A n/a B n/a |

| Conditions | Yield |
|---|---|
| With water; chiral ((substituted salen)Co)2-InCl3 In tetrahydrofuran at 20℃; for 6h; | A n/a B 44% |
-
-
556-52-5
oxiranyl-methanol

-
A
-
56-81-5
glycerol

-
B
-
57044-25-4
(R)-oxiranemethanol

-
C
-
60456-23-7
(S)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With water; dimeric chiral (salen)Co complex linked with Al at 20℃; for 4h; Product distribution; Further Variations:; Catalysts; | A 42% B n/a C n/a |

-
-
107-18-6
allyl alcohol

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; D-(-)-diisopropyl tartrate; ((CH3)3CCH2)3Ta=CHC(CH3)3 impergnated on silica; tantal content 4.92percent In dichloromethane at 0℃; for 48h; Product distribution / selectivity; | 29% |
| With L-(+)-diisopropyl tartrate; Cumene hydroperoxide; titanium tetraisopropoxide sublimed on silica; titanium content 1.8percent (C/Ti=6.7) In dichloromethane at 0℃; for 48h; Product distribution / selectivity; | 14% |
| With oxygen In N,N-dimethyl-formamide at 50℃; under 760.051 Torr; for 24h; Catalytic behavior; | 12% |
| Sharpless asymmetric epoxidation; | |
| With titanium(IV) isopropylate; tert.-butylhydroperoxide; calcium hydride; diethyl (2R,3R)-tartrate; silica gel In tetrahydrofuran at -20 - -15℃; for 6h; Temperature; Solvent; |
-
-
556-52-5
oxiranyl-methanol

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| 19% | |
| enantioselective resolution; CPO-catalysed oxidation with tert-butyl hydroperoxidase; |
-
-
41274-09-3
(R)-3-tosyloxy-1,2-propanediol

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With sodium methylate In diethyl ether for 1h; | |
| With sodium methylate In methanol; diethyl ether Yield given; | |
| With sodium In methanol | |
| With sodium methylate In diethyl ether |
-
-
110207-31-3
Pentanoic acid oxiranylmethyl ester

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| lipase membran-enclosed enzymatic catalysis; |
-
-
24553-03-5
phenylacetate of 2,3-epoxypropan-1-ol

-
A
-
57044-25-4
(R)-oxiranemethanol

-
B
-
60456-23-7
(S)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With immobilized penicillinacylase from E. coli In water; acetonitrile Ambient temperature; stopped at 50percent conversion; |
-
-
108-05-4
vinyl acetate

-
-
556-52-5
oxiranyl-methanol

-
A
-
65031-95-0
(2S)-oxiran-2-ylmethyl acetate

-
B
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| Lipase P In dichloromethane Ambient temperature; |

-
-
556-52-5
oxiranyl-methanol

-
A
-
115005-76-0
(2R)-oxirane-2-carboxylic acid

-
B
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With potassium phosphate buffer; Acetobacter pesteunanus; potassium hexacyanoferrate(III) Product distribution; other reagents, other C3-alcohol synthons, enantioselectivity of conversion; |

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With formic acid Verseifen des entstandenen Formyl-monochlorhydrins mit Salzsaeure und Behandeln des erhaltenen l-Glycerin-α-monochlorhydrins mit alkoh. Kalilauge; |

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With methanol; diethyl ether; sodium methylate |

| Conditions | Yield |
|---|---|
| With 2-(di(2-hydroxyethyl)amino)ethanesulfonic acid; lipase from Ophiostoma piliferum In water at 25℃; pH=7.20; Enzyme kinetics; |
-
-
115459-65-9
(2S)-(+)-glycidyl-4-nitrobenzoate

-
A
-
57044-25-4
(R)-oxiranemethanol

-
B
-
62-23-7
4-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With 2-(di(2-hydroxyethyl)amino)ethanesulfonic acid; esterase from Streptomyces diastatochromogenes In water at 25℃; pH=7.20; Enzyme kinetics; | |
| With 2-(di(2-hydroxyethyl)amino)ethanesulfonic acid; lipase from Bacillus thermocatenulanatus In water at 40℃; pH=7.20; Enzyme kinetics; |
-
-
106-91-2
2,3-Epoxypropyl methacrylate

-
A
-
79-41-4
poly(methacrylic acid)

-
B
-
57044-25-4
(R)-oxiranemethanol

-
C
-
78196-35-7
(S)-glycidyl methacrylate

-
D
-
130463-96-6
(R)-glycidyl methacrylate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Porcine Pancreas Lipase; type II (E.C. 3.1.1.3) In water at 25℃; for 24h; pH=7.8; Title compound not separated from byproducts; |

-
-
2461-40-7
(+/-)-glycidyl butyrate

-
A
-
60456-26-0
(R)-glycidyl butyrate

-
B
-
57044-25-4
(R)-oxiranemethanol

-
C
-
60456-23-7
(S)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With Rhizopus oryzae lipase on dextran sulfate coated Sepabeads In water at 25℃; pH=7; Enzymatic reaction; Title compound not separated from byproducts; |

-
-
556-52-5
oxiranyl-methanol

-
-
879-18-5
naphthalene-1-carbonic acid chloride

-
A
-
57044-25-4
(R)-oxiranemethanol

-
B
-
60456-23-7
(S)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve; N-ethyl-N,N-diisopropylamine; (S)-1-methyl-2-[(N-benzyl-N-methylamino)methyl]pyrrolidine In dichloromethane; N,N-dimethyl-formamide at -78℃; for 3h; Title compound not separated from byproducts; |

-
-
96-24-2
3-monochloro-1,2-propanediol

-
A
-
57044-25-4
(R)-oxiranemethanol

-
B
-
60456-23-7
(S)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With potassium phosphate; [(R,R)-(salen)Co(II)]2*Al(NO3)3 In tetrahydrofuran at 20℃; for 7h; | |
| With potassium phosphate; (R,R)-[Co(salen)]2*GaCl3 In tetrahydrofuran at 20℃; for 7h; |

| Conditions | Yield |
|---|---|
| In water |
-
-
98760-25-9, 120787-40-8, 121787-43-7, 13443-29-3
oxiran-2-ylmethyl benzoate

-
A
-
121787-43-7
(2S)-oxiran-2-ylmethyl benzoate

-
B
-
57044-25-4
(R)-oxiranemethanol

-
C
-
120787-40-8
(R)-2,3-epoxypropyl benzoate

| Conditions | Yield |
|---|---|
| With candidarugosa In 1,4-dioxane; aq. phosphate buffer at 30℃; for 2h; pH=7; Temperature; Resolution of racemate; Enzymatic reaction; |


| Conditions | Yield |
|---|---|
| With ethanol; porcine pancreas lipase II at 30℃; for 24h; Reagent/catalyst; Enzymatic reaction; Optical yield = 72 %ee; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 25℃; for 24h; | 100% |
| With TEA In dichloromethane at 0 - 20℃; for 24h; Etherification; | 88% |
| With dmap; triethylamine In dichloromethane at 20℃; for 24h; | 88% |
-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
78906-15-7, 114413-26-2, 124150-87-4, 123237-62-7
tert-butyldimethylsilyl (S)-glycidyl ether

| Conditions | Yield |
|---|---|
| With 1H-imidazole; triethylamine In dichloromethane at 0 - 20℃; Inert atmosphere; | 100% |
| With 1H-imidazole In tetrahydrofuran at 20℃; Inert atmosphere; | 100% |
| With 1H-imidazole; dmap In dichloromethane at 0 - 20℃; Inert atmosphere; | 96% |
-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
57044-25-4
(R)-oxiranemethanol

-
-
106731-74-2, 119944-29-5, 107033-46-5
tert-butyl[(2S)-oxiran-2-ylmethoxy]diphenylsilane

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 25℃; | 100% |
| With 1H-imidazole In dichloromethane at 25℃; for 12h; | 100% |
| With 1H-imidazole In dichloromethane at 0 - 25℃; | 99% |
-
-
100-39-0
benzyl bromide

-
-
57044-25-4
(R)-oxiranemethanol

-
-
2930-05-4, 14618-80-5, 89616-40-0, 16495-13-9
(S)-benzyl glycidyl ether

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; | 100% |
| With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; Inert atmosphere; | 100% |
| With tetra-(n-butyl)ammonium iodide; sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 98% |
-
-
40615-36-9
4,4'-dimethoxytrityl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
834906-31-9
(S)-glycidyl 4,4’-dimethoxytrityl ether

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 12h; | 100% |
| With pyridine at 20℃; | 89% |
| In dichloromethane; triethylamine Ambient temperature; | 65% |

| Conditions | Yield |
|---|---|
| With cesium fluoride at 120℃; for 5h; Sealed tube; regioselective reaction; | 100% |
| With cesium fluoride for 5h; Etherification; ring cleavage; Heating; | 99% |
-
-
121-51-7
3-nitrobenzenesulphonyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
115314-14-2
(2s)-(+)-glycidyl 3-nitrobenzenesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; Inert atmosphere; | 100% |
| With triethylamine In dichloromethane at 0℃; | 99% |
| With triethylamine at -20℃; for 96h; | 97% |
-
-
110-91-8
morpholine

-
-
57044-25-4
(R)-oxiranemethanol

-
-
180959-55-1
(2S)-2-hydroxy-3-morpholin-4-yl-propanol

| Conditions | Yield |
|---|---|
| In ethanol at 0 - 140℃; under 9.0009 Torr; for 0.0666667h; Microwave irradiation; | 100% |
| In ethanol at 140℃; for 0.0666667h; Microwave irradiation; | |
| In ethanol at 140℃; for 0.0666667h; Microwave irradiation; |
-
-
57044-25-4
(R)-oxiranemethanol

-
-
438056-69-0
4-(4-aminophenyl)morpholin-3-one

-
-
1447919-65-4
4-[4-{(R)-2,3-dihydroxy-propylamino}phenyl]morpholin-3-one

| Conditions | Yield |
|---|---|
| In methanol for 14h; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With cesium fluoride for 5h; Etherification; ring cleavage; Heating; | 99% |
-
-
3282-30-2
pivaloyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
162825-77-6
(S)-oxiranemethanol trimethylacetate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; for 5h; | 99% |
| With pyridine In dichloromethane at 0 - 20℃; for 5h; | 99% |
-
-
1072791-53-7
6-((R)-5-{3-[(3-fluoro-6-methoxy-[1,5]naphthyridin-4-ylmethyl)-amino]-propyl}-2-oxo-oxazolidin-3-yl)-4H-benzo[1,4]oxazin-3-one

-
-
57044-25-4
(R)-oxiranemethanol

-
-
1072791-54-8
6-((R)-5-{3-[((S)-2,3-dihydroxy-propyl)-(3-fluoro-6-methoxy-[1,5]naphthyridin-4-ylmethyl)-amino]-propyl}-2-oxo-oxazolidin-3-yl)-4H-benzo[1,4]oxazin-3-one

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; methanol at 70℃; for 6h; | 99% |
-
-
994-30-9
triethylsilyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
164031-18-9
(S)-(triethylsilyl)glycidyl ether

| Conditions | Yield |
|---|---|
| With dmap; triethylamine at -30℃; for 0.5h; | 98% |
| With 1H-imidazole In dichloromethane at 20℃; | 94% |
-
-
57044-25-4
(R)-oxiranemethanol

-
-
1066-54-2
trimethylsilylacetylene

-
-
241129-49-7
(S)-5-(trimethylsilyl)pent-4-yne-1,2-diol

| Conditions | Yield |
|---|---|
| With n-butyllithium; boron trifluoride diethyl etherate at -78 - -30℃; for 21h; | 98% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran at -78℃; Metallation; Stage #2: (R)-oxiranemethanol With n-butyllithium In tetrahydrofuran at -78 - 25℃; for 12h; Ring cleavage; Silylation; | |
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; | |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -65℃; for 0.333333h; Stage #2: With boron trifluoride diethyl etherate In tetrahydrofuran; hexane for 0.333333h; Stage #3: (R)-oxiranemethanol In tetrahydrofuran; hexane at 20℃; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 98% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 97% |
| With dmap; triethylamine In dichloromethane at 0℃; for 1.5h; Inert atmosphere; | 94% |
-
-
405-04-9
3-fluoro-4-hydroxybenzonitrile

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 2h; Mitsunobu Displacement; Inert atmosphere; | 98% |
-
-
13154-24-0
triisopropylsilyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
185140-87-8
(2S)-2,3-Epoxy-1-(triisopropylsilyloxy)propane

| Conditions | Yield |
|---|---|
| With 1H-imidazole; dmap In dichloromethane at 20℃; for 1.5h; Substitution; Silylation; | 97% |
| With 1H-imidazole In dichloromethane at 0 - 20℃; for 1.5h; Inert atmosphere; | 97% |
| Stage #1: (R)-oxiranemethanol With 1H-imidazole; dmap In dichloromethane at 0℃; for 0.166667h; Inert atmosphere; Sealed tube; Stage #2: triisopropylsilyl chloride In dichloromethane at 0 - 20℃; for 2.33333h; Inert atmosphere; | 94% |
| With 1H-imidazole; dmap In dichloromethane at 0 - 20℃; | 86% |
| With dmap; triethylamine In dichloromethane for 24h; Ambient temperature; | 81% |
-
-
121-51-7
3-nitrobenzenesulphonyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
152333-94-3
glycidyl nosylate

| Conditions | Yield |
|---|---|
| With triethanolamine | 97% |
| With triethylamine In dichloromethane at 0℃; for 2.5h; |

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 70℃; for 2h; | 96.2% |
-
-
589-15-1
1-bromomethyl-4-bromobenzene

-
-
57044-25-4
(R)-oxiranemethanol

-
-
897961-91-0
(S)-2-(((4-bromobenzyl)oxy)methyl)oxirane

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 96% |
| With tetra-(n-butyl)ammonium iodide; sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 18h; | 12.92 g |

| Conditions | Yield |
|---|---|
| at 70℃; for 2h; | 96% |
-
-
57044-25-4
(R)-oxiranemethanol

-
-
114990-90-8
(S)-oxirane-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With ruthenium trichloride; sodium periodate Oxidation; | 95% |
| With ruthenium trichloride; sodium periodate; water In acetonitrile at 25℃; for 3h; |
-
-
2746-25-0
p-Methoxybenzyl bromide

-
-
57044-25-4
(R)-oxiranemethanol

-
-
80910-01-6, 108836-41-5, 144069-33-0, 134733-19-0
(R)-2-(((4-methoxybenzyl)oxy)methyl)oxirane

| Conditions | Yield |
|---|---|
| Stage #1: (R)-oxiranemethanol With sodium hydride In tetrahydrofuran at 0℃; for 20h; Stage #2: p-Methoxybenzyl bromide In tetrahydrofuran at 20℃; for 3h; | 95% |
-
-
822-39-9
2-chloro-1,3,2-dioxaphospholan

-
-
57044-25-4
(R)-oxiranemethanol

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether at 0 - 20℃; for 3h; Reagent/catalyst; Solvent; | 94.8% |
-
-
106-48-9
4-chloro-phenol

-
-
57044-25-4
(R)-oxiranemethanol

-
-
112652-61-6
(R)-3-(4-chlorophenoxy)propane-1,2-diol

| Conditions | Yield |
|---|---|
| With cesium fluoride In N,N-dimethyl-formamide at 80℃; for 18h; Etherification; ring cleavage; | 94% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 20℃; for 18h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: propargyl bromide With iodine; magnesium; mercury dichloride In diethyl ether; toluene at 0 - 20℃; Inert atmosphere; Stage #2: (R)-oxiranemethanol In diethyl ether; toluene at -78 - 20℃; for 14h; Inert atmosphere; Stage #3: With water; ammonium chloride In diethyl ether; toluene Inert atmosphere; | 94% |
| Stage #1: propargyl bromide With iodine; magnesium; mercury dichloride In diethyl ether; toluene at 0 - 20℃; Grignard Reaction; Inert atmosphere; Stage #2: (R)-oxiranemethanol In diethyl ether; toluene at -78 - 20℃; for 14h; Inert atmosphere; Stage #3: With water; ammonium chloride In diethyl ether; toluene Inert atmosphere; | 94% |
| Stage #1: propargyl bromide With iodine; magnesium; mercury dichloride In diethyl ether; toluene at 0 - 20℃; for 3h; Inert atmosphere; Stage #2: (R)-oxiranemethanol In diethyl ether; toluene at -78 - 20℃; for 15h; Inert atmosphere; regioselective reaction; | 94% |

| Conditions | Yield |
|---|---|
| In diethyl ether at -20℃; for 0.333333h; | 93% |
-
-
57044-25-4
(R)-oxiranemethanol

-
-
1126-20-1
2-allyloxyphenol

-
-
66901-82-4
(R)-2-allyloxy-1-(2,3-dihydroxypropoxy)benzene

| Conditions | Yield |
|---|---|
| With cesium fluoride In N,N-dimethyl-formamide at 80℃; for 8h; Etherification; ring cleavage; | 93% |
-
-
76-86-8
Triphenylsilyl chloride

-
-
57044-25-4
(R)-oxiranemethanol

-
-
473543-52-1
((S)-1-Oxiranylmethoxy)-triphenyl-silane

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 18h; | 93% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 T,
T,  E
E