Hangzhou Dingyan Chem Co., Ltd
Hot selling quality guaranteed 1,3-Indanedione 606-23-5 with reasonable price Our Competitive Advantage: 1.Best quality in your requirement 2.Competitive price in Chi
Cas:606-23-5
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis
Cas:606-23-5
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:606-23-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name: 1,3-Indanedione Synonyms: 1,3-DIOXOINDANE;1,3-DIKETOHYDRINDENE;1,3-Indanedione for synthesis;1H-indene-1,3(2H)-dione-4,5,6,7-d4;1,3-HYDRINDENEDI
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:606-23-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:606-23-5
Min.Order:1 Kilogram
FOB Price: $76.0 / 85.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
1,3-Indanedione CAS:606-23-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediat
Cas:606-23-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Cas:606-23-5
Min.Order:1 Kilogram
FOB Price: $40.0 / 50.0
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:606-23-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:606-23-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
CAS Number: 606-23-5 Molecular formula: C9H6O2 Molecular Weight: 146.14 EINECS number: 210-109-7 Related Categories: Intermediates of Dyes and Pigments; Carbonyl Compounds; Organic Building Blocks; Pigments; General R
Cas:606-23-5
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Henan Wentao Chemical Product Co., Ltd.
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) reasonable & compet
Cas:606-23-5
Min.Order:100 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHenan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:606-23-5
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: 1,3-Indanedione Synonyms: 1,3-DIOXOINDANE;1,3-DIKETOHYDRINDENE;1,3-Indanedione for synthesis;1H-indene-1,3(2H)-dione-4,5,6,7-d4;1,3-HYDRINDENEDIONE;1,3-INDANDIONE;1,3-INDANEDIONE;1,3-Dioxoindan CAS: 606-23-5 MF: C9H6O2 MW:
Cas:606-23-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:606-23-5
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:606-23-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:606-23-5
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Synthetic route

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With Shvo's Catalyst In acetone at 35℃; for 20h; Air; | A n/a B 95% |
-
-
26976-59-0
3-hydroxy-2,3-dihydro-1H-inden-1-one

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With Jones reagent In acetone at 20℃; for 2h; | 95% |
-
-
60414-82-6
2,3-dihydro-1H-indene-1,3-diol

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In 1,4-dioxane at 20℃; for 0.333333h; | 92% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In nitrobenzene at 80℃; for 5h; Mechanism; other aromatic acyl chlorides; | 90% |
| With aluminium trichloride In nitrobenzene at 80℃; for 5h; | 90% |
-
-
117560-06-2
3-phenyliodonio-1,2,4-trioxo-1,2,3,4-tetrahydronaphthalenide

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| In acetonitrile for 1h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: phthalic acid dimethyl ester; ethyl acetate With sodium methylate at 12 - 80℃; Large scale; Stage #2: With hydrogenchloride In water at 45 - 55℃; for 5h; Large scale; | 83.2% |

| Conditions | Yield |
|---|---|
| With manganese(III) Schiff-base; dihydrogen peroxide at 20℃; for 5h; | A 14% B 83% |
| With C26H30F6MnN6O6S2; dihydrogen peroxide; acetic acid In acetonitrile at 20℃; for 1h; chemoselective reaction; | A 9% B 61% |

| Conditions | Yield |
|---|---|
| Stage #1: phthalic acid dimethyl ester; acetic acid With sodium methylate at 12 - 80℃; Stage #2: With hydrogenchloride In water at 45 - 55℃; for 5h; | 82.7% |

-
-
107428-05-7
dimethyl 2,2-di(but-2-yn-1-yl)malonate

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With calcium hydride; 1,5-hexadienerhodium(I)-chloride dimer; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In 1,2-dichloro-ethane at 80℃; for 15h; Schlenk technique; Inert atmosphere; | A 10% B 76% |


| Conditions | Yield |
|---|---|
| With sodium percarbonate; adogen 464; bis-(tributyltin oxide) dioxochromium(VI); toluene-4-sulfonic acid In acetonitrile at 80 - 83℃; for 20h; | A 4% B 5% C 70% |
| With sodium percarbonate; adogen 464; bis-(tributyltin oxide) dioxochromium(VI) In acetonitrile at 80 - 83℃; for 20h; Product distribution; benzylic oxidation with various reagents, solvent dependence; | A 2% B 10% C 60% |
| With dipyridinium dichromate; sodium percarbonate; adogen 464 In 1,2-dichloro-ethane at 80 - 83℃; for 20h; | A 6% B 7% C 30% |


| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; N-hydroxyphthalimide In acetonitrile at 80℃; for 24h; Schlenk technique; | 61% |
| With tert.-butylnitrite; N-hydroxyphthalimide; oxygen In acetonitrile at 80℃; under 760.051 Torr; for 24h; Schlenk technique; | 61% |
-
-
1807-62-1
2-(9-xanthylidene)indane-1,3-dione

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
87688-44-6
2-(9-xanthyl)indane-1,3-dione

| Conditions | Yield |
|---|---|
| With thiophenol at 100℃; for 10h; Product distribution; other reagent; | A 12% B 55% |
-
-
107428-05-7
dimethyl 2,2-di(but-2-yn-1-yl)malonate

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With silver tetrafluoroborate; chloro(1,5-cyclooctadiene)rhodium(I) dimer; calcium hydride; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In 1,2-dichloro-ethane at 80℃; Schlenk technique; Inert atmosphere; | A 27% B 14% C 45% |
| With calcium hydride; bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In 1,2-dichloro-ethane at 80℃; Reagent/catalyst; Schlenk technique; Inert atmosphere; | A 27% B 32% C 40% |
| With calcium hydride; bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In 1,2-dichloro-ethane at 80℃; Schlenk technique; Inert atmosphere; | A 10% B 39% C 12% |


| Conditions | Yield |
|---|---|
| With dipyridinium dichromate; adogen 464; dihydrogen peroxide; sodium carbonate In various solvent(s) for 24h; Oxidation; Heating; | A 18% B 38% |

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide at 90℃; for 1h; Inert atmosphere; | 35% |
-
-
14570-43-5
2-propylindan-1,3-dione

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| In acetonitrile for 4h; UV-irradiation; | 28% |
-
-
496-11-7
INDANE

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
26976-59-0
3-hydroxy-2,3-dihydro-1H-inden-1-one

-
C
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; [(pymox-Me2)2RuCl2]+BF4- In water at 20℃; for 2h; | A 5% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
7319-63-3
2-bromo-indan-1,3-dione

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
1685-97-8
2,2-dibromo-indan-1,3-dione

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide Uebersaettigen der Loesung mit Schwefelsaeure; |
-
-
103165-42-0
2,2-bis-(1,3-dioxo-indan-2-yl)-acenaphthen-1-one

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
850645-51-1
2-(2-oxo-acenaphthen-1-ylidene)-indan-1,3-dione

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
87768-18-1
3-methoxy-1-oxo-indene-2-carboxylic acid ethyl ester

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| at 100℃; |
-
-
74307-85-0
2-bromo-1,3-dioxo-indan-2-carboxylic acid ethyl ester

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
1685-97-8
2,2-dibromo-indan-1,3-dione

| Conditions | Yield |
|---|---|
| With water |

-
-
606-23-5
1H-indene-1,3(2H)-dione
-
-
15365-99-8
1-(1,3-dioxo-indan-2-yl)-4-(1,3-dioxo-indan-2-ylidene)-3,4-dihydro-2H-pyrrolium betaine

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| at 210 - 220℃; under 0.1 Torr; |
-
-
25299-18-7
2‑(2‑hydroxybenzylidene)‑1H‑indene‑1,3(2H)‑dione

-
-
100-63-0
phenylhydrazine

-
A
-
606-23-5
1H-indene-1,3(2H)-dione

-
B
-
90-02-8
salicylaldehyde


| Conditions | Yield |
|---|---|
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| at 1100℃; Mechanism; lower temperature; | |
| at 1100℃; flash thermolysis; Yield given; | |
| With potassium tert-butylate In tetrahydrofuran |
-
-
28164-53-6
2-diazo-3-hydroxy-1-indanone

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| tin(ll) chloride In dichloromethane Ambient temperature; |
-
-
19968-95-7
1,3-indandione anion

-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With 4-chlorobenzylmalononitrile; potassium chloride In water; dimethyl sulfoxide at 20℃; Rate constant; var. phenyl-substituted benzylmalononitriles; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dihydrogen peroxide; potassium bromide In toluene at 20℃; for 0.5h; | 100% |
| With hydroxy(tosyloxy)iodobenzene; copper(ll) bromide In acetonitrile at 0℃; for 0.0833333h; | 90% |
| With bromomalononitrile In N,N-dimethyl-formamide at 20℃; for 0.5h; Reagent/catalyst; | 90% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
551-93-9
2-aminoacetophenone

-
-
142790-83-8
10-methyl-11H-indeno[1,2-b]quinolin-11-one

| Conditions | Yield |
|---|---|
| With acetic acid at 130℃; for 4h; | 100% |
| With malic acid In neat (no solvent) at 70℃; for 2.4h; Friedlaender Quinoline Synthesis; Green chemistry; | 81% |
| With water; acetic acid at 130℃; for 4h; | 50% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
1620-98-0
3,5-di-t-butyl-4-hydroxybenzaldehyde

-
-
53566-09-9
(Z)-2-(3,5-di-tert-butyl-4-hydroxybenzylidene)-1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In chloroform for 21h; Knoevenagel reaction; Heating; | 100% |
| With sulfuric acid In acetic acid for 51h; Ambient temperature; | 75% |
| With piperidine In ethanol for 8h; Knoevenagel Condensation; Reflux; | 51.5% |
| With piperidine In acetic acid |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
234762-41-5
(Z)-5a,6,7,9a-Tetrahydro-5a,6-tetramethylene-11-[ethoxycarbonyl(nitro)methylene]naphtho[1',2':4,5]imidazo[1,2-a]pyridine

| Conditions | Yield |
|---|---|
| With sodium carbonate In N,N-dimethyl-formamide at 20℃; for 24h; methylene exchange; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indene-1,3(2H)-dione; 4-chlorobenzaldehyde With L-proline In methanol at 20℃; for 0.5h; organocatalytic Knoevenagel condensation; Stage #2: (E)-benzalacetone In methanol at 25℃; for 96h; Diels-Alder/epimerization reaction; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 1H-indene-1,3(2H)-dione; 1-naphthaldehyde With L-proline In methanol at 20℃; for 0.5h; organocatalytic Knoevenagel condensation; Stage #2: (E)-benzalacetone In methanol at 25℃; for 96h; Diels-Alder/epimerization reaction; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 1H-indene-1,3(2H)-dione; 4-hydroxy-benzaldehyde With L-proline In methanol at 20℃; for 0.5h; organocatalytic Knoevenagel condensation; Stage #2: (E)-benzalacetone In methanol at 25℃; for 96h; Diels-Alder/epimerization reaction; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 1H-indene-1,3(2H)-dione; 4-cyanobenzaldehyde With L-proline In methanol at 20℃; for 0.5h; organocatalytic Knoevenagel condensation; Stage #2: (E)-benzalacetone In methanol at 25℃; for 96h; Diels-Alder/epimerization reaction; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 1H-indene-1,3(2H)-dione; methyl 4-formylbenzoate With L-proline In methanol at 20℃; for 0.5h; organocatalytic Knoevenagel condensation; Stage #2: (E)-benzalacetone In methanol at 25℃; for 96h; Diels-Alder/epimerization reaction; | 100% |

-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
99-61-6
3-nitro-benzaldehyde

-
-
18691-32-2
3′-(3-nitrophenyl)dispiro[indene-2,1′-cyclopropane-2′,2′′-indene]-1,1′′,3,3′′-tetraone

| Conditions | Yield |
|---|---|
| With bromocyane; triethylamine In methanol at 0 - 20℃; for 0.00416667h; Sealed tube; | 100% |
| With N,N,N’,N’-tetrabromobenzene-1,3-disulfonamide; sodium acetate In ethanol at 20℃; for 0.0333333h; Reagent/catalyst; | 96% |
| With dmap; iodine at 20℃; for 1h; Neat (no solvent); Mechanical ball-milling; chemospecific reaction; | 88% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
555-16-8
4-nitrobenzaldehdye

-
-
18691-31-1
3'-(4-nitrophenyl)-dispiro[indan-2,1'-cyclopropane-2',2''-indan]-1,1'',3,3''-tetrone

| Conditions | Yield |
|---|---|
| With bromocyane; triethylamine In methanol at 0 - 20℃; for 0.00416667h; Sealed tube; | 100% |
| With dmap; iodine at 20℃; for 1h; Neat (no solvent); Mechanical ball-milling; chemospecific reaction; | 89% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
874-42-0
2,4-dichlorobenzaldeyhde

-
-
7090-75-7
3′-(2,4-dichlorophenyl)dispiro[indene-2,1′-cyclopropane-2′,2′′-indene]-1,1′′,3,3′′-tetraone

| Conditions | Yield |
|---|---|
| With bromocyane; triethylamine In methanol at 0 - 20℃; for 0.00416667h; Sealed tube; | 100% |
| With N,N,N’,N’-tetrabromobenzene-1,3-disulfonamide; sodium acetate In ethanol at 20℃; for 0.166667h; Reagent/catalyst; | 96% |
-
-
50-00-0
formaldehyd

-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
106144-67-6
dispiro[indene-2,1'-cyclopropane-2',2''-indene]-1,1'',3,3''-tetraone

| Conditions | Yield |
|---|---|
| With bromocyane; sodium ethanolate In methanol at 0 - 20℃; for 0.00416667h; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid In water at 20℃; Inert atmosphere; | 99.8% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
555-16-8
4-nitrobenzaldehdye

-
-
15875-60-2
2-(p-nitrobenzylidene)indan-1,3-dione

| Conditions | Yield |
|---|---|
| With L-proline In methanol at 25℃; for 12h; organocatalytic Knoevenagel condensation; | 99% |
| In water at 20 - 80℃; Knoevenagel condensation; | 97.5% |
| With magnesium oxide at 20℃; for 0.333333h; Knoevenagel condensation; | 94% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
93-02-7
2,5-dimethoxybenzaldehyde

-
-
67200-96-8
2-(2,5-dimethoxybenzylidene)-1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid In ethanol; water at 50 - 60℃; Reagent/catalyst; Solvent; | 99% |
| Stage #1: 1H-indene-1,3(2H)-dione With pyridine Stage #2: 2,5-dimethoxybenzaldehyde at 100℃; for 1h; | 75% |
| With ethanol |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
95-01-2
2,4-Dihydroxybenzaldehyde

-
-
78426-08-1
2-(2,4-dihydroxybenzylidene)-1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With pyridine In ethanol Reflux; | 99% |
| In ethanol for 3h; Heating; | 72% |
| With hydrogenchloride; acetic acid In water at 20℃; Inert atmosphere; | 43.1% |
| With piperidine; ethanol | |
| With potassium hydroxide bei der Kondensation; |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
106-96-7
propargyl bromide

-
-
24241-98-3
2,2-di(prop-2-yn-1-yl)-1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetone; toluene at 20℃; for 16h; | 99% |
| With potassium hydroxide In water; acetone at 2440℃; for 16h; Reflux; | 93% |
| With potassium hydroxide In water at 40℃; for 24h; | 93% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

- poly{oxy[(E)-but-2-enylene](1,3-indandione-2,2-diyl)[(E)-but-2-enylene]}, Mn=14000; monomer(s): (2Z,7Z)-non-2,7-diene-5-oxa-1,9-ol dibenzoic acid ester; 1,3-indane-2,4-diene
-
poly{oxy[(E)-but-2-enylene](1,3-indandione-2,2-diyl)[(E)-but-2-enylene]}, Mn=14000; monomer(s): (2Z,7Z)-non-2,7-diene-5-oxa-1,9-ol dibenzoic acid ester; 1,3-indane-2,4-diene

| Conditions | Yield |
|---|---|
| With N,O-bis-(trimethylsilyl)-acetamide; tris(dibenzylideneacetone)dipalladium (0); 1,4-di(diphenylphosphino)-butane In dichloromethane; N,N-dimethyl-formamide Tsuji-Trost reaction; | 99% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
107-18-6
allyl alcohol

-
-
81055-89-2
2,2-diallyl-1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With trisodium tris(3-sulfophenyl)phosphine; sodium carbonate; bis(η3-allyl-μ-chloropalladium(II)) In water; ethyl acetate at 20℃; for 18h; Tsuji-Trost reaction; | 99% |
| With Ru(Cp*)(η3-C3H5)(p-CH3C6H5SO3)2 In dichloromethane; acetonitrile at 50℃; for 3h; regioselective reaction; | 89% |

| Conditions | Yield |
|---|---|
| With 1,2-diamino-benzene; rac-Pro-OH In ethanol at 25℃; for 1h; | 99% |
| With 2,6-dimethyl-1,4-dihydropyrimidine-3,5-dicarboxylic acid diethyl ester; L-proline In ethanol at 25℃; Knoevenagel/hydrogenation reaction; | 88% |
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; L-proline In ethanol at 25℃; for 26h; cascade Knoevenagel olefination/hydrogenation reaction; Combinatorial reaction / High throughput screening (HTS); chemoselective reaction; | 88% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 20℃; for 24h; | 99% |
-
-
106518-84-7
3-(4-bromophenylamino)-5,5-dimethylcyclohex-2-enone

-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
89-98-5
2-chloro-benzaldehyde

-
-
1009033-46-8
10-(4-bromophenyl)-5-(2-chlorophenyl)-7,8-dihydro-7,7-dimethyl-5H-indeno[1,2-b]quinolin-9,11(6H,10H)-dione

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium Tetrafluoroborate at 90℃; for 8h; | 99% |

-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
18940-21-1
5,5-dimethyl-3-(phenylamino)cyclohex-2-en-1-one

-
-
552-89-6
2-nitro-benzaldehyde

-
-
1009033-44-6
7,8-dihydro-7,7-dimethyl-5-(2-nitrophenyl)-10-phenyl-5H-indeno[1,2-b]quinolin-9,11(6H,10H)-dione

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium Tetrafluoroborate at 90℃; for 6h; | 99% |

-
-
1603-41-4
(5-methyl-pyridin-2-yl)amine

-
-
606-23-5
1H-indene-1,3(2H)-dione

-
-
61066-33-9, 61066-34-0, 61066-35-1, 61127-23-9
pyrimidine-2,4,5,6(1H,3H)-tetraone

-
-
1367748-11-5
5-(1,3-dioxo-2,3-dihydro-1H-inden-2-yl)-5-hydroxypyrimidine-2,4,6(1H,3H,5H)-trione 2-amino-5-methylpyridinium salt

| Conditions | Yield |
|---|---|
| In chloroform for 1h; Reflux; | 99% |


| Conditions | Yield |
|---|---|
| With aluminum oxide In dichloromethane at 25℃; for 12h; Knoevenagel Condensation; | 99% |

| Conditions | Yield |
|---|---|
| With aluminum oxide In dichloromethane at 25℃; for 12h; Knoevenagel Condensation; | 99% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In 1,2-dichloro-ethane at 60℃; for 12h; Solvent; Molecular sieve; diastereoselective reaction; | 99% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In 1,2-dichloro-ethane at 60℃; for 12h; Molecular sieve; diastereoselective reaction; | 99% |
-
-
606-23-5
1H-indene-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| With magnesium sulfate; 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 20℃; for 24h; Inert atmosphere; Schlenk technique; | 99% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
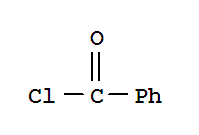













 Xi
Xi