-
Name
Tetrachloropropene
- EINECS
- CAS No. 10436-39-2
- Article Data33
- CAS DataBase
- Density 1.539g/cm3
- Solubility MISCIBLE
- Melting Point 29-31°C(lit.)
- Formula C3H2 Cl4
- Boiling Point 173.8°Cat760mmHg
- Molecular Weight 179.861
- Flash Point 62.3°C
- Transport Information
- Appearance clear colorless to yellowish liquid after melting
- Safety Poison by ingestion and skin contact. Moderately toxic by inhalation. Experimental reproductive effects. Mutation data reported. When heated to decomposition it emits toxic fumes of Cl−. See also CHLORINATED HYDROCARBONS, ALIPHATIC.
- Risk Codes R37/38; R41; R36/37/38; R34
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, C
C
- Synonyms Propene,1,1,2,3-tetrachloro- (6CI,7CI,8CI); 1,1,2,3-Tetrachloro-1-propene;1,1,2,3-Tetrachloropropene; 2,3,3-Trichloroallyl chloride; HCO 1230; HCO 1230xa
- PSA 0.00000
- LogP 3.11070
Synthetic route
-
-
16714-68-4
1,1,2,2,3-pentachloropropane

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cetylpyridinium bromide at 130℃; for 2h; | 97% |
| With potassium hydroxide | |
| With sodium hydroxide |
-
-
24425-97-6
1,1,1,2,2,3-hexachloropropane

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| With hydrogenchloride; zinc In 1,4-dioxane; water at 45℃; for 49h; Autoclave; Inert atmosphere; | 90% |
-
-
1070-78-6
1,1,1,3-tetrachloropropane

-
A
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
B
-
2567-14-8
3,3-dichloroallyl chloride

-
C
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| With iron(III) chloride; chlorine at 82℃; under 1277.21 Torr; for 15.33h; Product distribution / selectivity; | A 82.9% B 3.4% C 8.1% |

-
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
A
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
B
-
16500-91-7
2,3,3,3-tetrachloropropene

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 0℃; | |
| With potassium hydroxide; Aliquat 336 In water at 80℃; for 3h; | |
| With potassium hydroxide; Aliquat 336 In water at 80℃; for 3h; | |
| With potassium hydroxide; Aliquat 336 In water at 8℃; for 3h; |
-
-
16500-91-7
2,3,3,3-tetrachloropropene

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| at 180℃; | |
| With aluminium trichloride at 0℃; | |
| With antimonypentachloride | |
| at 150 - 180℃; |
-
-
67-56-1
methanol

-
-
3266-39-5
2,3,3-trichloro-2-propen-1-ol

-
A
-
80-63-7
methyl 2-chloroacrylate

-
B
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| With sulfuric acid; water 1) 105 deg C, 4 h, 2) 30 min; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
A
-
2233-00-3
3,3,3-trichloro-1-propene

-
B
-
10436-39-2
1,1,2,3-Tetrachloropropene

| Conditions | Yield |
|---|---|
| at 400℃; under 750.075 Torr; for 0.0130556h; Inert atmosphere; |
-
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
A
-
2252-87-1
2,3-dichloro-3,3-difluoroprop-1-ene

-
B
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
C
-
666-27-3
1,1,2,3-tetrachloro-1-fluoro-propane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride at 290℃; under 750.075 Torr; for 3h; Inert atmosphere; tubular reactor; |


| Conditions | Yield |
|---|---|
| Stage #1: prop-1-yne With chlorine at 60 - 80℃; Stage #2: With chlorine; dibenzoyl peroxide at 40 - 50℃; Stage #3: With sodium hydroxide at 40 - 50℃; |

| Conditions | Yield |
|---|---|
| at 100 - 450℃; under 1018.63 Torr; Temperature; Reagent/catalyst; Pressure; Inert atmosphere; | 8.1 %Spectr. |
-
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
A
-
2730-62-3
2-chloro-1,1,1-trifluoropropene

-
B
-
677-21-4
1,1,1-trifluoropropylene

-
C
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
D
-
16500-91-7
2,3,3,3-tetrachloropropene

-
E
-
421-42-1
2,3,3-trichloro-3-fluoro-propene

| Conditions | Yield |
|---|---|
| With hydrogen fluoride at 350℃; under 750.075 Torr; Temperature; | A 77 %Chromat. B 11 %Chromat. C 42.7 %Spectr. D 22.3 %Spectr. E 6.6 %Spectr. |

-
-
21700-31-2
1,1,1,2,3-pentachloro-propane

-
A
-
2730-62-3
2-chloro-1,1,1-trifluoropropene

-
B
-
677-21-4
1,1,1-trifluoropropylene

-
C
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
D
-
421-42-1
2,3,3-trichloro-3-fluoro-propene

-
E
-
666-27-3
1,1,2,3-tetrachloro-1-fluoro-propane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride at 275℃; under 750.075 Torr; | A 80 %Chromat. B 13 %Chromat. C 53.3 %Spectr. D 6.9 %Spectr. E 6.9 %Spectr. |

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
100-97-0
hexamethylenetetramine

| Conditions | Yield |
|---|---|
| In chloroform at 60℃; for 12h; | 99.6% |
-
-
110-85-0
piperazine

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
137554-28-0
N,N'-bis(1,2,2-trichloroallyl)piperazine

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 90 - 95℃; for 4h; | 86.8% |
-
-
5464-28-8
1,3-dioxolane-4-methanol

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
127719-54-4
4-(2,3,3-Trichloro-allyloxymethyl)-[1,3]dioxolane

| Conditions | Yield |
|---|---|
| With potassium hydroxide; 18-crown-6 ether for 4h; Ambient temperature; | 80% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
42967-74-8
dithiobenzoic acid triethylammonium salt

-
-
154424-51-8
dithiobenzoic acid β,γ,γ-trichloroallyl ester

| Conditions | Yield |
|---|---|
| In acetone | 78% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
2730-62-3
2-chloro-1,1,1-trifluoropropene

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimonypentachloride; chlorine; 1-ethyl-3-methyl-1H-imidazol-3-ium chloride In 1,1,2-trichloro-2,2-difluoroethane at 132.1℃; under 6000.6 Torr; Product distribution / selectivity; | 72.6% |
| With hydrogen fluoride; diisopropylamine; chromium(III) oxide at 200 - 300℃; under 760.051 Torr; for 0.000569444 - 0.00277778h; Product distribution / selectivity; | |
| With methoxyhydroquinone; hydrogen fluoride; chromium(III) oxide at 243 - 254℃; under 760.051 Torr; for 66.75h; Product distribution / selectivity; |
-
-
140-31-8
aminoethylpiperazine

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
137554-29-1
N-(1,1,2-trichloroallyl)-N'-(β-1,2,2-trichloroallylaminoethyl)piperazine

| Conditions | Yield |
|---|---|
| In isopropyl alcohol Heating; | 70% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
930-69-8
sodium thiophenolate

-
-
38491-40-6
phenyl-(2,3,3-trichloro-allyl)-sulfide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 70% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
111-40-0
1,5-diamino-3-azapentane

-
-
137554-22-4
N,N'-bis(1,2,3-trichloroallyl)diethylenetriamine

| Conditions | Yield |
|---|---|
| In isopropyl alcohol Heating; | 65% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
431-31-2
1,1,1,2,3-pentafluoropropane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimonypentachloride at 150℃; under 47821.8 Torr; for 8h; Product distribution / selectivity; | 59% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
A
-
2730-62-3
2-chloro-1,1,1-trifluoropropene

-
B
-
421-73-8
2-chloro-1,1,1,2-tetrafluoropropane

-
C
-
431-31-2
1,1,1,2,3-pentafluoropropane

-
D
-
338-75-0
2,3-dichloro-1,1,1-trifluoropropane

-
E
-
151771-09-4
1-chloro-2,3,3,3-tetrafluoropropane

-
F
-
117970-90-8
2-chloro-1,1,1,3-tetrafluoropropane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimonypentachloride at 100 - 125℃; under 33082.7 - 39185.2 Torr; for 12h; Product distribution / selectivity; | A n/a B n/a C 53% D n/a E n/a F n/a |

-
-
106-44-5
p-cresol

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
106119-06-6
4-methyl-2-(2,3,3-trichloro-2-propenyl)phenol

| Conditions | Yield |
|---|---|
| 31% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
A
-
2730-62-3
2-chloro-1,1,1-trifluoropropene

-
B
-
374-06-1
2-chloro-2,3,3-trifluoro-butane

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; antimonypentachloride; chlorine; 1-ethyl-3-methyl-1H-imidazol-3-ium chloride In 1,1,2-trichloro-2,2-difluoroethane at 133.2℃; under 6000.6 Torr; Product distribution / selectivity; | A 25.5% B 6.6% |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
124-41-4
sodium methylate

-
-
63578-64-3
methyl-(2,3,3-trichloro-allyl)-ether

| Conditions | Yield |
|---|---|
| With methanol |

| Conditions | Yield |
|---|---|
| With chlorine at 400℃; under 36775.4 Torr; |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
6066-14-4
3-bromo-1,1,2-trichloro-propene

| Conditions | Yield |
|---|---|
| With acetone; sodium bromide | |
| With hydrogen bromide; iron(III) chloride |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
21400-25-9
1,1,2-trichloropropene

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; lithium aluminium tetrahydride | |
| With ethanol; zinc |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
24425-97-6
1,1,1,2,2,3-hexachloropropane

| Conditions | Yield |
|---|---|
| With chlorine Irradiation.mit UV-Licht; |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
3266-39-5
2,3,3-trichloro-2-propen-1-ol

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
17078-21-6
1,1,2-trichloro-3-phenyl-propene

| Conditions | Yield |
|---|---|
| With aluminium trichloride; benzene |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
109-89-7
diethylamine

-
-
118378-57-7
diethyl-(2,3,3-trichloro-allyl)-amine

| Conditions | Yield |
|---|---|
| With methanol |
-
-
110-91-8
morpholine

-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
15111-02-1
4-(2,3,3-trichloro-allyl)-morpholine

| Conditions | Yield |
|---|---|
| In methanol Heating; |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
141-52-6
sodium ethanolate

-
-
15111-07-6
1,1,2-trichloro-3-ethoxy-propene

| Conditions | Yield |
|---|---|
| With phenol In ethanol |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
540-72-7
sodium thiocyanide

-
-
3266-21-5
2,3,3-Trichlor-allyl-thiocyanat

| Conditions | Yield |
|---|---|
| In ethanol |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
996-82-7
sodium diethylmalonate

-
-
31000-94-9
Bis(β.γ.γ-trichlorallylmalonic Acid)-Diethyl Ester

| Conditions | Yield |
|---|---|
| In ethanol |
-
-
10436-39-2
1,1,2,3-Tetrachloropropene

-
-
996-82-7
sodium diethylmalonate

-
-
31000-93-8
β.γ.γ-Trichlorallylmalonic Acid-Diethyl Ester

| Conditions | Yield |
|---|---|
| In ethanol |
1,1,2,3-Tetrachloropropene Chemical Properties
IUPAC Name: 1,1,2,3-Tetrachloroprop-1-ene
The MF of 1,1,2,3-Tetrachloropropene (CAS NO.10436-39-2) is C3H2Cl4.
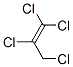
The MW of 1,1,2,3-Tetrachloropropene (CAS NO.10436-39-2) is 179.86.
Synonyms of 1,1,2,3-Tetrachloropropene (CAS NO.10436-39-2): Tetrachloropropene ; 1,1,2,3-Tetrachloropropene ; 1,1,2,3-Tetrachloro-1-propene ; 1,1,2,3-Tetrachloro-propen ; 2,3,3-Trichloroallyl chloride
Apperance: Colorless oily liquid
Index of Refraction: 1.512
EINECS: 233-920-8
Density: 1.539 g/ml
Flash Point: 62.3 °C
Boiling Point: 173.8 °C
1,1,2,3-Tetrachloropropene Uses
1,1,2,3-Tetrachloropropene (CAS NO.10436-39-2) is Ye Yan fear herbicide intermediate and used for the synthesis of heat-resistant plastic.
1,1,2,3-Tetrachloropropene Production
A dehydrochlorination: tirring 1,1,2,3 - Tetrachloro-propane,calcium hydroxide and a certain amount of water in the reactors, to a certain reaction temperature for several hours, add 1:1 hydrochloric acid and in to acidic deposition layered collection of mixed-bit three allyl chloride.
Secondary chloride: reaction equipment is the same chlorination, mixed with bits into three allyl chloride, were mixed with bits 5-chloropropane.
Second dehydrochlorination: Production equipment is the same dehydrochlorination in the reaction vessel into position five chlorine mixed with propane, adding calcium hydroxide, dehydrochlorination was four allyl chloride.
ClCH=CHCH2Cl+Cl2→ClCH2CHClCHCl2
ClCH2CHClCHCl2+OH-→ClCH2ClG=CHCl
ClCH2ClC=CHCl+Cl2→Cl2CHCCl2CH2Cl
Cl2CHCCl2CH2Cl+OH-→Cl2C=CClCH2Cl
1,1,2,3-Tetrachloropropene Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LC50 | inhalation | 3200mg/m3 (3200mg/m3) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(10), Pg. 51, 1979. | |
| mouse | LD50 | oral | 800mg/kg (800mg/kg) | LIVER: OTHER CHANGES | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 43(4), Pg. 15, 1978. |
| rabbit | LD50 | oral | 477mg/kg (477mg/kg) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(10), Pg. 51, 1979. | |
| rabbit | LD50 | skin | 400uL/kg (0.4mL/kg) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. | |
| rat | LC50 | inhalation | 1500mg/m3 (1500mg/m3) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 23(10), Pg. 51, 1979. | |
| rat | LD50 | oral | 350mg/kg (350mg/kg) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. |
1,1,2,3-Tetrachloropropene Consensus Reports
Reported in EPA TSCA Inventory.
1,1,2,3-Tetrachloropropene Safety Profile
Poison by ingestion and skin contact. Moderately toxic by inhalation. Experimental reproductive effects. Mutation data reported. When heated to decomposition it emits toxic fumes of Cl−. See also CHLORINATED HYDROCARBONS, ALIPHATIC.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 10436-57-4
- 104366-23-6
- 1043688-99-8
- 104-36-9
- 104371-51-9
- 104372-31-8
- 104375-88-4
- 104376-24-1
- 104376-72-9
- 104376-79-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View