-
Name
1,3,5-Trichlorobenzene
- EINECS 203-608-6
- CAS No. 108-70-3
- Article Data81
- CAS DataBase
- Density 1.448 g/cm3
- Solubility Insoluble in water.
- Melting Point 63 °C
- Formula C6H3Cl3
- Boiling Point 211.3 °C at 760 mmHg
- Molecular Weight 181.449
- Flash Point 126.7 °C
- Transport Information
- Appearance white to beige crystalline powder
- Safety 26-36-36/37-61
- Risk Codes 20/21/22-36/37/38-38-22-52/53
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Benzene, 1,3,5-trichloro-;sym-trichlorobenzene;s-Trichlorobenzene;1,3,5-trichloro benzene;1,3,5-trichlorobenzol;
- PSA 0.00000
- LogP 3.64680
Synthetic route
-
-
634-90-2
1,2,3,5-tetrachlorobenzene

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

| Conditions | Yield |
|---|---|
| With radical anion of p,p'-di-tert-butylbiphenyl In tetrahydrofuran at -25℃; Irradiation; | A 2.8% B 97.2% |
-
-
19752-55-7
1-bromo-3,5-dichlorobenzene

-
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| With chlorine; sodium thiosulfate | 97.2% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; hypophosphorous acid; sodium nitrite In water at -5 - 25℃; | 95% |
| With tetrafluoroboric acid; N,N-dimethyl-formamide; sodium nitrite In water at 45℃; for 0.25h; Product distribution; or benzene, room temperature, 3 h; | 94% |
| Stage #1: 2,4,6-trichloroaniline With sulfuric acid Stage #2: With sodium nitrite In water for 0.75h; Cooling with ice; Stage #3: With sodium hypophosphite hydrate In hexane; water at 20℃; Cooling with ice; | 84% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; N-benzyl-N,N,N-triethylammonium chloride; sodium nitrite In tetrachloromethane 1.) 0 deg C, 1.5 h, 2.) r.t., 14 h; | 95% |
-
-
14862-52-3
1,3-dibromo-5-chlorobenzene

-
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| With chlorine; sodium thiosulfate In tetrachloromethane | 91% |
-
-
58-89-9
LINDANE

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| With ammonia at 25℃; Product distribution; Further Variations:; Reagents; Dehalogenation; | A 5% B 87% C 8% |
| at 25℃; pH=8.32; Kinetics; Product distribution; Further Variations:; pH-values; | |
| With (5,10,15,20-tetrakis(pentafluorophenyl)porphyrinato)iron(III) chloride; tetrabutylammonium perchlorate In N,N-dimethyl-formamide for 4h; Electrolysis; |


| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid; hydrogen iodide In acetonitrile at 60℃; for 24h; | A 3% B 86% |
-
-
634-90-2
1,2,3,5-tetrachlorobenzene

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| With radical anion of p,p'-di-tert-butylbiphenyl; triethylamine In tetrahydrofuran at 45℃; Irradiation; | A 18.8% B 75.5% C 5.7% |
| With radical anion of p,p'-di-tert-butylbiphenyl; triethylamine In tetrahydrofuran at 45℃; Product distribution; Rate constant; Mechanism; Irradiation; radical anion of naphthalene, different temperatures, without Et3N; | A 18.8% B 75.5% C 5.7% |
| With radical anion of p,p'-di-tert-butylbiphenyl In tetrahydrofuran at 45℃; Irradiation; | A 59.2% B 40.5% C 0.3% |
| In acetonitrile at 40℃; Product distribution; Mechanism; Quantum yield; Irradiation; in the presence of triethylamine; | A 18.77 % Chromat. B 75.45 % Chromat. C 5.78 % Chromat. |
| With sodium bis(2-methoxyethoxy)aluminium dihydride In toluene at 70℃; Rate constant; relative reactivity in dehalogenation; |


-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
33979-03-2
2,2',4,4',6,6'-Hexachlorobiphenyl

-
C
-
134253-68-2
2,4,6-trichlorobenzenediazohydroxide

-
D
-
23914-43-4, 18490-44-3
2,2',4,4',6,6'-Hexachlor-azobenzen

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 34℃; for 24h; Irradiation; Further byproducts given; | A 51.4% B 12.4% C 1.5% D 15.3% |


-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
33979-03-2
2,2',4,4',6,6'-Hexachlorobiphenyl

-
C
-
134253-68-2
2,4,6-trichlorobenzenediazohydroxide

-
D
-
23914-43-4, 18490-44-3
2,2',4,4',6,6'-Hexachlor-azobenzen

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 34℃; for 24h; Product distribution; influence of magnetic field; | A 51.4% B 12.4% C 1.5% D 15.3% E n/a |


-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
33979-03-2
2,2',4,4',6,6'-Hexachlorobiphenyl

-
C
-
23914-43-4, 18490-44-3
2,2',4,4',6,6'-Hexachlor-azobenzen

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 34℃; for 24h; Further byproducts given; | A 40.4% B 3.5% C 2.3% |

-
-
104891-98-7
3-Amino-3-(2,4,6-trichlor-phenylazo)propensaeuremethylester

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
2724-66-5
2,4,6-trichlorophenylhydrazine hydrochloride

-
C
-
23914-43-4, 18490-44-3
2,2',4,4',6,6'-Hexachlor-azobenzen

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 0.5h; Ambient temperature; | A 37% B 35% C n/a |
| With hydrogenchloride In methanol for 0.5h; Product distribution; Mechanism; Ambient temperature; | A 37% B 35% C n/a |

-
-
36997-05-4
2-methoxyacrylic acid ethyl ester

-
-
50-43-1
2,4,6-trichlorobenzoic acid

-
A
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; silver carbonate In 1,4-dioxane; dimethyl sulfoxide at 130℃; for 24h; Solvent; Sealed tube; Inert atmosphere; stereoselective reaction; | A n/a B 24% C 16% D n/a |


-
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 34℃; for 24h; Irradiation; | 13% |
| With trifluorormethanesulfonic acid at 34℃; for 24h; | 6.8% |
-
-
110-86-1
pyridine

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

-
-
91-22-5
quinoline

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| at 105 - 110℃; |


| Conditions | Yield |
|---|---|
| With tetrachloromethane at 290℃; | |
| With hydrogenchloride at 260℃; |
-
-
64-17-5
ethanol

-
-
319-86-8
delta-lindane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
58-89-9
LINDANE

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
319-85-7
beta-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |

-
-
64-17-5
ethanol

-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene

| Conditions | Yield |
|---|---|
| Product distribution; |


| Conditions | Yield |
|---|---|
| With nitrogen bei Licht- und Waermestrahlung einer 100 Watt-Lampe; |

| Conditions | Yield |
|---|---|
| With sodium ethanolate | |
| With sodium methylate In methanol; dimethyl sulfoxide at 50.1℃; Rate constant; |

| Conditions | Yield |
|---|---|
| ueber mehrere Stufen; |
-
-
24473-00-5
2,4,6-trichlorobenzaldehyde

-
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
33786-89-9
5-chloro-1,3-benzenediamine

-
-
108-70-3
1,3,5-trichlorobenzene

| Conditions | Yield |
|---|---|
| diazotiert und mit CuCl+HCl behandelt; |
-
-
319-84-6
alpha-hexachlorocyclohexane

-
A
-
108-70-3
1,3,5-trichlorobenzene

-
B
-
120-82-1
1,2,4-Trichlorobenzene

-
C
-
87-61-6
1,2,3-trichlorobenzene


| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium diacetate; DavePhos In toluene at 90℃; Suzuki Coupling; Inert atmosphere; Schlenk technique; | 100% |
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 94% |
| With trans-chloro(9-phenanthrenyl)bis(triphenylphosphine)nickel(II); potassium carbonate; triphenylphosphine In toluene at 110℃; for 18h; Suzuki-Miyaura Coupling; Inert atmosphere; | 91% |
| With potassium carbonate In ethanol; water at 90℃; for 36h; Suzuki-Miyaura Coupling; | 21% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: acetaldehyde In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With magnesium; ethylene dibromide; lithium chloride In tetrahydrofuran at 25℃; for 0.5h; Stage #2: carbon dioxide In tetrahydrofuran at 25℃; for 1h; Further stages; | 99% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
5720-05-8
4-methylphenylboronic acid

-
-
50446-43-0
1,3,5-tris(4-methyl-phenyl)-benzene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrabutylammomium bromide In water at 95℃; for 20h; Suzuki-Miyaura Coupling; | 99% |
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 90% |
| With potassium carbonate In ethanol; water at 90℃; for 24h; Suzuki-Miyaura Coupling; | 12% |

| Conditions | Yield |
|---|---|
| Stage #1: n-decyl glycolate With sodium methylate at 120℃; Stage #2: 1,3,5-trichlorobenzene at 160℃; | 98.9% |

| Conditions | Yield |
|---|---|
| With nitric acid In sulfuric acid at 25 - 120℃; Product distribution; | 98.5% |
| Stage #1: 1,3,5-trichlorobenzene With nitric acid at 40℃; for 0.25h; Stage #2: With sulfuric acid at 40 - 80℃; for 1.5h; | 96% |
| With sulfuric acid; nitric acid | 61% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 80 - 90℃; Concentration; Reagent/catalyst; Solvent; Temperature; Time; | 98% |
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 120℃; for 1h; | 78% |
| In methanol; dimethyl sulfoxide at 50.1℃; Rate constant; | |
| With methanol at 180℃; |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
75-69-4
trichlorofluoromethane

-
-
567-59-9
1,3,5-trichloro-2-(trifluoromethyl)benzene

| Conditions | Yield |
|---|---|
| With aluminium trichloride Ambient temperature; | 98% |
| With aluminium trichloride 1a) 4 h, r.t., 1b) 4 h, -10 deg C; | 98% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
151721-79-8
1,3,5-trichloro-2,4,6-triiodo-benzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; iodine; periodic acid at 20℃; Inert atmosphere; | 98% |
| With sulfuric acid; iodine; periodic acid at 20℃; | 98% |
| With sulfuric acid; periodic acid | 96% |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene for 8h; Solvent; Time; Reflux; | 96.5% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
33119-38-9
α-chlorobis(pentachlorophenyl)methane

-
-
105633-30-5
Bis-(2,4,6-trichlorophenyl)-methane

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 85℃; for 2.5h; | 96% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
24473-00-5
2,4,6-trichlorobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran; hexane at -78℃; for 1.5h; | 96% |
| Stage #1: 1,3,5-trichlorobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran; hexane at -78℃; for 1.5h; Inert atmosphere; | 95% |
| Stage #1: 1,3,5-trichlorobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran; hexane at -78℃; for 1.5h; | 95% |

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 100℃; for 1h; | 95% |
| In N,N-dimethyl acetamide at 100℃; for 24h; |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
73183-34-3
bis(pinacol)diborane

-
-
365564-05-2
1,3,5-tris(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzene

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; tris-(dibenzylideneacetone)dipalladium(0); lithium chloride In toluene at 100℃; for 15h; | 95% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
86-74-8
9H-carbazole

-
-
148044-07-9
1,3,5-tris(9H-carbazol-9-yl)benzene

| Conditions | Yield |
|---|---|
| With di-tert-butyl(2,2-diphenyl-1-methyl-1-cyclopropyl)phosphine; bis(η3-allyl-μ-chloropalladium(II)); methylmagnesium chloride In tetrahydrofuran; 5,5-dimethyl-1,3-cyclohexadiene at 5℃; Buchwald-Hartwig Coupling; Inert atmosphere; Reflux; | 94.6% |
| With di-tert-butyl(2,2-diphenyl-1-methyl-1-cyclopropyl)phosphine; bis(η3-allyl-μ-chloropalladium(II)); sodium t-butanolate In 5,5-dimethyl-1,3-cyclohexadiene at 120℃; for 3h; Inert atmosphere; | 67% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
7509-20-8
1,3,5-tris(4-methoxyphenyl)benzene

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(dibenzylideneacetone)palladium(0); catacxium A In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 94% |
| With 1,3-bis(2,6-diisopropylphenyl)-1,3,2-diazaphospholidine-2-oxide; potassium phosphate; chloro(1-naphthyl)bis(triphenylphosphine)nickel(II) In toluene at 110℃; for 18h; Suzuki Coupling; Schlenk technique; Inert atmosphere; | 67% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
24473-00-5
2,4,6-trichlorobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 2,4,6-trichlorobenzaldehyde In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 94% |
| Stage #1: 1,3,5-trichlorobenzene With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 2,4,6-trichlorobenzaldehyde In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With ammonium sulfate; bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; ammonia; sodium t-butanolate; tert-butyl XPhos In 1,4-dioxane at 100℃; for 24h; Reagent/catalyst; Sealed tube; Inert atmosphere; | 93% |
| With lithium amide; ammonia at -50℃; | |
| With ammonia |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; (Tricyclohexyl-n-dodecyl)phosphonium bromide In toluene at 50℃; for 3h; | A n/a B 93% |

-
-
108-70-3
1,3,5-trichlorobenzene

-
-
17049-50-2
n-decyl magnesium bromide

-
-
87969-78-6
1,3,5-tridecylbenzene

| Conditions | Yield |
|---|---|
| 1,2-bis(diphenylphosphino)ethane nickel(II) chloride In diethyl ether 1.) 18 h, 0 deg C to room temperature, 2.) 2 h, reflux; | 93% |

| Conditions | Yield |
|---|---|
| With Pd-PEPPSI-IPrAn; potassium tert-butylate In toluene at 80℃; for 24h; Suzuki coupling; Inert atmosphere; | 93% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
22479-78-3
4-(4-nitrophenoxy)phenol

| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-nitrophenoxy)phenol With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 2h; Inert atmosphere; Stage #2: 1,3,5-trichlorobenzene In N,N-dimethyl-formamide at 150℃; for 12h; Inert atmosphere; | 92.8% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
108-70-3
1,3,5-trichlorobenzene

-
-
363598-46-3
(2,4,6-trichlorophenyl)trimethylsilane

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With sec.-butyllithium In tetrahydrofuran; cyclohexane at -100℃; for 2h; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; cyclohexane | 92% |
| Stage #1: 1,3,5-trichlorobenzene With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -75℃; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -75℃; | 72% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
67-66-3
chloroform

-
-
40860-87-5
α,α,α',α',α'',α'',2,4,6-nonachloromesitylene

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride at 125℃; for 72h; Friedel Crafts alkylation; pressure tube; | 91% |
| With aluminum (III) chloride at 125℃; for 72h; Autoclave; | 73% |
| With aluminium trichloride at 100℃; for 18h; | 64% |
| With aluminum (III) chloride for 67h; Friedel-Crafts Alkylation; Sealed tube; Heating; | 32% |
| With aluminum (III) chloride at 125℃; for 72h; |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichlorobenzene With 2,2,6,6-tetramethyl-piperidine; n-butyllithium In tetrahydrofuran; hexane at -75℃; for 2h; Stage #2: With iodine In tetrahydrofuran; hexane at -75℃; | 91% |
-
-
108-70-3
1,3,5-trichlorobenzene

-
-
4294-57-9
para-methylphenylmagnesium bromide

-
-
50446-43-0
1,3,5-tris(4-methyl-phenyl)-benzene

| Conditions | Yield |
|---|---|
| With 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether at 20℃; for 2h; | 91% |
| Stage #1: 1,3,5-trichlorobenzene; para-methylphenylmagnesium bromide; 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether for 2h; Stage #2: With methanol In diethyl ether Product distribution / selectivity; | 91% |
1,3,5-TRICHLOROBENZENE Chemical Properties
1,3,5-TRICHLOROBENZENE(108-70-3) is also named SYM-TRICHLOROBENZENE;1,3,5-trichloro-benzen;ai3-22031;Benzene,1,3,5-trichloro-;I,3,5-TrichlorbenzolVW,16;s-Trichlorobenzene;trichloro-1,3,5benzene;1,3,5-TCB
Meiting Point: 63 oC
Boiling Point: 208 oC(lit.)
Flash Point: 260 oF
Storage temp.: 0-6oC
Stability: Stable. Incompatible with acids, strong oxidizing agents.
Index of Refraction: 1.567
Molar Refractivity: 40.93 cm3
Molar Volume: 125.2 cm3
Surface Tension: 39.9 dyne/cm
Density: 1.448 g/cm3
Enthalpy of Vaporization: 42.93 kJ/mol
Vapour Pressure: 0.267 mmHg at 25oC
The chemical structure of 1,3,5-TRICHLOROBENZENE(108-70-3) is
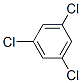 .
.1,3,5-TRICHLOROBENZENE Uses
1,3,5-TRICHLOROBENZENE Toxicity Data With Reference
| 1. | dlt-oin-oin-par 10 ppm | EVETBX Environmental Entomology. 2 (1973),1029. | ||
| 2. | dlt-oin-oin-skn 10 ppm | EVETBX Environmental Entomology. 2 (1973),1029. | ||
| 3. | mnt-mus-ipr 425 mg/kg/24H | MUTAEX Mutagenesis. 2 (1987),111. | ||
| 4. | orl-rat LD50:800 mg/kg | 48RKAL Industrial and Environmental Xenobiotics: Metabolism and Pharmacokinetics of Organic Chemicals and Metals, Proceedings of an International Conference, Prague ,Gut, I., et al.,Berlin, Fed. Rep, Ger..: Springer-Verlag,1981,389. | ||
| 5. | orl-mus LD50:3350 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) UCRL-13701 . | ||
| 6. | ipr-mus LD50:2260 mg/kg | MUTAEX Mutagenesis. 2 (1987),111. |
1,3,5-TRICHLOROBENZENE Consensus Reports
1,3,5-TRICHLOROBENZENE Safety Profile
Hazard Codes:
 Xn,Xi
Xn,Xi Risk Statements: 20/21/22-36/37/38-38-22-52/53
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
38: Irritating to the skin
22: Harmful if swallowed
52/53: Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment
Safety Statements: 26-36-36/37-61
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
36/37: Wear suitable protective clothing and gloves
61: Avoid release to the environment. Refer to special instructions safety data sheet
RIDADR: 3077
WGK Germany: 3
RTECS: DC2100100
Hazard Note: Harmful/Irritant
HazardClass: 9
PackingGroup: III
Hazardous Substances Data: 108-70-3(Hazardous Substances Data) .
1,3,5-TRICHLOROBENZENE Standards and Recommendations
1,3,5-TRICHLOROBENZENE Specification
(1)It must be separated from strong oxidants and keep in a well-ventilated room.
(2)When spilled,sweep it into containers; if appropriate, moisten first to prevent dusting. Carefully collect remainder, then remove to safe place. Do NOT let this chemical enter the environment.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 108710-70-9
- 108-71-4
- 108714-73-4
- 108723-79-1
- 108724-15-8
- 108-72-5
- 108729-78-8
- 108731-70-0
- 108731-71-1
- 108-73-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View