-
Name
N-Ethyl-2-pyrrolidone
- EINECS 220-250-6
- CAS No. 2687-91-4
- Article Data53
- CAS DataBase
- Density 0.993 g/cm3
- Solubility 1000g/L at 23℃
- Melting Point 87.5-88.5 °C
- Formula C6H11NO
- Boiling Point 203.6 °C at 760 mmHg
- Molecular Weight 113.159
- Flash Point 76.1 °C
- Transport Information
- Appearance clear liquid
- Safety 26-36
- Risk Codes 22-36
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 1-Ethyl-2-pyrrolidone;1-Ethylazacyclopentan-2-one;N-Ethyl-a-pyrrolidinone;N-Ethyl-g-butyrolactam;N-Ethylbutyrolactam;N-Ethylpyrrolidone;NEP;NEP 100;1-Ethyl-2-pyrrolidinone;
- PSA 20.31000
- LogP 0.56660
Synthetic route

| Conditions | Yield |
|---|---|
| With C7H14N3(1+)*Cl(1-); sodium triethylborohydride; cobalt(II) chloride In tetrahydrofuran at 25℃; under 7500.75 Torr; for 16h; | 99% |
| With ammonium formate; palladium on activated charcoal In methanol for 0.5h; Heating; | 94% |
| With oxygen; hydrazine hydrate; 7-(trifluoromethyl)-1,10-ethyleneisoalloxazinium chloride In water at 100℃; under 760.051 Torr; for 18h; | 88% |

| Conditions | Yield |
|---|---|
| In water; ethylene glycol at 290℃; under 38787.1 Torr; Kinetics; Temperature; Concentration; Flow reactor; | 93.9% |
| at 300℃; | |
| at 215℃; |
-
-
88-12-0
1-ethenyl-2-pyrrolidinone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
1190639-19-0
1-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)ethyl)pyrrolidin-2-one

| Conditions | Yield |
|---|---|
| With Rh(acac)(dppb) In tetrahydrofuran for 18h; Inert atmosphere; regioselective reaction; | A n/a B 83% |


| Conditions | Yield |
|---|---|
| dirhodium tetraacetate In dichloromethane at 40℃; for 5h; | 77% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-pyrrolidinon; ethyl bromide With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; for 2h; Stage #2: at 0℃; | 75% |
-
-
14468-90-7
N-trimethylsilyl-pyrrolidin-2-one

-
-
515-46-8
Ethyl benzenesulfonate

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
17882-06-3
trimethylsilyl benzenesulfonate

| Conditions | Yield |
|---|---|
| Heating; | A 59% B 72% |


| Conditions | Yield |
|---|---|
| ZSM-5 | 69% |

| Conditions | Yield |
|---|---|
| ZSM-5 | A 68% B 13% |


| Conditions | Yield |
|---|---|
| With silver tetrafluoroborate; dibromo[1,3-dihydro-1-(2-pyridinyl)-3-(2,4,6-trimethylphenyl)-2H-imidazol-2-ylidene]platinum(II) In nitrobenzene-d5 at 20 - 150℃; under 4125.41 Torr; for 26h; Inert atmosphere; | 59% |
-
-
6112-00-1
N,N-diethyl-2-diazoacetamide

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
31366-13-9
1-ethyl-4-methyl-2-azetidinone

-
C
-
111328-65-5
C12H22N2O2

| Conditions | Yield |
|---|---|
| With Cu(1+)*HB(C3N2Br3)3(1-)=(HB(C3N2Br3)3)Cu In dichloromethane at 20℃; Inert atmosphere; chemoselective reaction; | A 51% B 20% C 17% |
| With copper(I) hydrotris(3,5-dimethylpyrazol-1-yl)borate In dichloromethane at 20℃; Inert atmosphere; chemoselective reaction; |


| Conditions | Yield |
|---|---|
| With sodium hydride Heating; | 42% |
-
-
6112-00-1
N,N-diethyl-2-diazoacetamide

-
B
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
C
-
111328-65-5, 138895-86-0, 37511-04-9
N1,N1,N4,N4-tetraethylfumaramide

| Conditions | Yield |
|---|---|
| Ru2(CO)4[(PrO)4-5,17-Br2-11,23-(CO2-)2-calix[4]arene] In dichloromethane at 40℃; for 24h; | A 17% B 23% C 14% |

-
-
96-48-0
4-butanolide

-
-
7732-18-5
water

-
-
121-44-8
triethylamine

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
64-17-5
ethanol

| Conditions | Yield |
|---|---|
| Conversion of starting material; | A n/a B 13% |


| Conditions | Yield |
|---|---|
| With sulfuric acid; water Bei der elektrolytischen Reduktion an einer Blei-Kathode.; | |
| With 5% active carbon-supported ruthenium; hydrogen In water at 150℃; under 112511 Torr; for 6h; Green chemistry; |
-
-
63867-13-0
4-(N,N-diethylamino)butanoic acid

-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| With phosphorus trichloride Erhitzen des Reaktionsprodukts auf 155-160grad.; |
-
-
854852-25-8
3-hydroxy-4-nitro-butyric acid ethyl ester

-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; ethanol; copper oxide-chromium oxide at 260℃; under 191232 Torr; Hydrogenation; |
-
-
67-56-1
methanol

-
-
6112-00-1
N,N-diethyl-2-diazoacetamide

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
31366-13-9
1-ethyl-4-methyl-2-azetidinone

-
C
-
30280-35-4
N,N-diethylglycine methyl ester

-
D
-
70814-00-5
2-methoxy-N,N-diethyl-acetamide

| Conditions | Yield |
|---|---|
| at 0℃; for 0h; Product distribution; Irradiation; variuos temp.; | A 16.8 % Chromat. B 37.5 % Chromat. C 11.9 % Chromat. D n/a |


-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| With sulfuric acid at 45℃; elektrolytische Reduktion; |
-
-
2314-78-5
N-ethylsuccinimide

-
-
7664-93-9
sulfuric acid

-
-
7732-18-5
water

-
A
-
7335-06-0
1-ethylpyrrolidine

-
B
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| at 28℃; elektrochemischen Reduktion an einer Blei-Kathode; |

-
-
123-91-1
1,4-dioxane

-
-
64-17-5
ethanol

-
-
854852-25-8
3-hydroxy-4-nitro-butyric acid ethyl ester

-
A
-
7335-06-0
1-ethylpyrrolidine

-
B
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| at 260℃; under 191232 Torr; Hydrogenation; |


| Conditions | Yield |
|---|---|
| at 100℃; vom pH 1.2 bis pH 12.7; |
-
-
76572-84-4
4-(ethylamino)butyric acid

-
-
71-36-3
butan-1-ol

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| at 100℃; |


| Conditions | Yield |
|---|---|
| With sodium hydride at 60 - 70℃; |
-
-
3445-11-2
1-(2-hydroxyethyl)-2-pyrrolidinone

-
A
-
616-45-5
2-pyrrolidinon

-
B
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
C
-
88-12-0
1-ethenyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| 0.37 - 1.6 % cesium on silica In water at 350 - 375℃; for 3 - 95h; Product distribution / selectivity; | |
| 0.37 - 1.6 % cesium on silica at 350 - 375℃; for 3 - 95h; Product distribution / selectivity; |

-
-
88-12-0
1-ethenyl-2-pyrrolidinone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
1190639-18-9
((CH3)4C2O2)BCHCHNC4H6O

-
C
-
1190639-19-0
1-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)ethyl)pyrrolidin-2-one

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)] In benzene-d6 for 18h; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| With ammonium bromide at 250℃; for 5h; Autoclave; |
-
-
1455-13-6
deuteromethanol

-
-
6112-00-1
N,N-diethyl-2-diazoacetamide

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
31366-13-9
1-ethyl-4-methyl-2-azetidinone

-
C
-
1167575-44-1
C7H14(2)HNO2

| Conditions | Yield |
|---|---|
| Irradiation; |

-
-
6112-00-1
N,N-diethyl-2-diazoacetamide

-
A
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
B
-
31366-13-9
1-ethyl-4-methyl-2-azetidinone

| Conditions | Yield |
|---|---|
| In chloroform Irradiation; |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In N,N-dimethyl-formamide at 60℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| With ruthenium(IV) oxide; sodium periodate In ethyl acetate for 15h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; for 12h; Inert atmosphere; | 92% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
4083-64-1
Tosyl isocyanate

-
-
131657-47-1
(E)-N-(1-ethylpyrrolidin-2-ylidene)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 18h; | 91% |
-
-
292638-84-7
styrene

-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| With potassium tert-butylate at 80℃; for 2h; Sealed tube; | A 86% B 5% |

-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone With bis(cyclopentadienyl)dihydrozirconium; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane at 20℃; under 760.051 Torr; for 12h; Inert atmosphere; Stage #2: With hydrogenchloride In diethyl ether Inert atmosphere; | 86% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

| Conditions | Yield |
|---|---|
| With Lawessons reagent In tert-butyl methyl ether at 20℃; for 16h; Inert atmosphere; | 85% |
| With Lawessons reagent In benzene for 4h; Heating; | 83% |
| Multi-step reaction with 2 steps 1: POCl3 / CH2Cl2 / 0.5 h / -78 °C 2: hexamethyldisilathiane / CH2Cl2 / 6 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| With sodium hydride In toluene for 48h; Heating; | 85% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
700-58-3
2-Adamantanone

-
-
913335-86-1
2-(1-ethyl-2-oxopyrrolidin-3-yl)tricyclo[3.3.1.13,7]decan-2-ol

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone With lithium diisopropyl amide In tetrahydrofuran; diethyl ether; hexane at -80℃; for 0.333333h; Stage #2: 2-Adamantanone In tetrahydrofuran; diethyl ether; hexane at -80 - 20℃; Further stages.; | 84% |
| Stage #1: 1-ethyl-2-pyrrolidinone With lithium diisopropyl amide at -70℃; Stage #2: 2-Adamantanone In tetrahydrofuran at -80℃; |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
1333433-21-8
(E)-methyl 2-(2-methoxy-3-oxoisoindolin-1-ylidene)acetate

| Conditions | Yield |
|---|---|
| With air at 100℃; for 3h; Green chemistry; regioselective reaction; | 82% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
67217-55-4
mono-6-deoxy-6-(p-tolylsulphonyl)-β-cyclodextrin

| Conditions | Yield |
|---|---|
| at 90℃; | 80% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
1352122-79-2
(E)-methyl 2-(2-methoxy-4-methyl-3-oxoisoindolin-1-ylidene)acetate

| Conditions | Yield |
|---|---|
| With air at 100℃; for 12h; Green chemistry; regioselective reaction; | 79% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
18620-02-5
(4-bromophenyl)magnesium bromide

-
-
1109230-92-3
5-(4-bromophenyl)-1-ethyl-2,3-dihydropyrrole

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone; (4-bromophenyl)magnesium bromide In diethyl ether at 20℃; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride; water In diethyl ether pH=3 - 4; Cooling with ice; Stage #3: With sodium hydroxide In water | 78% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
100-58-3
phenylmagnesium bromide

-
-
1109230-81-0
1-ethyl-5-phenyl-2,3-dihydropyrrole

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone; phenylmagnesium bromide In diethyl ether at 20℃; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride; water In diethyl ether pH=3 - 4; Cooling with ice; Stage #3: With sodium hydroxide In water | 78% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
39216-86-9
4-(ethylamino)butan-1-ol

| Conditions | Yield |
|---|---|
| With ethanol; sodium In hexane at 0℃; for 0.333333h; Inert atmosphere; chemoselective reaction; | 78% |
| With ethanol; sodium In hexane; paraffin oil at 0℃; for 0.0833333h; Inert atmosphere; | 22% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
873-77-8
(4-chlorphenyl)magnesium bromide

-
-
1109230-91-2
5-(4-chlorophenyl)-1-ethyl-2,3-dihydropyrrole

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone; (4-chlorphenyl)magnesium bromide In diethyl ether at 20℃; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride; water In diethyl ether pH=3 - 4; Cooling with ice; Stage #3: With sodium hydroxide In water | 75% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate at 110℃; for 16h; | 74% |

| Conditions | Yield |
|---|---|
| With pyridine; N-hydroxyphthalimide; sodium perchlorate In acetonitrile Product distribution; Mechanism; controlled potential electrolysis, electrodes: glassy-carbon vs. SCE; | A 73% B 11% |
| With pyridine; N-hydroxyphthalimide; sodium perchlorate In acetonitrile controlled potential electrolysis, electrodes: glassy-carbon vs. SCE; | A 73% B 11% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
70-55-3
toluene-4-sulfonamide

-
-
131657-47-1
(E)-N-(1-ethylpyrrolidin-2-ylidene)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| With diazoacetic acid ethyl ester; zinc trifluoromethanesulfonate In cyclohexane for 12h; Reflux; stereoselective reaction; | 73% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
1352122-82-7
(E)-methyl 2-(2,5-dimethoxy-3-oxoisoindolin-1-ylidene)acetate

| Conditions | Yield |
|---|---|
| With air at 100℃; for 12h; Green chemistry; regioselective reaction; | 72% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
13139-86-1
4-methoxyphenyl magnesium bromide

-
-
1109230-89-8
1-ethyl-5-(4-methoxyphenyl)-2,3-dihydropyrrole

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone; 4-methoxyphenyl magnesium bromide In diethyl ether at 20℃; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride; water In diethyl ether pH=3 - 4; Cooling with ice; Stage #3: With sodium hydroxide In water | 71% |
-
-
2687-91-4
1-ethyl-2-pyrrolidinone

-
-
352-13-6
4-flourophenylmagnesium bromide

-
-
1109230-90-1
1-ethyl-5-(4-fluorophenyl)-2,3-dihydropyrrole

| Conditions | Yield |
|---|---|
| Stage #1: 1-ethyl-2-pyrrolidinone; 4-flourophenylmagnesium bromide In diethyl ether at 20℃; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride; water pH=3 - 4; Cooling with ice; Stage #3: With sodium hydroxide In water | 70% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid; dibenzoyl peroxide In water at 35 - 40℃; for 24h; Inert atmosphere; Irradiation; regioselective reaction; | 70% |
1-Ethyl-2-pyrrolidinone Chemical Properties
Molecular Formula: C6H11NO
Molecular Weight: 113.16
EINECS: 220-250-6
Index of Refraction: 1.464
Molar Refractivity: 31.47 cm3
Molar Volume: 113.8 cm3
Surface Tension: 32.6 dyne/cm
Density: 0.993 g/cm3
Flash Point: 76.1 °C
Enthalpy of Vaporization: 43.98 kJ/mol
Boiling Point: 203.6 °C at 760 mmHg
Vapour Pressure: 0.276 mmHg at 25 °C
Appearance: Clear liquid
Structure of 1-Ethyl-2-pyrrolidinone (CAS NO.2687-91-4):
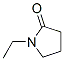
IUPAC Name: 1-Ethylpyrrolidin-2-one
Canonical SMILES: CCN1CCCC1=O
InChI: InChI=1S/C6H11NO/c1-2-7-5-3-4-6(7)8/h2-5H2,1H3
InChIKey: ZFPGARUNNKGOBB-UHFFFAOYSA-N
Product Category of 1-Ethyl-2-pyrrolidinone (CAS NO.2687-91-4): Heterocyclic Building Blocks;Pyrrolidines
1-Ethyl-2-pyrrolidinone Uses
1-Ethyl-2-pyrrolidinone (CAS NO.2687-91-4) is mainly used in the production of pharmaceuticals, dyestuff, pesticide, etc.
1-Ethyl-2-pyrrolidinone Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 1350mg/kg (1350mg/kg) | GASTROINTESTINAL: OTHER CHANGES LIVER: OTHER CHANGES KIDNEY, URETER, AND BLADDER: OTHER CHANGES | Food and Chemical Toxicology. Vol. 26, Pg. 475, 1988. |
| rat | LD50 | subcutaneous | > 2gm/kg (2000mg/kg) | United States Patent Document. Vol. #4882359, |
1-Ethyl-2-pyrrolidinone (CAS NO.2687-91-4) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
1-Ethyl-2-pyrrolidinone Consensus Reports
Reported in EPA TSCA Inventory.
1-Ethyl-2-pyrrolidinone Safety Profile
Moderately toxic by ingestion. An eye irritant. Combustible. When heated to decomposition it emits toxic fumes of NOx.Potential
The Hazard Codes:  Xn
Xn
The Risk Statements:
22: Harmful if swallowed
26: Very Toxic by inhalation
The Safety Statements:
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
1-Ethyl-2-pyrrolidinone Specification
1-Ethyl-2-pyrrolidinone , its cas register number is 2687-91-4. It also can be called N-Ethylpyrrolidinone ; NEP ; and N-Ethyl-2-pyrrolidone . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately.
In addition, 1-Ethyl-2-pyrrolidinone (CAS NO.2687-91-4) could be stable under normal temperatures and pressures. It is not compatible with strong oxidizing agents, strong acids, strong bases, acid chlorides, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Related Products
- 1-Ethyl-2-pyrrolidinone
- 2687-94-7
- 26879-47-0
- 2687-96-9
- 2687-97-0
- 2688-22-4
- 2688-48-4
- 2688-49-5
- 26885-67-6
- 26885-71-2
- 2688-84-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View