-
Name
1-Hydroxy-2-naphthoic acid
- EINECS 201-674-0
- CAS No. 86-48-6
- Article Data54
- CAS DataBase
- Density 1.399 g/cm3
- Solubility soluble in hot water
- Melting Point 195-200 °C (dec.)(lit.)
- Formula C11H8O3
- Boiling Point 367.7 °C at 760 mmHg
- Molecular Weight 188.183
- Flash Point 190.4 °C
- Transport Information
- Appearance beige to brown-grey powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Naphthoicacid, 1-hydroxy- (8CI);1-Hydroxy-2-naphthalenecarboxylic acid;
- PSA 57.53000
- LogP 2.24360
Synthetic route

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 180℃; under 25857.4 Torr; for 6h; | 96% |

| Conditions | Yield |
|---|---|
| at 140 - 190℃; under 7600 Torr; for 5h; | 93.4% |

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate at 200℃; for 5h; | 60% |
| With salicylic acid decarboxylase from Trichosporon moniliiforme; potassium hydrogencarbonate In acetonitrile at 30℃; for 24h; pH=8.5; aq. phosphate buffer; Enzymatic reaction; regioselective reaction; | |
| Multi-step reaction with 4 steps 1.1: 1H-imidazole / N,N-dimethyl-formamide / 0.5 h / 0 - 20 °C / Inert atmosphere 1.2: 0 - 20 °C / Inert atmosphere 2.1: water; sodium hydrogencarbonate / N,N-dimethyl-formamide; diethyl ether / 0.5 h / 20 °C / Inert atmosphere 3.1: palladium diacetate; silver(I) acetate; N-tert-butoxycarbonyl-L-leucine; 2,2,2-trifluoroethanol / 1,2-dichloro-ethane / 0.08 h / 95 °C / Inert atmosphere 3.2: 18 h / 95 °C / Inert atmosphere 4.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 0.5 h / Inert atmosphere View Scheme |
-
-
623-11-0
1-methyl-4-nitrosobenzene

-
-
858438-04-7
4-isopropyl-benzo[h]chromen-2-one

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With barium dihydroxide |
-
-
69796-59-4
3-acetyl-2-methyl-benzo[h]chromen-4-one

-
-
86-48-6
1-hydroxy-2-naphthoic acid
-
-
67176-26-5
1-hydroxynaphthalene-2-carbonitrile

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With alkaline solution |
-
-
3019-88-3
sodium naphtholate

-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With 1,4-dioxane | |
| With pyridine |

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| at 200 - 220℃; In der Kalischmelze; |
-
-
104095-33-2
3-benzoyl-5-chloromethyl-dihydro-furan-2-one

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction; |
-
-
61035-03-8
2-(diethylamino)-4H-naphtho[1,2-b]pyran-4-one

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol Heating; |
-
-
28591-61-9
6-Diazo-benzocycloheptene-5,7-dione

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With water |
-
-
574-96-9
1-hydroxy-2-naphthaldehyde

-
A
-
59648-32-7
2-(hydroxymethyl)-1-naphthol

-
B
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With barium dihydroxide; formaldehyd at 100 - 110℃; for 4h; | A 85 % Chromat. B 9 % Chromat. |
-
-
875224-19-4
3-ethyl-2-methyl-benzo[h]chromen-4-one

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
86-48-6
1-hydroxy-2-naphthoic acid
-
-
95703-28-9
3-benzyl-4-methyl-benzo[h]chromen-2-one

-
A
-
2550-26-7
4-Phenyl-2-butanone

-
B
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
861353-04-0
4-methyl-3-phenyl-benzo[h]chromen-2-one

-
A
-
10166-08-2
2-methyl-benzeneacetaldehyde

-
B
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
7446-70-0
aluminium trichloride

-
-
104095-33-2
3-benzoyl-5-chloromethyl-dihydro-furan-2-one

-
-
71-43-2
benzene

-
A
-
94540-41-7
phenyl(1,2,3,4-tetrahydronaphthalen-1-yl)methanone

-
B
-
112863-69-1
1,4,5-triphenyl-pentan-1-one

-
C
-
86-48-6
1-hydroxy-2-naphthoic acid


| Conditions | Yield |
|---|---|
| With sodium | |
| With sodium; toluene | |
| With potassium carbonate under 36775.4 Torr; | |
| With potassium hydrogencarbonate; glycerol |

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With sodium; sodium cyclohexanolate; cyclohexanol at 190 - 200℃; |

| Conditions | Yield |
|---|---|
| at 135℃; |

| Conditions | Yield |
|---|---|
| With alkali sulfite |
-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| Erhitzen des entstandenen Salzes im Autoklaven auf 120-140grad; | |
| Erhitzen des entstandenen Salzes im Autoklaven auf 120-140grad; |

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| at 200 - 220℃; durch Kalischmelze; |


-
-
77-78-1
dimethyl sulfate

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
948-03-8
1-hydroxy-naphthalene-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With lithium hydroxide In tetrahydrofuran for 1h; Methylation; esterification; Heating; | 100% |
| Stage #1: 1-hydroxy-2-naphthoic acid With lithium hydroxide In tetrahydrofuran for 0.5h; Stage #2: dimethyl sulfate In tetrahydrofuran for 3h; Heating; | 95.9% |
| Stage #1: 1-hydroxy-2-naphthoic acid With triethylamine In acetone for 0.25h; Inert atmosphere; Stage #2: dimethyl sulfate In acetone at 55℃; for 12h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Reflux; | 100% |
| With potassium carbonate In acetone Reflux; | 96% |
| With potassium carbonate In acetone for 18h; Reflux; | 95% |
-
-
71125-38-7
meloxicam

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1174325-92-8
4-hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide 1-hydroxy-2-naphthoic acid (1:1)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 0.5h; | 100% |
| In tetrahydrofuran Product distribution / selectivity; | |
| In dimethylsulfoxide-d6; water at 25 - 90℃; Solvent; | 71 g |
-
-
59721-28-7
camostat

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| In water; isopropyl alcohol at 25 - 60℃; | 99.1% |

| Conditions | Yield |
|---|---|
| With bromine In acetic acid at 20℃; | 99% |
| With bromine; acetic acid at 20℃; | 99% |
| With bromine In acetic acid at 20℃; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| In propan-1-ol (argon); storing in a Schlenk tube (ambient temp., 14 d); | 99% |

-
-
81-16-3
2-aminonaphthalenesulfonic acid

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
A
-
1402066-96-9
2-[(1-hydroxy-2-naphthalenyl)azo]-1-naphthalenesulfonic acid sodium salt

| Conditions | Yield |
|---|---|
| Stage #1: 2-aminonaphthalenesulfonic acid With hydrogenchloride; sodium hydroxide; sodium nitrite In water at 0℃; for 0.5h; Stage #2: 1-hydroxy-2-naphthoic acid With sodium carbonate In ethanol; water at 0℃; | A 12% B 97% |


| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic anhydride at 100℃; for 0.166667h; | 97% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 50℃; Temperature; | 96% |
| Stage #1: 1-hydroxy-2-naphthoic acid With potassium carbonate In acetone for 0.25h; Inert atmosphere; Stage #2: dimethyl sulfate In acetone at 55℃; for 12h; Inert atmosphere; | 95% |
| With potassium carbonate In acetone Heating; | |
| With potassium carbonate In acetone for 8h; Reflux; |
-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
A
-
179041-74-8
9-methoxy-2-amino-1,2,3,4-tetrahydroanthracene

-
B
-
179041-75-9
10-methoxy-2-amino-1,2,3,4-tetrahydrophenanthrene

| Conditions | Yield |
|---|---|
| A 95% B n/a |
-
-
1310717-02-2
3,6-diazabicyclo[3.1.1]heptan-3-yl(cyclopropyl) methanone

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1400593-10-3
C9H14N2O*C11H8O3

| Conditions | Yield |
|---|---|
| In Isopropyl acetate; water at 60℃; | 95% |

-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| Stage #1: (R)-N-(3-(5-fluoro-2-(2-fluoro-3-(methylsulfonyl)phenylamino)pyrimidin-4-yl)-1H-indol-7-yl)-3-methoxy-2-(4-methylpiperazin-1-yl)propanamide In methanol; ethyl acetate at 65℃; Inert atmosphere; Stage #2: 1-hydroxy-2-naphthoic acid In methanol; ethyl acetate at 50℃; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| In acetone at 30℃; for 0.666667h; Temperature; Solvent; | 94.1% |
| In acetone at 25 - 45℃; for 1.5h; | 69% |
| In diethyl ether at 20℃; for 16h; | |
| In isopropyl alcohol | |
| Stage #1: salmeterol With pyrographite In methanol at 25 - 30℃; for 0.5h; Stage #2: 1-hydroxy-2-naphthoic acid In methanol at 18 - 30℃; for 3h; | 20.75 g |
-
-
50-00-0
formaldehyd

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1175-41-3
4,4'-methylenebis(1-hydroxy-2-naphthoic acid)

| Conditions | Yield |
|---|---|
| With sulfuric acid 1.) 90 deg C, 6 h, 2.) RT, 12 h; | 94% |

| Conditions | Yield |
|---|---|
| With pyridine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In toluene at 20℃; for 7.5h; Inert atmosphere; | 93% |
| With sulfuric acid | |
| Stage #1: 1-hydroxy-2-naphthoic acid With thionyl chloride Stage #2: ethanol With triethylamine | |
| With sulfuric acid Reflux; |

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl sulfate; 1-hydroxy-2-naphthoic acid With potassium carbonate Stage #2: With lithium hydroxide In tetrahydrofuran; water | 92% |
| With sodium hydroxide In water |
-
-
1416767-53-7
5,5-dibenzyl-2,3,4,5-tetrahydropyridine

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1416767-44-6
12,12-dibenzyl-10,11,12,12a-tetrahydronaphtho[2,1-e]pyrido[2,1-b][1,3]oxazin-7(9H)-one

| Conditions | Yield |
|---|---|
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; N-ethyl-N,N-diisopropylamine In toluene at 90℃; for 20h; Inert atmosphere; | 92% |
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; N-ethyl-N,N-diisopropylamine In toluene at 90℃; for 20h; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrabutylammomium bromide In tetrahydrofuran at 30℃; for 0.166667h; | 91% |
-
-
67-56-1
methanol

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
948-03-8
1-hydroxy-naphthalene-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid | 90% |
| With sulfuric acid | 90% |
| With sulfuric acid at 80℃; for 8h; | 80% |
-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
33950-71-9
ethyl 1-hydroxy-2-naphthoate

| Conditions | Yield |
|---|---|
| With ethanol; sulfuric acid for 8h; Reflux; | 90% |
| Multi-step reaction with 2 steps 1: SOCl2 / tetrahydrofuran / 2 h / 20 °C 2: 88 percent / 0.17 h / 120 °C / microwave irradiation View Scheme | |
| Multi-step reaction with 2 steps 1: petroleum ether; thionyl chloride View Scheme |
-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
352662-32-9
2-chloro-4H-naphtho[1,2-d][1,3,2]dioxaphosphinin-4-one

| Conditions | Yield |
|---|---|
| With triethylamine; phosphorus trichloride In toluene at 20℃; | 90% |
| With anhydrous phosphorus trichloride In toluene | 75% |
| With Lewatit MP-62; phosphorus trichloride In toluene at 20℃; Product distribution / selectivity; | 75% |
| With triethylamine; phosphorus trichloride In toluene at 0 - 20℃; Product distribution / selectivity; | 74.35% |
-
-
105-36-2
ethyl bromoacetate

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1009813-72-2
1-ethoxycarbonylmethoxynaphthalene-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrabutylammomium bromide In tetrahydrofuran at 30℃; for 0.166667h; | 90% |
-
-
106-95-6
allyl bromide

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
1009813-71-1
1-(2-propenoxy)-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrabutylammomium bromide In tetrahydrofuran at 30℃; for 0.166667h; | 90% |

-
-
7732-18-5
water

-
-
86-48-6
1-hydroxy-2-naphthoic acid

-
-
869582-71-8
tetraaquabis(1-hydroxy-2-naphthoato-κO)manganese(II)

| Conditions | Yield |
|---|---|
| In water suspn. of Mn(OH)2 in H2O added to suspn. of acid in water; mixt. heated at 313 K for 6 wk; sepd. by decantation; dried at room temp.; | 90% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; for 10h; Cooling with ice; | 90% |
-
-
86-48-6
1-hydroxy-2-naphthoic acid

| Conditions | Yield |
|---|---|
| With Vilsmeier reagent; hydrazine hydrate; triethylamine In acetonitrile at 20℃; for 8h; | 90% |

| Conditions | Yield |
|---|---|
| With polyphosphoric acid for 0.166667h; Microwave irradiation; | 90% |
1-Hydroxy-2-naphthoic acid Chemical Properties
Product Name: 1-Hydroxy-2-naphthoic acid (CAS NO.86-48-6)
IUPAC Name: 1-hydroxynaphthalene-2-carboxylic acid
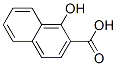
MF: C11H8O3
MW: 188.18
EINECS: 201-674-0
MP: 195-200 °C (dec.)(lit.)
Fp: 150 °C
Density: 1.399 g/cm3
Boiling Point: 367.7 °C at 760 mmHg
Water Solubility: Solubility in hot water
Appearance: Slightly gray granules.
Index of Refraction: 1.716
Molar Refractivity: 52.9 cm3
Molar Volume: 134.5 cm3
Categories: Intermediates of Dyes and Pigments
1-Hydroxy-2-naphthoic acid Uses
1-Hydroxy-2-naphthoic acid (CAS NO.86-48-6) is used as coupler and also used as the additive of battery.
1-Hydroxy-2-naphthoic acid Safety Profile
Safety Information of 1-Hydroxy-2-naphthoic acid (CAS NO.86-48-6):
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 26-36-37/39
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37/39: Wear suitable gloves and eye/face protection
WGK Germany: 3
HS Code: 29182910
1-Hydroxy-2-naphthoic acid Specification
1-Hydroxy-2-naphthoic acid , its CAS NO. is 86-48-6, the synonyms are 1-hydroxy-2-naphthalenecarboxylicaci ;1-hydroxy-2-Naphthalenecarboxylicacid
Related Products
- 1-Hydroxy-2-naphthoic acid
- 864866-56-8
- 864866-92-2
- 864872-31-1
- 86489-89-6
- 86490-14-4
- 86491-51-2
- 864947-20-6
- 864960-96-3
- 86499-23-2
- 86499-24-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View