-
Name
1-Indanone
- EINECS 201-470-1
- CAS No. 83-33-0
- Article Data477
- CAS DataBase
- Density 1.148 g/cm3
- Solubility 6.5 g/L (20°C) in water
- Melting Point 38-40 °C
- Formula C9H8O
- Boiling Point 244 °C at 760 mmHg
- Molecular Weight 132.162
- Flash Point 111.7 °C
- Transport Information
- Appearance pale yellow liquid
- Safety 22-24/25-36-26
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, Xn
Xn
- Synonyms alpha-indanone;Hydrindone;r-Hydrindone;alpha-hydrindone;1H-Inden-1-one, 2,3-dihydro-;2,3-dihydroinden-1-one;1H-Inden-1-one,2,3-dihydro-;Indanone (VAN);Indan-1-one;1-Indone;2,3-Dihydro-1H-inden-1-one;1-Indanone(1H-Inden-1-one,2,3-dihydro-);
- PSA 17.07000
- LogP 1.81550
Synthetic route

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In dichloromethane at 80℃; for 3h; Friedel-Crafts Acylation; High pressure; Inert atmosphere; Green chemistry; | 100% |
| With trifluorormethanesulfonic acid at 5 - 20℃; for 2h; Cyclization; | 99% |
| With polyphosphoric acid at 90℃; for 0.5h; | 98.2% |
-
-
68253-35-0, 100485-57-2, 3349-60-8
indan-1-one oxime

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With potassium permanganate; tungstate sulfuric acid In dichloromethane; water at 20℃; for 0.133333h; | 94% |
| With diphenyl ditelluride; oxygen In ethyl acetate for 24h; Irradiation; | 94% |
| With water; 2-nitro-4,5-dichloropyridazin-3(2H)-one In methanol at 150℃; under 10336 Torr; for 0.166667h; Microwave irradiation; | 90% |

| Conditions | Yield |
|---|---|
| With iodosylbenzene; potassium bromide In water for 2h; Oxidation; | 100% |
| With cerium(III) sulfate; barium bromate In water; acetonitrile for 2h; Heating; | 100% |
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In ethyl acetate at 80℃; | 100% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water at 75℃; for 12h; | 100% |
| With tert.-butylhydroperoxide; C21H19N5Pd(2+)*2BF4(1-) In decane; acetonitrile at 70℃; for 24h; Temperature; Wacker Oxidation; | 95% |
| With iron(II) chloride In ethanol at 80℃; for 6h; | 93% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; C24H18N6O3*Cu(1+)*I(1-) In water at 20℃; for 4h; Reagent/catalyst; | 98% |
| With tert.-butylhydroperoxide; copper(II) choride dihydrate; tetrabutyl-ammonium chloride; sodium carbonate; 2,2′‐biquinoline‐4,4′‐dicarboxylic acid dipotassium salt In water at 20℃; for 17h; Green chemistry; | 98% |
| With tert.-butylhydroperoxide; 0.5C34H18N16(4-)*2H2O*2Cu(1+)*C3H7NO In water at 20℃; for 4h; Reagent/catalyst; Sonication; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Phenylpropionic acid With trifluoroacetic anhydride In dichloromethane at 20℃; for 0.25h; Stage #2: With trifluorormethanesulfonic acid In dichloromethane at 20℃; for 0.5h; | A n/a B 94% |
| Stage #1: 3-Phenylpropionic acid With trifluoroacetic anhydride In dichloromethane at 20℃; for 0.25h; Stage #2: With trifluorormethanesulfonic acid In dichloromethane at 20℃; for 2h; | A 75% B 16% |
-
-
13225-61-1
1,7-diphenyl-4-oxahepta-1,6-diyne

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With water; PtCl4-CO In tetrahydrofuran at 80℃; under 1034.3 Torr; for 1.5h; | 94% |

| Conditions | Yield |
|---|---|
| With pyridine; palladium diacetate; tetrabutyl-ammonium chloride In N,N-dimethyl-formamide at 100℃; under 760 Torr; for 8h; | 100% |
-
-
40774-41-2
3-Bromoindan-1-one

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With water; zinc In acetonitrile at 25℃; for 6h; Sealed tube; Inert atmosphere; | 62% |
| With water; zinc In acetonitrile at 25℃; for 6h; Inert atmosphere; Sealed tube; | 62% |

| Conditions | Yield |
|---|---|
| With palladium dichloro (η-2,5-norbornadiene); oxygen; (-)-sparteine In toluene at 60℃; for 54h; Title compound not separated from byproducts; | A n/a B n/a C 63% |
| With 3 A molecular sieve; oxygen; (-)-sparteine; palladium dichloro (η-2,5-norbornadiene) In toluene at 60℃; under 760 Torr; for 54h; | A n/a B n/a C 63% |
| With (1R)-N-oxyl-1-(N-benzylcarbamoyl)-8-azabicyclo[3.2.1]octane; sodium hydrogencarbonate; sodium bromide In dichloromethane; water at 0℃; Electrochemical reaction; optical yield given as %ee; enantioselective reaction; | A n/a B n/a C 53% |


| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; oxygen; butane-2,3-dione dioxime In acetonitrile at 80℃; under 3750.38 Torr; for 8h; | A 10.4% B 75.3% |
| With N-hydroxyphthalimide; 1,4-diamino-2,3-dichloroanthraquinone; oxygen In acetonitrile at 80℃; under 1875.15 - 2250.18 Torr; for 6h; | A 15 % Chromat. B 64% |
| With 1H-imidazole; sodium periodate; Mn(TDCPP)OAc In dichloromethane for 24h; Ambient temperature; | A 26% B 60% |
-
-
78196-99-3
2-(phenylthio)-2,3-dihydro-1H-inden-1-one

-
A
-
83-33-0
inden-1-one

-
B
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In ethanol at 20℃; Irradiation; chemoselective reaction; | A 36% B 25% |

| Conditions | Yield |
|---|---|
| With water; triethylamine; triphenylphosphine; chloro(1,5-cyclooctadiene)rhodium(I) dimer In tetrahydrofuran at 160℃; under 51485.6 Torr; for 4h; | 61% |

| Conditions | Yield |
|---|---|
| With Novozym 435; 2,6-dimethyl-4-heptanol; (Ph4C4CO)2H(μ-H)(CO)4Ru2 In toluene at 70℃; for 72h; Reduction; Enzymatic reaction; | A 13 % Spectr. B 85% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With diethylamino-sulfur trifluoride In dichloromethane at -78 - 20℃; for 4.5h; Inert atmosphere; | 37% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate In butan-1-ol for 6h; Solvent; Reflux; | 86% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate In butan-1-ol for 6h; Reflux; | 88% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate In butan-1-ol for 12h; Reflux; | 85% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate In butan-1-ol for 6h; Reflux; | 90% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With indium(III) triflate In butan-1-ol for 4h; Reflux; | 86% |
-
-
28272-96-0
2-vinylbenzaldehyde

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; 6-diphenylphosphinomethyl-3-methyl-2-aminopyridine In toluene at 150℃; for 1h; Inert atmosphere; | 97% |
| With acetic acid; L-proline at 120℃; for 24h; Green chemistry; | 45% |
| (iPrO)Ti(μ:η1,η1-OCMe2CH2PPh2)RhCl In dichloromethane at 25℃; for 11h; | 40% |

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With sodium carbonate; acetic acid In methanol; water | 82.4% |

| Conditions | Yield |
|---|---|
| With pyridine; tetrabutyl-ammonium chloride; tri tert-butylphosphoniumtetrafluoroborate; palladium diacetate In 1,4-dioxane at 150℃; for 0.333333h; microwave irradiation; | 75% |

-
A
-
10277-74-4, 34698-41-4, 61341-86-4, 61949-83-5
(S)-1-Aminoindane

-
B
-
10277-74-4
(R)-1-Aminoindane

-
C
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With [Rh(OH)(cod)]2; Ureaphos-2; hydrogen; caesium carbonate In isopropyl alcohol at 55℃; under 97509.8 Torr; for 17h; Reagent/catalyst; Schlenk technique; Autoclave; Optical yield = < 2 %ee; | A n/a B n/a C 6 %Chromat. |


| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium bromide; chiral azabicyclo-N-oxyl In dichloromethane at 0℃; Electrochemical reaction; | A n/a B 52% |
| With C32H36IrNO2S In cyclohexanone at 30℃; for 7h; optical yield given as %ee; enantioselective reaction; | A 50% B n/a |
| With C50H42ClN2O3Ru In chloroform at 25℃; Kinetics; Time; Reagent/catalyst; Molecular sieve; Resolution of racemate; enantioselective reaction; | A 33.7% B n/a |
-
-
27326-43-8
2-vinylbenzoic acid

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With potassium phosphate; fac-tris(2-phenylpyridinato-N,C2')iridium(III); tris-(trimethylsilyl)silane; dimethyl dicarbonate In acetonitrile at 20℃; for 24h; Inert atmosphere; Schlenk technique; Irradiation; Sealed tube; | 76% |
| Multi-step reaction with 2 steps 1: 56 percent / tributylphosphine / CH2Cl2 / 4 h / -30 °C 2: 56 percent / Bu3SnH; AIBN / benzene / 10 h / Heating View Scheme |
-
-
52427-11-9
3-(4-methoxyphenyl)-2-methylpropionic acid

-
A
-
5464-10-8
6-methoxy-2-methylindanone

-
B
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With PPA | |
| With PPA | |
| With PPA In (2S)-N-methyl-1-phenylpropan-2-amine hydrate | |
| With PPA | |
| With PPA |
-
-
201230-82-2
carbon monoxide

-
-
2170-06-1
1-Phenyl-2-(trimethylsilyl)acetylene

-
A
-
104-53-0
3-phenyl-propionaldehyde

-
B
-
59556-86-4
2-Phenyl-3-(trimethylsilyl)-propanal

-
C
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine; triphenylphosphine; chloro(1,5-cyclooctadiene)rhodium(I) dimer In tetrahydrofuran at 160℃; under 36775.4 Torr; for 4h; | A 25% B 36% C 10% |


| Conditions | Yield |
|---|---|
| With triethylsilane; oxygen; [5,10,15,20-tetrakis(2,6-dichlorophenyl)porphinato]cobalt(II) In dichloromethane; isopropyl alcohol at 28℃; under 760 Torr; for 1.5h; | A 60% B n/a |
-
-
14381-42-1
Indan-1-carboxylic acid

-
-
83-33-0
inden-1-one

| Conditions | Yield |
|---|---|
| With (2,2'-dipyridyl)bis(5-methyl-2-(4-fluoro)phenylpyridine-N,C)iridium(III) hexafluorophosphate; oxygen; sodium carbonate; 1,1'-diethyl-4,4'-bipyridinium diperchlorate In dimethyl sulfoxide at 20℃; under 760.051 Torr; for 16h; Irradiation; | 77% |
| With iron(III) chloride; oxygen In N,N-dimethyl-formamide at 110℃; | 49% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid In water at 100℃; for 16h; | 100% |
| With hydrogenchloride; acetic acid at 100℃; for 16h; | 98% |
| With hydrogenchloride; acetic acid In water at 120℃; for 10h; | 90% |

| Conditions | Yield |
|---|---|
| With 15percent aqueous acidic TiCl3; 30percent aqueous NH3 In methanol at 0 - 20℃; for 0.0833333h; | 100% |
| With sodium tetrahydroborate | 99% |
| With C15H18BF3; hydrogen; tert-butylimino-tri(pyrrolidino)phosphorane In tetrahydrofuran at 75℃; under 75007.5 Torr; for 20h; Glovebox; | 99% |
-
-
83-33-0
inden-1-one

-
-
68253-35-0, 100485-57-2, 3349-60-8
indan-1-one oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride | 100% |
| With hydroxylamine hydrochloride In ethanol at 20 - 25℃; for 0.166667h; | 91.6% |
| With hydroxylamine hydrochloride; sodium hydroxide In ethanol at 20 - 25℃; for 1h; | 91.6% |
-
-
83-33-0
inden-1-one

-
-
25501-32-0
(S)-indanol

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran at 0℃; for 0.5h; enantioselective reaction; | 100% |
| With formic acid; [RuCl2(hexamethylbenzene)]2; (S)-2-piperidinemethanethiol hydrochloride; triethylamine at 30℃; for 24h; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |
| With formic acid; triethylamine at 28℃; for 48h; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With acetic acid at 120℃; for 20h; | 100% |

| Conditions | Yield |
|---|---|
| With (1R,2R)-PhCH(NH2)CH(Ph)NH-p-SO2-C6H4-CH2CH2-silica gel; tetrabutylammomium bromide; sodium formate; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2 at 40℃; | 100% |
| With dimethylsulfide borane complex; (S)-1-methyl-3,3-diphenyl-hexahydropyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran at 0℃; for 0.5h; enantioselective reaction; | 100% |
| With formic acid; C38H40ClN2O2RhS; triethylamine In neat (no solvent) at 24 - 30℃; for 5h; Reagent/catalyst; Inert atmosphere; enantioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With titanium(IV) oxide; potassium hydroxide In toluene at 100℃; for 10h; Sealed tube; Irradiation; | 100% |
| With dichloro[1,3‐diethyl(5,6‐dimethyl)benzimidazolin‐2‐ylidene](benzene)ruthenium (II); sodium hydroxide In toluene at 105℃; for 8h; | 88% |
| With bis(μ-chloro)-bis[1,3-di(2-pyridyl)-4,6-dimethylbenzene-N,C(2'),N-iridium chloride]; caesium carbonate In tert-Amyl alcohol for 12h; Reflux; | 87% |

| Conditions | Yield |
|---|---|
| With ethanol; lithium hydride In benzene Reflux; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With sulfuric acid Inert atmosphere; | 100% |
-
-
500-22-1
3-pyridinecarboxaldehyde

-
-
83-33-0
inden-1-one

-
-
4875-90-5
2-(pyridin-3-ylmethylene)-2,3-dihydro-1H-inden-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 0℃; Claisen-Schmidt Condensation; | 99.8% |
| With sodium hydroxide In methanol at 20℃; for 12h; |
-
-
500-22-1
3-pyridinecarboxaldehyde

-
-
83-33-0
inden-1-one

-
-
4875-90-5
(2E)-2-(pyridin-3-ylmethylidene)-2,3-dihydro-1H-inden-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 3-pyridinecarboxaldehyde; inden-1-one In ethanol for 0.0833333h; Inert atmosphere; Stage #2: With sodium hydroxide In ethanol; water for 0.5h; Inert atmosphere; | 99.8% |
| With sodium hydroxide In ethanol; water at 0℃; Claisen-Schmidt Condensation; | 73% |
| In methanol at 20℃; Alkaline conditions; | 29% |

| Conditions | Yield |
|---|---|
| Stage #1: m-Chlorobenzaldehyde; inden-1-one In ethanol for 0.0833333h; Inert atmosphere; Stage #2: With sodium hydroxide In ethanol; water for 0.5h; Inert atmosphere; | 99.7% |
-
-
872-85-5
pyridine-4-carbaldehyde

-
-
83-33-0
inden-1-one

-
-
4875-89-2
(E)-2-(pyridin-4-ylmethylene)-2,3-dihydro-1H-inden-1-one

| Conditions | Yield |
|---|---|
| Stage #1: pyridine-4-carbaldehyde; inden-1-one In ethanol for 0.0833333h; Inert atmosphere; Stage #2: With sodium hydroxide In ethanol; water for 0.5h; Inert atmosphere; | 99.4% |
| With piperidine; acetic acid at 130℃; for 1.5h; | 34% |
-
-
872-85-5
pyridine-4-carbaldehyde

-
-
83-33-0
inden-1-one

-
-
4875-89-2
2-(pyridin-4-ylmethylene)-2,3-dihydro-1H-inden-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 0℃; Claisen-Schmidt Condensation; | 99.4% |
-
-
89-98-5
2-chloro-benzaldehyde

-
-
83-33-0
inden-1-one

-
-
103457-37-0
(E)-2-(2-chlorobenzylidene)-2,3-dihydro-1H-inden-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-benzaldehyde; inden-1-one In ethanol for 0.0833333h; Inert atmosphere; Stage #2: With sodium hydroxide In ethanol; water for 0.5h; Inert atmosphere; | 99.2% |
| With sodium hydroxide In ethanol for 3h; | 84% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid at 20℃; for 24h; | 99.1% |
| basic condition; | |
| Claisen-Schmidt Condensation; Alkaline conditions; |

| Conditions | Yield |
|---|---|
| Stage #1: inden-1-one With sodium hydride In N,N-dimethyl-formamide for 1h; Stage #2: methyl iodide In N,N-dimethyl-formamide for 1.5h; | 99% |
| Stage #1: inden-1-one With sodium hydride In 1,2-dimethoxyethane at 10 - 35℃; for 0.166667h; Stage #2: methyl iodide In 1,2-dimethoxyethane at 10 - 35℃; for 1h; | 99% |
| Stage #1: inden-1-one With sodium hydride In 1,2-dimethoxyethane at 20℃; for 0.166667h; Stage #2: methyl iodide In 1,2-dimethoxyethane at 20℃; for 1h; | 99% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; toluene-4-sulfonic acid at 80℃; for 1h; | 99% |
| With bromine In acetic acid at 100℃; for 0.75h; | 97% |
| With hydrogen bromide; dihydrogen peroxide In methanol; water for 20h; Heating; | 89% |

| Conditions | Yield |
|---|---|
| 99% | |
| With bromine In diethyl ether Cooling with ice; | 99% |
| With copper bromide In chloroform; ethyl acetate for 1h; Reflux; | 96% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
83-33-0
inden-1-one

-
-
31928-65-1
1-((trimethylsilyl)oxy)-2,3-dihydro-1H-indene-1-carbonitrile

| Conditions | Yield |
|---|---|
| With 4-methylmorpholine N-oxide In dichloromethane at 20℃; for 18h; | 99% |
| Stage #1: inden-1-one With scandium tris(trifluoromethanesulfonate); 1-n-butyl-3-methylimidazolium hexafluoroantimonate at 20℃; for 0.166667h; Stage #2: trimethylsilyl cyanide at 20℃; for 2h; Inert atmosphere; | 99% |
| With C29H46LaN3Si2 at 15℃; for 24h; Inert atmosphere; Glovebox; Schlenk technique; | 99% |
-
-
83-33-0
inden-1-one

-
-
616-38-6
carbonic acid dimethyl ester

-
-
22955-77-7
methyl 1-indanone-2-carboxylate

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 99% |
| With sodium hydride In toluene; mineral oil at 0 - 120℃; for 18h; | 95% |
| With sodium hydride In tetrahydrofuran; mineral oil for 2h; Reflux; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex; (S)-2-(anilinodiphenylmethyl)pyrrolidine In tetrahydrofuran at 20℃; for 2h; enantioselective reaction; | A 99% B n/a |
| With dimethylsulfide borane complex In tetrahydrofuran at 20℃; for 2h; enantioselective reaction; | A 96% B n/a |
| With sprouted Pisum sativa In water at 25℃; for 58h; optical yield given as %ee; enantioselective reaction; | A n/a B 42% |

| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethyl acetamide; inden-1-one With trifluorormethanesulfonic acid; thallium(III) acetate at 60℃; for 0.333333h; Stage #2: With water for 0.166667h; Further stages.; | 99% |
-
-
83-33-0
inden-1-one

-
-
90271-93-5
β-bromoindenone

| Conditions | Yield |
|---|---|
| Stage #1: inden-1-one With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane for 2h; Heating; irradiation; Stage #2: With triethylamine In tetrachloromethane at 0 - 20℃; for 12h; | 99% |
| Stage #1: inden-1-one With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane for 2h; Photolysis; Reflux; Stage #2: With triethylamine In tetrachloromethane at 0℃; for 9h; | 95% |
| Stage #1: inden-1-one With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane for 2.5h; Reflux; Stage #2: With triethylamine In tetrachloromethane at 0℃; | 85% |

-
-
60556-33-4
N-(tert-butyl)-N-(1-chloro-1,1-dimethylsilyl)amine

-
-
83-33-0
inden-1-one

-
-
308846-97-1
N-(tert-butyl)-N-(1-(1H-3-indenyloxy)-1,1-dimethylsilyl)amine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | 99% |

1-Indanone Chemical Properties
IUPAC Name: 2,3-Dihydroinden-1-one
Synonyms of 1-Indanone (CAS NO.83-33-0): 1-Indone ; 2,3-Dihydro-1H-inden-1-one ; alpha-Hydrindone ; alpha-Indanone ; 1H-Inden-1-one, 2,3-dihydro-
CAS NO: 83-33-0
Molecular Formula: C9H8O
Molecular Weight: 132.16
Molecular Structure: 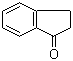
EINECS: 201-470-1
H bond acceptors: 1
H bond donors: 0
Freely Rotating Bonds: 0
Polar Surface Area: 17.07 Å2
Index of Refraction: 1.585
Molar Refractivity: 38.55 cm3
Molar Volume: 115 cm3
Surface Tension: 44.8 dyne/cm
Density: 1.148 g/cm3
Flash Point: 111.7 °C
Enthalpy of Vaporization: 48.1 kJ/mol
Boiling Point: 244 °C at 760 mmHg
Vapour Pressure: 0.0311 mmHg at 25°C
Melting point : 38-40 °C
Water Solubility: 6.5 g/L (20°C)
Appearance: Pale yellow liquid
Product Categories of 1-Indanone (CAS NO.83-33-0): Drug Intermediates;Indane/Indanone and Derivatives;Indanone & Indene;Fused Ring Systems
1-Indanone Safety Profile
Safety Information about 1-Indanone (CAS NO.83-33-0):
Hazard Codes:  Xn,Xi
Xn,Xi
Risk Statements: 22-36/37/38
R22: Harmful if swallowed.
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 22-24/25-36-26
S22: Do not breathe dust.
S24/25: Avoid contact with skin and eyes.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
WGK Germany: 3
Hazard Note: Irritant
HS Code: 29143900
1-Indanone Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media: In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam.
Handling: Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage: Store in a cool, dry place. Keep container closed when not in use.
Related Products
- 1-Indanone
- 1-Indanone oxime
- 1-Indanone-6-carboxylic acid
- 83335-41-5
- 83-34-1
- 83344-22-3
- 833445-84-4
- 83345-46-4
- 833472-82-5
- 83348-31-6
- 833486-94-5
- 833489-37-5
- 833-50-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View