-
Name
1-Methyl-2-phenylindole
- EINECS 222-618-1
- CAS No. 3558-24-5
- Article Data177
- CAS DataBase
- Density 1.04 g/cm3
- Solubility
- Melting Point 98-100 °C(lit.)
- Formula C15H13N
- Boiling Point 380.8 °C at 760 mmHg
- Molecular Weight 207.275
- Flash Point 184.1 °C
- Transport Information
- Appearance white to light green-tan crystals or crystal powder
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms 1-methyl-2-phenyl-1H-indole;1-Methyl-2-phenyl-1H-indole;2-Phenyl-N-methylindole;Indole, 1-methyl-2-phenyl-;N-Methyl-2-phenylindole;
- PSA 4.93000
- LogP 3.84530
Synthetic route
-
-
170956-92-0
N-methyl-2-(2-phenylethynyl)aniline

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With chloro(1,3-bis(2,6-di-i-propylphenyl)imidazol-2-ylidene)gold(I); silver trifluoromethanesulfonate In 1,2-dichloro-ethane at 20℃; for 0.0166667h; Catalytic behavior; Sealed tube; | 100% |
| With silver nitrate In acetonitrile at 80℃; for 8h; Reagent/catalyst; Temperature; Solvent; Time; | 99% |
| With [(THD-Dipp)AuCl]; silver trifluoromethanesulfonate In ethanol at 20℃; for 0.5h; Reagent/catalyst; Inert atmosphere; Schlenk technique; | 97% |
-
-
20878-31-3, 65053-50-1, 65053-51-2
1-methyl-2-phenylindoline

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With ferrocene; tris(pentafluorophenyl)borate In toluene at 120℃; under 75.0075 Torr; for 16.5h; Inert atmosphere; Schlenk technique; Glovebox; | 99% |
| With N-hydroxyphthalimide In acetonitrile at 80℃; for 6h; Sealed tube; | 74% |
| With N-hydroxyphthalimide In acetonitrile at 80℃; for 6h; | 74% |
-
-
948-65-2
2-phenyl-indole

-
-
616-38-6
carbonic acid dimethyl ester

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene at 90℃; for 28h; Product distribution; Further Variations:; Temperatures; Solvents; Pressures; Reagents; reaction times; microwave irradiation; | 98% |
| With 1,4-diaza-bicyclo[2.2.2]octane In N,N-dimethyl-formamide at 94 - 95℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With Pd2[(C6H4)PPh2]2[CF3C(O)CHC(O)CF3]2 In acetic acid at 24.84℃; for 30h; Reagent/catalyst; Time; Solvent; Inert atmosphere; Schlenk technique; | 98% |
| With palladium containing [Zr6O4(OH)4(terephthalate)4.75(1,2,4,5-benzenetetracarboxylate)1.25] at 80℃; for 5h; Reagent/catalyst; Concentration; | 94% |
| With (1,3-di(2,4,6-(CH3)3-C6H2)-imidazol-2-ylidene)Pd(OAc)2; acetic acid at 25℃; for 18h; | 86% |
| With palladium diacetate In ethanol at 50℃; for 24h; Kinetics; Reagent/catalyst; | 51% |
-
-
603-76-9
1-methylindole

-
-
58109-40-3
diphenyliodonium hexafluorophosphate

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With Pd2[(C6H4)PPh2]2[CF3C(O)CHC(O)CF3]2 In acetic acid at 24.84℃; for 30h; Catalytic behavior; Reagent/catalyst; Time; Solvent; Inert atmosphere; Schlenk technique; | 98% |
| Stage #1: 1-methylindole With Pd2[(C6H4)PPh2]2[O2CC6H5]2; acetic acid at 24.84℃; for 0.0833333h; Inert atmosphere; Schlenk technique; Stage #2: diphenyliodonium hexafluorophosphate at 24.84℃; for 10h; Reagent/catalyst; Inert atmosphere; Schlenk technique; | 97% |
-
-
948-65-2
2-phenyl-indole

-
-
373-68-2
tetramethylammonium fluoride

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| In toluene at 100℃; for 6h; | 98% |
-
-
128746-62-3
2,3-dibromo-1-methyl-1H-indole

-
-
98-80-6
phenylboronic acid

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water at 90℃; for 6h; Suzuki-Miyaura Coupling; | 97% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium bis(2,2,6,6-tetramethyl-3,5-heptanedionate) In 1-methyl-pyrrolidin-2-one at 125℃; for 36h; | 96% |
| With water; palladium diacetate; silver cyclohexanecarboxylate at 30℃; for 16h; Time; Reagent/catalyst; | 93% |
| With ortho-nitrobenzoic acid; silver(l) oxide; palladium diacetate In N,N-dimethyl-formamide at 25℃; for 7h; Product distribution; Further Variations:; Reagents; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-phenylethynylaniline With potassium hydride In 1-methyl-pyrrolidin-2-one at 20℃; for 2h; Stage #2: methyl iodide In 1-methyl-pyrrolidin-2-one for 0.5h; | 96% |
-
-
21375-88-2
1-bromo-2-(phenylethenyl)benzene

-
-
74-89-5
methylamine

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; OTips-DalPhos; potassium tert-butylate In toluene at 90℃; for 12h; Reagent/catalyst; Buchwald-Hartwig Coupling; Inert atmosphere; Sealed tube; | 95% |
| With (R)-1-[(SP)-2-(dicyclohexylphosphino)ferrocenyl]ethyldi-tert-butylphosphine; bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; potassium tert-butylate In tetrahydrofuran; toluene at 90℃; Inert atmosphere; | 88% |
| With bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; OTips-DalPhos; potassium tert-butylate In tetrahydrofuran; toluene at 90℃; for 12h; Sealed vial; | 83% |
-
-
54655-08-2
N,N-dimethyl-2-(2-phenylethynyl)aniline

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid; silver(l) oxide at 40℃; for 3h; | 95% |
| With ethanol at 120℃; for 10h; Microwave irradiation; Inert atmosphere; Green chemistry; | 93% |
| With toluene-4-sulfonic acid; silver(l) oxide In N,N-dimethyl-formamide at 60℃; for 12h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Schlenk technique; | 88% |
| Multi-step reaction with 2 steps 1.1: iron(III) chloride / dichloromethane / 0.33 h / 25 °C 1.2: air / 24 h / 25 °C 2.1: n-butyllithium / tetrahydrofuran; hexane / 0 °C / Inert atmosphere 2.2: 0 - 25 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 2-phenyl-indole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; for 1h; Stage #2: methyl iodide In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; | 94% |
| Stage #1: 2-phenyl-indole With potassium hydroxide In N,N-dimethyl-formamide at 0℃; for 0.25h; Stage #2: methyl iodide In N,N-dimethyl-formamide at 0 - 20℃; for 6h; | 93% |
| Stage #1: 2-phenyl-indole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; for 0.5h; Stage #2: methyl iodide In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 24h; | 90% |
-
-
118486-94-5
2-(tributylstannyl)furan

-
-
170956-92-0
N-methyl-2-(2-phenylethynyl)aniline

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate In 1,2-dichloro-ethane at 80℃; Inert atmosphere; Glovebox; Schlenk technique; | 91% |
-
-
603-76-9
1-methylindole

-
-
13056-98-9
3,3-diethyl-1-phenyltriazene

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With C9H18N2O3S*H2O4S; palladium diacetate In water; N,N-dimethyl-formamide at 20℃; for 5h; Reagent/catalyst; regioselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide 60 degC 30 min then 80-90 degC 1 h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylindole With 2,2,6,6-tetramethylpiperidinyl-lithium In tetrahydrofuran; hexane; pentane at 20℃; for 0.5h; Inert atmosphere; Stage #2: chlorobenzene In tetrahydrofuran; hexane; pentane at 20℃; for 1.5h; Inert atmosphere; | 90% |
| With cesium acetate In N,N-dimethyl-formamide at 150℃; for 48h; under air; | 21% |
-
-
603-76-9
1-methylindole

-
-
369-57-3
benzenediazonium tetrafluoroborate

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylindole With palladium diacetate In 2-methyltetrahydrofuran; ethyl acetate for 2h; Stage #2: benzenediazonium tetrafluoroborate In 2-methyltetrahydrofuran; ethyl acetate at 20℃; for 0.5h; Catalytic behavior; Solvent; Reagent/catalyst; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: styrene oxide; N-methylaniline With zinc trifluoromethanesulfonate at 20℃; for 1h; Inert atmosphere; Stage #2: With 1,1'-bis-(diphenylphosphino)ferrocene; dodecacarbonyl-triangulo-triruthenium; toluene-4-sulfonic acid In 1,4-dioxane at 150℃; for 22h; Inert atmosphere; regioselective reaction; | 89% |
-
-
603-76-9
1-methylindole

-
-
591-50-4
iodobenzene

-
A
-
30020-98-5
N-methyl-3-phenylindole

-
B
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With palladium dichloride In dimethyl sulfoxide; acetonitrile at 100℃; for 8h; Catalytic behavior; Time; Reagent/catalyst; Temperature; Solvent; regioselective reaction; | A n/a B 88% |
| With 2,6-bis[(N-phenyl)imino]phenylpalladium bromide; potassium acetate at 80℃; for 24h; Reagent/catalyst; Solvent; Time; Overall yield = 98 %Chromat.; regioselective reaction; | A n/a B 85% |
| With PdBr2(bipy)*DMSO; silver carbonate In 1,4-dioxane at 150℃; for 13h; Inert atmosphere; | A n/a B 60% |
| With potassium acetate In water at 100℃; for 24h; Overall yield = 73.4 %; | |
| With sodium acetate; palladium dichloride In water; N,N-dimethyl-formamide at 140℃; Reagent/catalyst; regioselective reaction; |


| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; oxygen; C39H47N3O4Pd In dimethyl sulfoxide at 60℃; under 760.051 Torr; for 24h; Reagent/catalyst; Sealed tube; | 88% |
| With trifluorormethanesulfonic acid; oxygen; palladium diacetate In dimethyl sulfoxide at 70℃; under 760.051 Torr; for 20h; | 84% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; silver trifluoroacetate In methanol at 50℃; for 7h; Reagent/catalyst; Solvent; regioselective reaction; | 86% |
| With [bis(acetoxy)iodo]benzene; palladium diacetate In acetic acid at 20℃; for 16h; | 84% |
| With oxygen; palladium diacetate; acetic acid at 20℃; Schlenk technique; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: C9H8ClNZn*LiCl With trans-[1,3-bis(2,6-diisopropylphenyl)imidazolin-2-ylidene]PdCl2(NC5H5) In tetrahydrofuran at 20℃; for 0.05h; Inert atmosphere; Stage #2: methyl-phenyl-thioether In tetrahydrofuran; acetonitrile at 20℃; for 3h; Inert atmosphere; | 86% |
-
-
603-76-9
1-methylindole

-
-
7227-91-0
3,3-dimethyl-1-phenyltriazene

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With C9H18N2O3S*H2O4S; palladium diacetate In water; N,N-dimethyl-formamide at 20℃; for 5h; Reagent/catalyst; regioselective reaction; | 86% |

| Conditions | Yield |
|---|---|
| With [Pd(OTs)2(MeCN)2]; oxygen; acetic acid at 25℃; for 12h; Reagent/catalyst; Temperature; Schlenk technique; | 86% |
-
-
603-76-9
1-methylindole

-
-
13056-98-9
3,3-diethyl-1-phenyltriaz-1-ene

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; palladium diacetate In N,N-dimethyl acetamide at 80℃; for 12h; Sealed tube; | 85% |

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With water In benzene-d6 for 0.0833333h; Inert atmosphere; | 85% |

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With thiosalicilyc acid; trifluoroacetic acid at 72℃; for 1.5h; | 84% |
| With thiosalicilyc acid In trifluoroacetic acid at 72℃; for 1.5h; | 84% |
| With thiosalicilyc acid; trifluoroacetic acid at 72℃; for 1.5h; Mechanism; further 3-indolyl sulfides; | 84% |

| Conditions | Yield |
|---|---|
| (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride | 84% |
-
-
603-76-9
1-methylindole

-
-
36719-71-8
(E)-1-phenyl-2-(pyrrolidin-1-yl)diazene

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With hexafluorophosphoric acid; palladium diacetate In water; N,N-dimethyl-formamide at 0 - 20℃; for 4h; Catalytic behavior; Concentration; Reagent/catalyst; Solvent; Temperature; regioselective reaction; | 84% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
1666-13-3
diphenyl diselenide

-
-
1094425-08-7
1-Methyl-2-phenyl-3-(phenylselenyl)-1H-indole

| Conditions | Yield |
|---|---|
| With potassium iodate; glycerol In neat (no solvent) at 100℃; for 6h; Inert atmosphere; Schlenk technique; | 100% |
| With Rose Bengal lactone In acetonitrile at 20℃; for 6h; Schlenk technique; Irradiation; | 99% |
| With iodine; dimethyl sulfoxide In neat (no solvent) at 80℃; for 0.0833333h; Microwave irradiation; Sealed tube; Green chemistry; | 95% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
882-33-7
diphenyldisulfane

-
-
82946-84-7
1-methyl-2-phenyl-3-(phenylsulfenyl)indole

| Conditions | Yield |
|---|---|
| With potassium iodate; glycerol In neat (no solvent) at 100℃; for 6h; Inert atmosphere; Schlenk technique; | 100% |
| With sodium tetrafluoroborate; potassium iodide In acetonitrile at 60℃; for 12h; Electrolysis; | 89% |
| With sodium tetrafluoroborate; potassium iodide In acetonitrile at 60℃; for 12h; Electrochemical reaction; | 89% |
| With ammonium iodide; water; acetic acid; dimethyl sulfoxide at 110℃; for 6h; Green chemistry; regioselective reaction; | 77% |
| With sodium iodide In acetonitrile at 20℃; for 18h; Irradiation; | 52% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With hydrogen bromide; dimethyl sulfoxide In water; ethyl acetate at 60℃; for 0.166667h; | 99% |
| With N-Bromosuccinimide In chloroform at 20℃; for 0.5h; | 97% |
| With tetrabutylammomium bromide; sodium hydrogencarbonate; Selectfluor In toluene at 20℃; for 2h; Reagent/catalyst; Solvent; Schlenk technique; regioselective reaction; | 96% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
1135130-14-1
(R)-diisopropyl 3-((R)-(1-methyl-2-phenyl-1H-indol-3-yl)(phenyl)methyl)-4-methylenecyclopentane-1,1-dicarboxylate

| Conditions | Yield |
|---|---|
| With (R)-4-MeO-3,5-(t-Bu)2MeOBIPHEP(AuCl)2; silver trifluoromethanesulfonate In diethyl ether at 20℃; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With (R)-4-MeO-3,5-(t-Bu)2-MeOBIPHEP(AuCl)2; silver trifluoromethanesulfonate In diethyl ether at 20℃; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
452962-71-9
3-iodo-1-methyl-2-phenyl 1H-indole

| Conditions | Yield |
|---|---|
| With 1,3-Diiodo-5,5-dimethyl-2,4-imidazolidinedione; 4,4'-dimethoxyphenyl disulfide In acetonitrile at 20℃; for 0.75h; | 99% |
| With 1,3-Diiodo-5,5-dimethyl-2,4-imidazolidinedione; diphenyldisulfane In acetonitrile at 20℃; for 1.5h; | 99% |
| With sodium hydrogencarbonate; Selectfluor; potassium iodide In toluene at 20℃; for 2h; Reagent/catalyst; Schlenk technique; regioselective reaction; | 96% |
| With ammonium iodide; sulfuric acid; dimethyl sulfoxide In ethyl acetate at 60℃; for 5h; | 94% |
| With [bis(acetoxy)iodo]benzene; sodium iodide In water; acetonitrile at 20℃; regioselective reaction; | 94% |

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate) In acetonitrile at 25℃; for 2h; regioselective reaction; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
1147550-11-5
ammonium thiocyanate

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 3h; Electrochemical reaction; Inert atmosphere; | 99% |
| With oxygen In tetrahydrofuran at 25℃; under 750.075 Torr; for 10h; Irradiation; | 95% |
| With Oxone In methanol at 20℃; for 0.5h; | 87% |
| With TiO2/MoS2 In acetonitrile at 20℃; for 16h; Irradiation; | 84% |

| Conditions | Yield |
|---|---|
| With 7.78 wtpercent Cu supported on porous hypercrosslinked polymer (from N,N'-dibenzylbenzimidazolium chloride and benzene) In ethanol at 80℃; for 3h; | 99% |

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
106-54-7
p-Chlorothiophenol

-
-
1346164-88-2
3-((4-chlorophenyl)thio)-1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With lithium perchlorate In acetonitrile at 20℃; for 3h; Inert atmosphere; Electrochemical reaction; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
13056-98-9
3,3-diethyl-1-phenyltriazene

| Conditions | Yield |
|---|---|
| With C8H16N2*BrH*FeCl3 In 2,2,2-trifluoroethanol at 0 - 20℃; for 2h; Reagent/catalyst; regioselective reaction; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With Diphenylphosphinic chloride In acetonitrile at 60℃; for 1h; Schlenk technique; Inert atmosphere; | 99% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
124-38-9
carbon dioxide

-
-
59050-41-8
2-phenyl-1-methylindole-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With dimethylaluminum chloride In hexane; toluene at 80℃; under 22502.3 Torr; for 3h; Autoclave; | 98% |
| With dimethylaluminum chloride In hexane; toluene at 20℃; under 22502.3 Torr; for 3h; Friedel-Crafts reaction; regioselective reaction; | 61% |
| With N,N-dimethyl-aniline In acetonitrile Irradiation; | 2.5% |
-
-
1076-38-6
4-hydroxy[1]benzopyran-2-one

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
89-98-5
2-chloro-benzaldehyde

-
-
1609076-44-9
3-[(2-chlorophenyl)(1-methyl-2-phenyl-1H-indol-3-yl)-methyl]-4-hydroxy-2H-chromen-2-one

| Conditions | Yield |
|---|---|
| With L-proline In ethanol at 80℃; for 4h; | 98% |

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In 1,2-dichloro-ethane at 80 - 120℃; for 16h; Schlenk technique; | 98% |
| With chloro-trimethyl-silane at 80 - 120℃; Inert atmosphere; Sealed tube; | 98% |

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate In 1,2-dichloro-ethane at 95℃; for 16h; | 98% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
609769-63-3
N-benzyl-N-(p-toluenesulfonyl)-2-phenylethynylamine

| Conditions | Yield |
|---|---|
| With erbium(III) triflate In 1,2-dichloro-ethane at 70℃; for 2h; regioselective reaction; | 98% |
| With erbium(III) triflate In 1,2-dichloro-ethane at 70℃; for 2h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; | 98% |

| Conditions | Yield |
|---|---|
| With acetic anhydride at 90℃; for 1h; | 97% |
| With propionic acid anhydride at 65 - 75℃; for 0.116667h; | 93% |
-
-
860772-38-9
(E)‐1‐(1‐methyl‐1H‐imidazole‐2‐yl)‐but‐2‐en‐1‐one

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With C38H38IrN4O2(1+)*F6P(1-) In tetrahydrofuran at 20℃; for 40h; Friedel-Crafts Alkylation; Inert atmosphere; enantioselective reaction; | 97% |
-
-
51626-58-5
(1H-indol-3-yl)(phenyl)methanol

-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
78602-03-6
3-((1H-indol-3-yl)(phenyl)methyl)-1-methyl-2-phenyl-1H-indole

| Conditions | Yield |
|---|---|
| With silica gel In toluene at 20℃; for 2h; Friedel-Crafts Alkylation; | 97% |
| With 2,2,2-trifluoroethanol at 20℃; Sealed tube; Green chemistry; | 72% |
-
-
3558-24-5
1-methyl-2-phenyl-1H-indole

-
-
53841-59-1
1,1,3,3,3-pentafluoro-2-trimethylsiloxypropene

| Conditions | Yield |
|---|---|
| With copper(l) chloride In 1,2-dichloro-ethane at 80℃; for 16h; Inert atmosphere; | 97% |
| With copper(l) chloride In 1,2-dichloro-ethane at 80℃; for 24h; Inert atmosphere; | 97% |
1-Methyl-2-phenylindole Specification
The 1H-Indole,1-methyl-2-phenyl-, with the CAS registry number 3558-24-5 and EINECS registry number 222-618-1, has the systematic name of 1-methyl-2-phenyl-1H-indole. It is a kind of white to light green-tan crystals or crystal powder, and belongs to the following product categories: Intermediates of Dyes and Pigments; Indole; Indoles; Simple Indoles; Building Blocks; Heterocyclic Building Blocks. And the molecular formula of the chemical is C15H13N. What's more, while dealing with this chemical, you should avoid contacting with skin and eyes.
The characteristics of 1H-Indole,1-methyl-2-phenyl- are as followings: (1)ACD/LogP: 4.30; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 4.3; (4)ACD/LogD (pH 7.4): 4.3; (5)ACD/BCF (pH 5.5): 1086.41; (6)ACD/BCF (pH 7.4): 1086.41; (7)ACD/KOC (pH 5.5): 5185.19; (8)ACD/KOC (pH 7.4): 5185.19; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 4.93 Å2; (13)Index of Refraction: 1.599; (14)Molar Refractivity: 67.79 cm3; (15)Molar Volume: 198.4 cm3; (16)Polarizability: 26.87×10-24cm3; (17)Surface Tension: 38.9 dyne/cm; (18)Density: 1.04 g/cm3; (19)Flash Point: 184.1 °C; (20)Enthalpy of Vaporization: 60.43 kJ/mol; (21)Boiling Point: 380.8 °C at 760 mmHg; (22)Vapour Pressure: 1.17E-05 mmHg at 25°C.
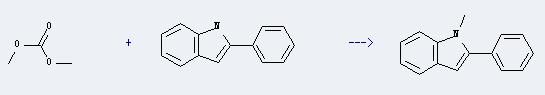
Preparation of 1H-Indole,1-methyl-2-phenyl-: This chemical can be prepared by 2-phenyl-indole and carbonic acid dimethyl ester. The reaction will need reagent 1,8-diazabicyclo[5.4.0]undec-7-ene. The reaction time is 28 hours with temperature of 90°C, and the yield is about 98%.
Uses of 1H-Indole,1-methyl-2-phenyl-: It can react with hexanal to produce 1,1-Bis(N-methyl-2'-phenylindol-3-yl)hexane. This reaction will need reagent methanesulfonic acid, and the menstruum acetonitrile. The reaction time is 4 hours, and the yield is about 61%.
.jpg)
Addtionally, the following datas could be converted into the molecular structure:
(1)SMILES: c1cccc3c1cc(c2ccccc2)n3C
(2)InChI: InChI=1/C15H13N/c1-16-14-10-6-5-9-13(14)11-15(16)12-7-3-2-4-8-12/h2-11H,1H3
(3)InChIKey: SFWZZSXCWQTORH-UHFFFAOYAK
Related Products
- 1-Methyl-2-phenylindole
- 1-Methyl-2-phenylindole-3-carboxaldehyde
- 35582-80-0
- 355836-10-1
- 35585-58-1
- 3558-60-9
- 3558-61-0
- 355-86-2
- 3558-69-8
- 35588-36-4
- 35588-53-5
- 35589-32-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View