 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
-
Name
1-Methylcyclopentene
- EINECS 211-762-0
- CAS No. 693-89-0
- Article Data146
- CAS DataBase
- Density 0.821 g/cm3
- Solubility
- Melting Point -142 °C
- Formula C6H10
- Boiling Point 78.4 °C at 760 mmHg
- Molecular Weight 82.1454
- Flash Point 75-77 °C
- Transport Information UN 3295
- Appearance clear colourless liquid
- Safety 16-62-7
- Risk Codes 11-65
-
Molecular Structure
-
Hazard Symbols
 F,
F,  Xn
Xn
- Synonyms 1-Methyl-1-cyclopentene;1-Methylcyclopentene;NSC 64657;
- PSA 0.00000
- LogP 2.11660
Synthetic route

| Conditions | Yield |
|---|---|
| In liquid sulphur dioxide Ambient temperature; | 99% |
| With bromine chloride | 67 % Spectr. |
| With (allylsulfonyl)benzene In Cyclohexane-d12; toluene at 130℃; for 36.5h; Product distribution; Further Variations:; Reagents; Solvents; Temperatures; | 95 % Spectr. |
| With diphenyl disulfone In chloroform-d1 at 80℃; for 1h; Kinetics; |

| Conditions | Yield |
|---|---|
| Stage #1: methyllithium; cyclopentanone at -10 - 0℃; for 0.5h; Stage #2: With toluene-4-sulfonic acid In 1,2-dichloro-ethane Reflux; | 96% |
| With acid |

| Conditions | Yield |
|---|---|
| 95% | |
| With iodine; oxalic acid | 87% |
| With iodine Erhitzen des erhaltenen Gemisches von 1-Methyl-cyclopenten und Methylencyclopentan mit Essigsaeure und wenig Toluol-4-sulfonsaeure; |

| Conditions | Yield |
|---|---|
| Stage #1: methyl magnesium iodide; cyclopentanone In diethyl ether for 1h; Heating; Stage #2: With iodine In toluene for 3h; Heating; | 94% |
| In diethyl ether for 3h; Ambient temperature; | 34.8% |
| 2) dehydration; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; | 87% |
-
-
30043-41-5
(hex-5-enyl)magnesium bromide

-
A
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With isopropylmagnesium bromide; bis(cyclopentadienyl)titanium dichloride In diethyl ether at 36℃; for 5h; | A 66% B n/a |
-
-
115-10-6
Dimethyl ether

-
-
142-29-0
cyclopentene


-
B
-
187737-37-7
propene

-
C
-
74-85-1
ethene

-
D
-
693-89-0
1-methylcyclopent-1-ene

-
E
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| MFI280 zeolite at 400℃; under 750.075 Torr; for 20h; Product distribution / selectivity; Gas phase; | A 59.5% B 22.9% C 6% D 9.5% E 2.1% |
| MTT47 zeolite at 400℃; under 750.075 Torr; for 20h; Product distribution / selectivity; Gas phase; | A 52.2% B 6.7% C 1.4% D 39.3% E 0.4% |

-
-
110-83-8
cyclohexene

-
A
-
110-54-3
hexane

-
B
-
96-37-7
methyl-cyclopentane

-
C
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| ultrastabilized Y zeolite at 350℃; for 0.0333333h; | A 5.6% B 57.7% C 8.3% |

-
-
592-42-7
1,5-Hexadien

-
A
-
5194-50-3
(E,Z)-2,4-hexadiene

-
B
-
7319-00-8
(E)-1,4-hexadiene

-
C
-
1528-30-9
methylenecyclopentane

-
D
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With isopropylmagnesium bromide; bis(cyclopentadienyl)titanium dichloride In diethyl ether at 20℃; for 3h; Further byproducts given. Yields of byproduct given; | A n/a B n/a C n/a D 57% E n/a |

-
-
110-83-8
cyclohexene

-
A
-
1528-30-9
methylenecyclopentane

-
B
-
693-89-0
1-methylcyclopent-1-ene

-
C
-
1120-62-3
3-methyl-1-cyclopentene

| Conditions | Yield |
|---|---|
| ultrastabilized Y zeolite at 450℃; for 1h; | A 1.7% B 54.4% C 9.9% |

-
-
110-83-8
cyclohexene

-
A
-
693-89-0
1-methylcyclopent-1-ene

-
B
-
1759-81-5
4-methylcyclopentene

-
C
-
1120-62-3
3-methyl-1-cyclopentene

| Conditions | Yield |
|---|---|
| ultrastabilized Y zeolite at 450℃; for 1h; | A 54.4% B 4.9% C 9.9% |
| aluminum hydroxide contg. TiO2 and Na2O at 350℃; Kinetics; | |
| With hydrogenchloride; water In decalin at 225℃; for 49h; Product distribution; Mechanism; in sealed tube; also with DCl/D2O, deuteration investigated; also at 175 deg C; | A 45.2 % Chromat. B n/a C n/a |

-
-
110-83-8
cyclohexene

-
A
-
110-54-3
hexane

-
B
-
1528-30-9
methylenecyclopentane

-
C
-
96-37-7
methyl-cyclopentane

-
D
-
693-89-0
1-methylcyclopent-1-ene

-
E
-
1759-81-5
4-methylcyclopentene

-
F
-
1120-62-3
3-methyl-1-cyclopentene

| Conditions | Yield |
|---|---|
| ultrastabilized pprotonic Y zeolite In gas at 350℃; for 1h; Product distribution; Kinetics; other temperatures, other products; | A 0.5% B 1.4% C 1% D 19.6% E 1.8% F 3.6% |


| Conditions | Yield |
|---|---|
| With formaldehyd; ruthenium (III) bromide; hydrogen bromide; hydrogen; tetrabutyl phosphonium bromide In water at 200℃; under 30003 Torr; for 8h; | 13% |

| Conditions | Yield |
|---|---|
| With formaldehyd; ruthenium (III) bromide; hydrogen bromide; hydrogen; tetrabutyl phosphonium bromide In water at 200℃; under 30003 Torr; for 8h; | A 12% B 7% |

| Conditions | Yield |
|---|---|
| at 190 - 200℃; |
-
-
917-64-6
methyl magnesium iodide

-
-
6196-85-6
1-chloro-1-methylcyclopentane

-
A
-
1638-26-2
1,1-Dimethylcyclopentane

-
B
-
693-89-0
1-methylcyclopent-1-ene

-
-
19872-99-2
1-bromo-1-methyl-cyclopentane

-
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With quinoline at 190 - 200℃; | |
| With 1,3,5-triphenylverdasyl at 25℃; Rate constant; other solvents; | |
| In ethanol at 25℃; Kinetics; Further Variations:; Solvents; |

| Conditions | Yield |
|---|---|
| With water at 35℃; | |
| With quinoline at 160 - 190℃; | |
| With sodium hydroxide In water at 20℃; for 2h; Dehydrochlorination; elimination; | |
| With 1,3,5-triphenylverdazyl In acetonitrile at 25℃; Kinetics; Further Variations:; Solvents; Temperatures; | |
| With 1,3,5-triphenylverdazyl In methanol at 25℃; Kinetics; Further Variations:; Solvents; |
-
-
6196-85-6
1-chloro-1-methylcyclopentane

-
A
-
693-89-0
1-methylcyclopent-1-ene

-
B
-
1462-03-9
1-methylcyclopentanol

| Conditions | Yield |
|---|---|
| With water In ethanol at 30℃; Rate constant; solvolysis reaction; | |
| With calcium hydroxide; water at 35℃; |
-
-
26600-59-9
1-methylcyclopentyl acetate

-
A
-
1528-30-9
methylenecyclopentane

-
B
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| at 450℃; |
-
-
26600-59-9
1-methylcyclopentyl acetate

-
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| at 450℃; |
-
-
18729-48-1
3-methylcyclopentanol

-
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
53501-51-2
1-chloro-2-methyl-cyclopentane

-
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With quinoline at 160 - 190℃; |
-
-
26600-49-7
cyclopentylmethyl acetate

-
A
-
1528-30-9
methylenecyclopentane

-
B
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| at 450 - 570℃; |
-
-
859181-91-2
1-methyl-cyclopentanecarboxylic acid phenyl ester

-
A
-
693-89-0
1-methylcyclopent-1-ene

-
B
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| at 390℃; im Rohr; |

| Conditions | Yield |
|---|---|
| at 90 - 120℃; |

| Conditions | Yield |
|---|---|
| Erhitzen des Reaktionsprodukts mit Chinolin auf 190-200grad; |
-
-
82166-21-0
methyl cyclohexane

-
-
693-89-0
1-methylcyclopent-1-ene

| Conditions | Yield |
|---|---|
| With chromium (III)-oxide-aluminium oxide contacts at 550℃; |

| Conditions | Yield |
|---|---|
| at 200 - 300℃; Leiten ueber zuvor mit Phosphorsaeure behandeltes Kieselgur; | |
| at 400 - 500℃; Leiten ueber γ-Aluminiumoxyd; |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
123-38-6
propionaldehyde

-
-
81977-75-5
(methyl-2' cyclopentyl)-1 propanone-1

| Conditions | Yield |
|---|---|
| With dibenzoyl peroxide at 90℃; for 10h; | 100% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
16240-42-9
1,2-epoxy-1-methylcyclopentane

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 5 - 20℃; Inert atmosphere; | 99% |
| With sodium hydrogencarbonate; 3-chloro-benzenecarboperoxoic acid In dichloromethane at 24℃; for 4h; | 84% |
| With gallium oxide; dihydrogen peroxide; acetonitrile at 89.84℃; for 4h; Sealed tube; | 53% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
1198746-23-4
(R)-methyl 2-(cyclopentenylmethyl)-3,3,3-trifluoro-2-hydroxypropanoate

| Conditions | Yield |
|---|---|
| With 2,6-bis[(3aR,8aS)-3a,8a-dihydro-8H-indeno[1,2-d]oxazolin-2-yl]pyridine; indium(III) chloride; silver hexafluoroantimonate In 1,2-dichloro-ethane at 20℃; Ionic liquid; Molecular sieve; optical yield given as %ee; enantioselective reaction; | 98% |
-
-
924-44-7
glyoxylic acid ethyl ester

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
208242-85-7
ethyl (R)-3-(cyclopent-1-en-1-yl)-2-hydroxypropanoate

| Conditions | Yield |
|---|---|
| With 2,6-bis[(3aR,8aS)-3a,8a-dihydro-8H-indeno[1,2-d]oxazolin-2-yl]pyridine; silver hexafluoroantimonate; indium(III) chloride In 1,2-dichloro-ethane at 20℃; for 16h; Molecular sieve; Inert atmosphere; stereoselective reaction; | 98% |
-
-
91-01-0
1,1-Diphenylmethanol

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
1352552-71-6
((2-methylcyclopent-1-enyl)methylene)dibenzene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In ethylene dibromide at 60℃; Air atmosphere; regioselective reaction; | 96% |
| With N-octadecyl-N-(4-sulfobutyl)pyrrolidinium trifluoromethanesulfonate In dichloromethane at 80℃; for 6h; | 92% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
130575-06-3
2-Iodo-2-cyanopent-4-ynenitrile

-
-
141517-23-9
trans-Propargyl-(2-iodo-2-methylcyclopentyl)-malononitrile

| Conditions | Yield |
|---|---|
| In benzene at 80℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| With air In tetrahydrofuran byproducts: methylcyclopentane; N2-atmosphere; treatment of B2H6 with olefin, addn. of MeSH (stirring), injecting small amt. of air (in two steps during 2 h), stirring for 12 h; evapn. of volatiles (reduced pressure), fractional distn. (reduced pressure); | 95% |


| Conditions | Yield |
|---|---|
| With 9-(2-mesityl)-10-methylacridinium perchlorate In 1,2-dichloro-ethane at 20℃; Irradiation; Green chemistry; regioselective reaction; | 94% |
-
-
201230-82-2
carbon monoxide

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
20106-44-9
trans-2-Methylcyclopentanecarbaldehyde

| Conditions | Yield |
|---|---|
| With dicarbonylacetylacetonato rhodium (I); C37H50NO4P; hydrogen In toluene at 100℃; under 15001.5 Torr; for 12h; Autoclave; regioselective reaction; | 92% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
151-10-0
1,3-Dimethoxybenzene

-
-
1258741-73-9
2,4-dimethoxy-1-(1-methylcyclopentyl)benzene

| Conditions | Yield |
|---|---|
| With calcium(II) bis(trifluoromethanesulfonyl)imide; tert-butylammonium hexafluorophosphate(V) In dichloromethane at 20℃; for 1h; | 91% |
-
-
34069-94-8
trichloro-acetyl bromide

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
51284-43-6
7,7-dichloro-1-methylbicyclo[3.2.0]heptan-6-one

| Conditions | Yield |
|---|---|
| With zinc In diethyl ether Heating; | 90% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
119741-57-0
5-cyano-1H-pyrazole-4-carboxylic acid, ethyl ester

-
-
119741-58-1
5-cyano-1-(1-methylcyclopentyl)-1H-pyrazole-4-carboxylic acid, ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In dichloromethane; acetonitrile for 2h; Ambient temperature; | 90% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
545-06-2
trichloroacetonitrile

-
-
97963-20-7
2,2,2-Trichloro-1-(2-methyl-cyclopent-2-enyl)-ethanone

| Conditions | Yield |
|---|---|
| With boron trifluoride; water In dichloromethane at -78℃; | 90% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
114124-63-9
[Chloro-(3,4-dichloro-phenylsulfanyl)-methyl]-trimethyl-silane

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane at -20℃; for 0.5h; | 90% |
-
-
693-89-0
1-methylcyclopent-1-ene

-
-
39947-48-3
(1S,2S)-trans-2-methylcyclopentanol

| Conditions | Yield |
|---|---|
| With (R,R)-C6H12BH2(1-)*Li(1+)*OEt2; methyl iodide In diethyl ether for 9.5h; Ambient temperature; | 89% |

| Conditions | Yield |
|---|---|
| With dichlorobis(tri-O-tolylphosphine)palladium; potassium carbonate In N,N-dimethyl acetamide; ethylene glycol at 75℃; for 22h; Schlenk technique; Inert atmosphere; Sealed tube; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-methylcyclopent-1-ene With ozone In dichloromethane at -78℃; Stage #2: With triphenylphosphine In dichloromethane at -78 - 20℃; for 10h; | 87% |
| Stage #1: 1-methylcyclopent-1-ene With ozone In dichloromethane at -78 - 20℃; Stage #2: With dimethylsulfide In dichloromethane at 20℃; for 5h; Stage #3: With triphenylphosphine In dichloromethane at 20℃; for 5h; Further stages.; | 87% |
| Stage #1: 1-methylcyclopent-1-ene With osmium(VIII) oxide; 4-methylmorpholine N-oxide; tert-butyl alcohol In acetone at 20℃; for 4h; Stage #2: With sodium periodate In water at 20℃; for 1h; | 85% |

| Conditions | Yield |
|---|---|
| With zeolite-P at 80℃; for 3h; | 86.4% |
| cationite KU-23 at 80℃; for 5h; Product distribution; other alkyl- and alkenylcyclenes; var. temp. and time; | |
| cationite KU-23 at 80℃; for 5h; |

| Conditions | Yield |
|---|---|
| With diethylzinc In cyclohexane for 120h; Ambient temperature; | 86% |
| With iodine; copper; zinc In diethyl ether |

| Conditions | Yield |
|---|---|
| With Lindlar's catalyst; ozone-containing oxygen; ammonium chloride; sodium methylate 1) -75 Deg C, 2) room temperature, 3) 48 h.; | 86% |
| With dimethylsulfide; ozone 1.) methylene chloride, -78 deg C, 2.) room temperature, 3 h; Multistep reaction; |

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; for 24h; Title compound not separated from byproducts.; | A 85% B 15% |

-
-
110077-68-4, 89509-77-3
{IrH2(acetone)2(PPh3)2}SbF6

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
106864-47-5
hydrido(η-methylcyclopentadienyl)bis(triphenylphosphine)iridium(III) hexafluoroantimonate

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-benzene refluxed for 6 h under inert atmosphere; cooled, evapd., residue recrystd. from CH2Cl2-Et2O; | 85% |

-
-
693-89-0
1-methylcyclopent-1-ene

-
-
500569-41-5
(C5(CH3)5)Y(η(3)-C5H6CH3)

| Conditions | Yield |
|---|---|
| In further solvent(s) under inert atm. using Schlenk techniques; 1-methylcyclopentene added to((C5Me5)YH)2 dissolved in C6D11CD3 at -196°C; mixt. warmed to 0. degree.C; monitored by NMR (about 4 h); | 85% |

| Conditions | Yield |
|---|---|
| In diethyl ether for 8h; Cycloaddition; Heating; | 84.7% |
-
-
67-56-1
methanol

-
-
693-89-0
1-methylcyclopent-1-ene

-
A
-
159335-79-2
7-Methoxy-7-methyl-1,2-dioxepan-3-ol

-
C
-
116972-39-5
1-hydroperoxy-1-methoxy-5-oxo-hexane

| Conditions | Yield |
|---|---|
| With ozone at -78℃; Yields of byproduct given; | A 84% B n/a C n/a |
| With ozone at -78℃; Yield given. Title compound not separated from byproducts; | A 84% B n/a C n/a |

1-Methylcyclopentene Specification
The IUPAC name of 1-Methylcyclopentene is 1-Methylcyclopentene. With the CAS registry number 693-89-0, it is also named as 1-Methyl-1-cyclopentene. The product's categories are Alkenes; Cyclic and Organic Building Blocks. Besides, it is clear colourless liquid, which should be stored in sealed containers and placed in a cool and dry place. And the place is away from oxidizing agents. In addition, its molecular formula is C6H10 and molecular weight is 82.14.
The other characteristics of this product can be summarized as: (1)EINECS: 211-762-0; (2)ACD/LogP: 2.92; (3)# of Rule of 5 Violations: 0; (4)ACD/LogD (pH 5.5): 2.92; (5)ACD/LogD (pH 7.4): 2.92; (6)ACD/BCF (pH 5.5): 97.68; (7)ACD/BCF (pH 7.4): 97.68; (8)ACD/KOC (pH 5.5): 924.51; (9)ACD/KOC (pH 7.4): 924.51; (10)H bond acceptors: 0; (11)H bond donors: 0; (12)Freely Rotating Bonds: 0; (13)Index of Refraction: 1.459; (14)Molar Refractivity: 27.36 cm3; (15)Molar Volume: 99.9 cm3; (16)Surface Tension: 25.4 dyne/cm; (17)Density: 0.821 g/cm3; (18)Flash Point: 75-77 °C; (19)Melting Point: -142 °C; (20)Enthalpy of Vaporization: 30.61 kJ/mol; (21)Boiling Point: 78.4 °C at 760 mmHg; (22)Vapour Pressure: 103 mmHg at 25 °C.
Preparation of 1-Methylcyclopentene: this chemical can be prepared by Methylenecyclopentane.

This reaction needs liquid Sulphur dioxide at ambient temperature. The yield is 99 %.
Uses of 1-Methylcyclopentene: this chemical is used in organic synthesis. Furthermore, it can react with Diiodomethane to get 1-Methylbicyclo[3.1.0]hexan.
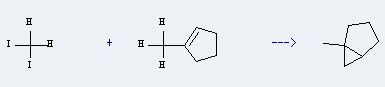
This reaction needs ZnEt2 and Cyclohexane at ambient temperature. The reaction time is 5 days. The yield is 86 %.
When you are using this chemical, please be cautious about it as the following: 1-Methylcyclopentene is highly flammable. Please keep away from sources of ignition. Moreover, it is harmful that may cause lung damage if swallowed. Please keep container tightly closed. Additionally, if swallowed, do not induce vomitting, please seek medical advice immediately and show this container or label.
People can use the following data to convert to the molecule structure.
(1)SMILES:C\1=C(/C)CCC/1
(2)InChI:InChI=1/C6H10/c1-6-4-2-3-5-6/h4H,2-3,5H2,1H3
(3)InChIKey:ATQUFXWBVZUTKO-UHFFFAOYAO
(4)Std. InChI:InChI=1S/C6H10/c1-6-4-2-3-5-6/h4H,2-3,5H2,1H3
(5)Std. InChIKey:ATQUFXWBVZUTKO-UHFFFAOYSA-N
Related Products
- 1-Methylcyclopentene
- 6938-94-9
- 69390-17-6
- 69390-33-6
- 69390-34-7
- 69390-36-9
- 69390-37-0
- 69390-40-5
- 69390-41-6
- 69390-43-8
- 69390-44-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View