-
Name
1 -Methylnaphthalene
- EINECS 201-966-8
- CAS No. 90-12-0
- Article Data339
- CAS DataBase
- Density 1.016 g/cm3
- Solubility Water: 0.026 g/L (25 °C)
- Melting Point -22 °C
- Formula C11H10
- Boiling Point 242.8 °C at 760 mmHg
- Molecular Weight 142.2
- Flash Point 82.2 °C
- Transport Information UN 3082 9/PG 3
- Appearance Colorless liquid
- Safety 7-26-36/37/39-61-45-36/37-23-36
- Risk Codes 22-36/37/38-42/43-51/53-39/23/24/25-23/24/25-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, N,
N, T
T
- Synonyms 1-Methylnaphthalene;Naphthalene, alpha-methyl-;Methynaph H;NSC 3574;a-Methylnaphthalene;alpha-Methylnaphthalene;AI3-15378;CCRIS 6151;
- PSA 0.00000
- LogP 3.14820
Synthetic route

| Conditions | Yield |
|---|---|
| In benzene at 10℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In 1,4-dioxane for 2h; Heating; | 100% |
| Stage #1: 1-Bromonaphthalene; dimethyl zinc(II) With n-butyllithium In tetrahydrofuran; diethyl ether; hexane at 0℃; for 0.5h; Substitution; Stage #2: With oxovanadium(V) ethoxydichloride In tetrahydrofuran; diethyl ether; hexane at 20℃; for 3h; Oxidation; Alkylation; Further stages.; | 73% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; for 15h; | 99% |

| Conditions | Yield |
|---|---|
| With potassium formate; PdCl22 In benzene at 60℃; for 4h; | 98% |
| With potassium formate; PdCl22 In benzene at 60℃; for 4h; Product distribution; further catalyst, reagent; solid or aqueous solution of reagent; | 98% |
| With sodium tetrahydroborate; water In methanol at 20℃; for 0.333333h; | 94% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; for 12h; | 98% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; {MoPdcp[μ-(CO)2][μ3-(CO)](PPh3)}2 In benzene at 78℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-Iodonaphthalene With [K{(Me)Ni[N(SiMe3)2,6-diisopropylphenyl]2}] In tetrahydrofuran at -35℃; for 0.333333h; Inert atmosphere; Stage #2: methylmagnesium bromide In tetrahydrofuran at 20℃; for 1.5h; Reagent/catalyst; Inert atmosphere; | 98% |
-
-
1351445-39-0
N,N,N-trimethyl-1-naphthalenaminium trifluoromethanesulfonate

-
-
75-24-1
trimethylaluminum

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,4-dioxane at 110℃; for 24h; Reagent/catalyst; Solvent; Temperature; Time; Schlenk technique; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With nickel In ethanol for 4h; Heating; | 95% |
| With nickel In ethanol for 4h; Heating; | 95% |
-
-
118226-43-0
N,N-dimethyl-N-(1,4-dihydronaphthyl-1-methyl)amine oxide

-
A
-
40476-27-5
1-methylene-1,4-dihydronaphthalene (benzo-p-isotoluene)

-
B
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| at 85 - 105℃; under 0.5 Torr; | A 95% B 5% |

| Conditions | Yield |
|---|---|
| With Me3Co(CN)2Li3 In tetrahydrofuran for 18h; Product distribution; for -78 deg C to r.t., variation of reagent; | A 4% B 95% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; for 17h; | A 3% B 95% |


| Conditions | Yield |
|---|---|
| bis(triphenylphosphine)nickel(II) chloride In benzene at 80℃; for 5h; Substitution; | 95% |
-
-
85924-73-8
C11H7Li

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol under 1034.3 Torr; for 2h; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: methyllithium With indium(III) chloride In tetrahydrofuran Stage #2: 1-Iodonaphthalene With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran Heating; | 94% |
| bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran; diethyl ether at 20℃; for 24h; | 113 mg |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonato)palladium(II); 18-crown-6 ether; potassium phosphate tribasic trihydrate; 2-(2,6-dimethylphenyl)-5-(2,4,6-triisopropylphenyl)imidazo[1,5-a]pyridin-2-ium chloride In 1,4-dioxane at 130℃; for 48h; Suzuki Coupling; Sealed tube; Inert atmosphere; | 94% |
| With potassium phosphate tribasic trihydrate; Tris(3,6-dioxaheptyl)amine; 2-(2,6-dimethylphenyl)-5-(2,4,6-triisopropylphenyl)imidazo[1,5-a]pyridin-2-ium chloride; palladium(II) acetylacetonate In 1,4-dioxane at 20 - 130℃; for 48.5h; Suzuki-Miyaura Coupling; Sealed tube; Inert atmosphere; | 87% |
| With dicyclohexyl-(2′,4′,6′-triisopropyl-3,6-dimethoxy-[1,1′-biphenyl]-2-yl)phosphine; palladium diacetate; caesium carbonate In toluene at 150℃; for 24h; Inert atmosphere; | 55% |

| Conditions | Yield |
|---|---|
| bis(tricyclohexylphosphine)nickel(II) dichloride In diethyl ether; toluene at 80℃; for 0.333333h; | 93% |
| With C30H36NiOP2 In tetrahydrofuran at 25℃; for 16h; Inert atmosphere; Schlenk technique; | 78% |

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; palladium diacetate; XPhos In tert-butyl alcohol at 50℃; for 12h; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 93% |
-
-
512-56-1
trimethyl phosphite

-
-
68716-52-9
1-naphthylboronic acid pinacol ester

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; lithium iodide; lithium tert-butoxide at 50℃; for 16h; Catalytic behavior; Kinetics; Reagent/catalyst; Temperature; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; water In methanol at 20℃; for 0.25h; | 92% |
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene Heating; | 89% |
| With TETRAHYDROPYRANE; 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride for 2h; Heating; | 82% |
| With phosphonic Acid; iodine In 1,2-dichloro-ethane at 120℃; for 36h; Inert atmosphere; | 78% |
| With ethanol; platinum Hydrogenolyse; |

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With TpRu(PPh3)(CH3CN)2PF6 In toluene at 110℃; for 6h; | 92% |
| tungsten pentacarbonyl tetrahydrofuran In tetrahydrofuran at 20℃; for 24h; Cyclization; | 83% |

| Conditions | Yield |
|---|---|
| bis(tricyclohexylphosphine)nickel(II) dichloride In diethyl ether; toluene at 80℃; for 0.333333h; | 92% |
| With tricyclohexylphosphine In toluene at 40℃; for 2h; Inert atmosphere; | 93 %Chromat. |

| Conditions | Yield |
|---|---|
| With palladium; triphenylphosphine In tetrahydrofuran at 70℃; for 12h; Catalytic behavior; Negishi Coupling; Inert atmosphere; Green chemistry; | 92% |

| Conditions | Yield |
|---|---|
| With ethoxyethoxyethanol; hydrazine hydrate; potassium hydroxide at 220℃; under 10343.2 Torr; Wolff-Kishner Reduction; Sonication; | 91% |
| With W-7 Raney-Nickel In ethanol for 2h; Heating; | 89% |
| With sodium hydroxide; water; hydrazine hydrate In ethylene glycol for 3h; Reduction; Heating; | 54% |
| With triethylsilane; tris(pentafluorophenyl)borate In dichloromethane |
-
-
90-11-9
1-Bromonaphthalene

-
-
60228-61-7
bis{bis(trimethylsilyl)amido}(methyl)(iodo)tin(IV)

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 101℃; for 4h; Stille reaction; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene With bis(tri-t-butylphosphine)palladium(0); oxygen In toluene at 20℃; for 0.0166667h; Schlenk technique; Stage #2: methyllithium In toluene at 20℃; for 0.0333333h; Catalytic behavior; Reagent/catalyst; Schlenk technique; | 91% |
| With bis(tri-t-butylphosphine)palladium(0); water; sodium chloride In diethyl ether at 20℃; for 0.00555556h; pH=6; Schlenk technique; | 85% |
-
-
63171-65-3
4-Hydro-1-methylnaphtalene-1-carboxylic acid

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | 90% |
-
-
137203-34-0
bis(trimethylaluminum)–1,4-diazabicyclo[2.2.2]octane adduct

-
-
90-14-2
1-Iodonaphthalene

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); dicyclohexyl(2',6'-diisopropyl-4'-sulfobiphenyl-2-yl)phosphonium hydrogen sulfate In tetrahydrofuran at 65℃; for 2h; Inert atmosphere; Ionic liquid; chemoselective reaction; | 90% |
-
-
87306-73-8
2-Methyl-2-[(naphthalen-1-ylmethyl)-amino]-propan-1-ol

-
-
90-12-0
1-Methylnaphthalene

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol under 2585.7 Torr; for 8h; Ambient temperature; | 88% |

| Conditions | Yield |
|---|---|
| With 1,2-bis(diphenylphosphino)ethane nickel(II) chloride In tetrahydrofuran at 65℃; for 24h; Kumada Cross-Coupling; Inert atmosphere; Schlenk technique; | 87% |
| With cobalt (III) fluoride; N,N′-bis(2,6-diisopropylphenyl)imidazol-2-ylidene hydrochloride In tetrahydrofuran at 80℃; for 5h; | 82% |
| With iron(III)-acetylacetonate In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 23℃; for 1h; |
-
-
98-88-4
benzoyl chloride

-
-
90-12-0
1-Methylnaphthalene

-
-
116672-26-5
(4-methylnaphthalen-1-yl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane for 1h; Friedel-Crafts Acylation; | 100% |
| With sulfated zirconia In chlorobenzene at 130℃; for 20h; | 53% |
| With carbon disulfide; aluminium trichloride at 0 - 10℃; | |
| With carbon disulfide; aluminium trichloride |

| Conditions | Yield |
|---|---|
| With aluminum oxide; copper(ll) bromide In tetrachloromethane at 50℃; for 2h; | 100% |
| With benzyltrimethylazanium tribroman-2-uide; zinc(II) chloride In acetic acid for 3h; Ambient temperature; | 99% |
| With N-Bromosuccinimide In acetonitrile at 30 - 40℃; for 12h; | 98% |
-
-
90-12-0
1-Methylnaphthalene

-
-
17075-39-7
4-chloro-1-methyl-naphthalene

| Conditions | Yield |
|---|---|
| With aluminum oxide; copper dichloride In chlorobenzene at 130℃; for 2h; | 100% |
| With lead(IV) acetate; tin(IV) chloride In dichloromethane for 0.25h; | 85% |
| With benzyltrimethylazanium tetrachloro-λ3-iodanuide In acetic acid at 70℃; for 18h; | 73% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; copper(ll) bromide In tetrachloromethane at 80℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide; Triphenylsilyl chloride In chlorobenzene at 20℃; under 22502.3 Torr; for 3h; Autoclave; regioselective reaction; | 99% |
| With aluminum tri-bromide In benzene at 40℃; under 22501.8 Torr; for 24h; | 33% |
-
-
90-12-0
1-Methylnaphthalene

-
-
73453-32-4, 73453-33-5, 19224-41-0
4,4'-dimethyl-1,1'-binaphthalene

| Conditions | Yield |
|---|---|
| With air; trifluorormethanesulfonic acid; sodium nitrite In acetonitrile at 0℃; for 1h; Product distribution; Mechanism; effect of further reagents; | 97% |
| With air; trifluorormethanesulfonic acid; sodium nitrite In acetonitrile at 0℃; for 1h; | 97% |
| With boron trifluoride diethyl etherate; thallium(III) trifluoroacetate In tetrachloromethane Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With Acetyl bromide; silica gel; zinc dibromide In benzene at 50℃; for 3h; | 97% |

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; N-hydroxyphthalimide; palladium diacetate In acetonitrile at 70℃; for 24h; Inert atmosphere; Sealed tube; | 97% |
| With tert.-butylhydroperoxide; ammonium fluoride; iodine; oxygen In water; dimethyl sulfoxide at 70℃; for 48h; Reagent/catalyst; Solvent; Time; Schlenk technique; Sealed tube; | 80% |
| Stage #1: 1-Methylnaphthalene With hydrogen bromide; dihydrogen peroxide In tetrachloromethane; water at 20℃; for 1h; Irradiation; Stage #2: With ammonia; iodine In tetrachloromethane; water; acetonitrile at 20 - 60℃; for 4h; | 73% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; dibenzoyl peroxide In tetrachloromethane | 96% |
| With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In benzene at 80℃; for 2.5h; Inert atmosphere; | 95% |
| With N,N,N’,N’-tetrabromobenzene-1,3-disulfonamide; dibenzoyl peroxide In ethyl acetate for 4.5h; Reflux; | 90% |
-
-
64-19-7
acetic acid

-
-
90-12-0
1-Methylnaphthalene

-
A
-
13098-88-9
naphthalen-1-ylmethyl acetate

-
B
-
66-77-3
1-naphthaldehyde

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In water at 85℃; for 2h; | A 3% B 95% |


| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In water; acetic acid at 85℃; for 2h; | A 3% B 95% |
| With *5H2O; lithium chloride In acetic acid at 100℃; for 27h; | A 35% B 39% |
-
-
90-12-0
1-Methylnaphthalene

-
-
20601-22-3
2-bromo-1-methylnaphthalene

| Conditions | Yield |
|---|---|
| With hydrogen bromide; dihydrogen peroxide In water at 20℃; for 24h; Darkness; | 94% |
| With hydrogen bromide; dihydrogen peroxide In water at 20℃; for 64h; Darkness; | 81% |
1-Methylnaphthalene Consensus Reports
1-Methylnaphthalene Specification
The 1-Methylnaphthalene with CAS registry number of 90-12-0 is also known as Naphthalene, alpha-methyl-. The IUPAC name and product name are the same. It belongs to product categories of Alpha Sort; Chemical Class; Hydrocarbons; M; MAlphabetic; Meta-methAnalytical Standards; NaphthalenesChemical Class; Neats; Volatiles/ Semivolatiles; Arenes; Building Blocks; Organic Building Blocks; Alphabetical Listings; Flavors and Fragrances; M-N; Naphthalenes. Its EINECS registry number is 201-966-8. In addition, the formula is C11H10 and the molecular weight is 142.20. This chemical is a colorless liquid and should be stored in ventilated and dry room below 30 °C.1-Methylnaphthalene is sensitive to heat. Reacts with strong oxidizing agents. Incompatible with oxygen and peroxides .
Physical properties about 1-Methylnaphthalene are:
(1)ACD/LogP: 3.91; (2)ACD/LogD (pH 5.5): 3.91; (3)ACD/LogD (pH 7.4): 3.91; (4)ACD/BCF (pH 5.5): 550.66; (5)ACD/BCF (pH 7.4): 550.66; (6)ACD/KOC (pH 5.5): 3188.07; (7)ACD/KOC (pH 7.4): 3188.07; (8)Index of Refraction: 1.616; (9)Molar Refractivity: 48.92 cm3; (10)Molar Volume: 139.8 cm3; (11)Surface Tension: 38.7 dyne/cm; (12)Density: 1.016 g/cm3; (13)Flash Point: 82.2 °C; (14)Enthalpy of Vaporization: 46.03 kJ/mol; (15)Boiling Point: 242.8 °C at 760 mmHg; (16)Vapour Pressure: 0.0518 mmHg at 25 °C.
Preparation of 1-Methylnaphthalene:
It is exist in high temperature tar about 0.8%-1.2%. Phenol and pyridine base are removed from the fraction of 230-300 °C. Then the fraction is distillated to collect fraction at 240-245 °C. At last, the fraction is cooled to obtain the product.
Uses of 1-Methylnaphthalene:
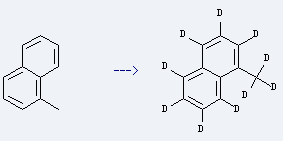
It is used as raw material of surfactant, water reducer, dispersant, drugs and other organic synthesis. It is used to produce 1-methylnaphthalene-d10. The reaction occurs with reagents deuterium oxide, sodium deuteroxide and other condition of supercritical pressure at 420-430 °C for 24 hours. The yield is about 75%.
Safety information of 1-Methylnaphthalene:
When you are using 1-Methylnaphthalene, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. It is harmful by inhalation, in contact with skin and if swallowed. What's more, may cause sensitisation by inhalation and skin contact. It also is toxic to aquatic organisms and may cause long-term adverse effects in the aquatic environment. During using it, wear suitable protective clothing, gloves and eye/face protection. Keep container tightly closed and avoid release to the environment. Do not breathe gas/fumes/vapour/spray. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If accident happens or you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: CC1=CC=CC2=CC=CC=C12
2. InChI: InChI=1S/C11H10/c1-9-5-4-7-10-6-2-3-8-11(9)10/h2-8H,1H3
3. InChIKey: QPUYECUOLPXSFR-UHFFFAOYSA-N
The toxicity data of 1-Methylnaphthalene is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LDLo | skin | 7500mg/kg (7500mg/kg) | National Technical Information Service. Vol. OTS0556357, | |
| rat | LD50 | oral | 1840mg/kg (1840mg/kg) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 85, 1982. |
Related Products
- 1-Methylnaphthalene
- 1-Methylnaphthalene-4-boronic acid
- 1-Methylnaphthalene-D10
- 9012-09-3
- 9012-31-1
- 9012-33-3
- 9012-36-6
- 9012-37-7
- 9012-54-8
- 9012-59-3
- 9012-63-9
- 9012-72-0
- 9012-76-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View