-
Name
2,2'-Dithiosalicylic acid
- EINECS 204-352-8
- CAS No. 119-80-2
- Article Data162
- CAS DataBase
- Density 1.53 g/cm3
- Solubility insoluble in water
- Melting Point 287-290 °C
- Formula C14H10O4S2
- Boiling Point 493.1 °C at 760 mmHg
- Molecular Weight 306.363
- Flash Point 252 °C
- Transport Information
- Appearance off white to light yellow powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2, 2-Dithiobis[benzoic acid];Bis(o-carboxylphenyl) disulfide;Bis(2-carboxyphenyl) disulfide;Diphenyldisulfide-2,2-dicarboxylic acid;2,2-Dithiobis(benzoic acid);Benzoic acid, 2,2-dithiobis-;2,2-Dithiodi(benzoic acid);2-(2-carboxylatophenyl)disulfanylbenzoate;2-(2-carboxyphenyl)disulfanylbenzoic acid;Bis(o-carboxyphenyl) disulfide;2,2'-Dithiosalicy acid;Benzoic acid, 2,2-dithiodi-;2-[(2-Carboxyphenyl)disulfanyl]benzoic acid;2,2'-Dithio Dibenzoic Acid;2,2-Dithiosalicylic acid;
- PSA 125.20000
- LogP 3.88240
Synthetic route

| Conditions | Yield |
|---|---|
| With xenon difluoride In dichloromethane at 25℃; | 99% |
| With 1-oxa-2-azaspiro[2.5]octane In toluene | 99% |
| With polyvinylpolypyrrolidonium tribromide In ethanol at 20℃; for 1.5h; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With nickel(II) chloride hexahydrate; morpholinium morpholine-1-carbodithioate; potassium hydroxide In water; ethylene glycol; N,N-dimethyl-formamide at 130℃; | 96% |
| With potassium sulfide; nickel(II) chloride hexahydrate; acetylacetone; potassium hydroxide In water; N,N-dimethyl-formamide at 110℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| With indium(III) oxide; ammonia; water; sulfur In ethanol at 60℃; Green chemistry; | 82% |
| With copper(l) iodide; tetra(n-butyl)ammonium hydroxide; sulfur In water at 80℃; for 48h; Inert atmosphere; Sealed tube; chemoselective reaction; |

| Conditions | Yield |
|---|---|
| With ozone; acetic acid at 16.84℃; | 26.7% |

| Conditions | Yield |
|---|---|
| With ozone; acetic acid at 16.84℃; | 14% |
-
-
19602-82-5
2,2'-dithiodibenzoic acid dichloride

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| bei laengerem Liegen an feuchter Luft; |
-
-
493-57-2
benzo-[b]thiophene-2,3-dione

-
A
-
16807-21-9
1,2-benzisothiazole-3-carboxamide

-
B
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; dihydrogen peroxide |

| Conditions | Yield |
|---|---|
| With sodium ethanolate |

| Conditions | Yield |
|---|---|
| With alkali | |
| With ethanol; sodium |
-
-
13993-58-3
4-chloro-3,5-diiodo-pyridine

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
-
147-93-3
Thiosalicylic acid

-
A
-
7153-08-4
4-hydroxy-3,5-diiodopyridine

-
C
-
119-80-2
2,2'-dithiobenzoic acid

-
-
784-62-3
1-thioflavone

-
-
141-52-6
sodium ethanolate

-
A
-
119-80-2
2,2'-dithiobenzoic acid

-
B
-
26824-02-2
1-(2-mercaptophenyl)ethan-1-one

-
C
-
147-93-3
Thiosalicylic acid

-
D
-
98-86-2
acetophenone

-
-
17333-86-7
o-Carboxybenzenediazonium

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With sodium disulfide | |
| With sodium disulfide |
-
-
623-11-0
1-methyl-4-nitrosobenzene

-
-
25803-71-8
2-[(2-oxo-2-phenylethyl)thio]-benzoic acid

-
-
119-80-2
2,2'-dithiobenzoic acid
-
-
13165-80-5
o-(hydroxysulfinyl)benzoic acid

-
A
-
632-25-7
2-sulfobenzoic acid

-
B
-
119-80-2
2,2'-dithiobenzoic acid
-
-
13165-80-5
o-(hydroxysulfinyl)benzoic acid

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; tin(ll) chloride |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
60-29-7
diethyl ether

-
-
108-98-5
thiophenol

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| anschliessendes Behandeln mit festem CO2; |

-
-
32147-20-9
(Z)-2-bromo-1,3-diphenylprop-2-en-1-one

-
-
147-93-3
Thiosalicylic acid

-
A
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With ethanol; sodium carbonate; acetone |

-
-
147-93-3
Thiosalicylic acid

-
-
2032-35-1
Bromoacetaldehyde diethyl acetal

-
A
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With ethanol; sodium acetate |

-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
60-29-7
diethyl ether

-
-
882-33-7
diphenyldisulfane

-
A
-
1126-80-3
(n-butylthio)benzene

-
B
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| anschliessendes Behandeln mit festem CO2; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; copper |
-
-
36665-23-3
benzo[d]-1,3-oxathiin-4-one 1-oxide

-
A
-
50-00-0
formaldehyd

-
B
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6 at 110℃; for 10h; |

| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) hexane, r.t., overnight, 2.) r.t.; Yield given. Multistep reaction; |
-
-
118-91-2
ortho-chlorobenzoic acid

-
A
-
119-80-2
2,2'-dithiobenzoic acid

-
B
-
147-93-3
Thiosalicylic acid

-
C
-
65-85-0
benzoic acid

-
D
-
69-72-7
salicylic acid

| Conditions | Yield |
|---|---|
| With sulfur In melt at 270℃; Product distribution; effect of various molten salts, amount of sulfur, molar ratio of sulfur to molten salts, composition of molten salt and various mercaptylation agents; |

-
-
124-38-9
carbon dioxide

-
-
622-38-8
Ethyl phenyl sulfide

-
-
629-19-6
Dipropyl disulfide

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; n-butyllithium Yield given. Multistep reaction; |


| Conditions | Yield |
|---|---|
| With triethylamine for 18h; | 100% |
| Stage #1: 2,2'-dithiobenzoic acid With thionyl chloride for 2h; Reflux; Stage #2: methanol With triethylamine for 2h; Reflux; | 92% |
| With sulfuric acid for 24h; Reflux; | 91% |

| Conditions | Yield |
|---|---|
| 99.5% | |
| With sulfuric acid at 10 - 40℃; for 4 - 5h; | 73.8% |
| With sulfuric acid at -5 - 50℃; for 58h; | 53.8% |
| With sulfuric acid at -5 - 50℃; for 58h; | |
| With sulfuric acid at 20℃; for 2h; |
-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
106-48-9
4-chloro-phenol

-
-
59803-22-4
1-chloro-4-hydroxy-9H-thioxanthen-9-one

| Conditions | Yield |
|---|---|
| With sulfuric acid at -5 - 70℃; for 21h; | 99% |
| Stage #1: 2,2'-dithiobenzoic acid With sulfuric acid In water for 0.5h; Stage #2: 4-chloro-phenol In water at 40 - 100℃; for 5h; | 77.9% |
| In sulfuric acid | 69.7% |

| Conditions | Yield |
|---|---|
| With chlorine; sulfur In 1,1,2,2-tetrachloroethylene at 70℃; Temperature; Solvent; | 99% |
| Stage #1: 2,2'-dithiobenzoic acid With thionyl chloride; 1-methyl-pyrrolidin-2-one at 80℃; Heating / reflux; Stage #2: With sulfuryl dichloride at 50℃; for 0.5h; | 93% |
| With thionyl chloride; chlorine; N,N-dimethyl-formamide 1.) 1,2-dichloroethane, reflux, 1 h, 2.) 10 deg C, 20 min; Multistep reaction; |
-
-
98-82-8
Isopropylbenzene

-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
5495-84-1
2-isopropyl-9H-thioxanthen-9-one

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 0.5h; | 99% |
| With methanesulfonic acid; zinc In chlorobenzene at 115℃; | 89.5% |
-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
19602-82-5
2,2'-dithiodibenzoic acid dichloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In N,N-dimethyl-formamide; toluene at 82℃; for 20h; | 97% |
| With thionyl chloride In N,N-dimethyl-formamide for 4h; Reflux; | 95% |
| With thionyl chloride; N,N-dimethyl-formamide In toluene at 82℃; for 20h; | 91.3% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; for 48h; Reagent/catalyst; Solvent; Temperature; Green chemistry; | 95% |
| Stage #1: 2,2'-dithiobenzoic acid With ammonium peroxydisulfate In methanol at 70℃; for 3h; Stage #2: indole In methanol at 20 - 70℃; | 46% |

| Conditions | Yield |
|---|---|
| With tetraphosphorus decasulfide In 1,4-dioxane for 48h; Inert atmosphere; Reflux; | 93% |
| With phosphorous (V) sulfide; xylene | |
| With Lawessons reagent In benzene Heating; Yield given; | |
| With tetraphosphorus decasulfide In pyridine for 1h; Heating; | |
| With pyridine; tetraphosphorus decasulfide |
-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In neat (no solvent) at 20℃; for 0.5h; Reagent/catalyst; chemoselective reaction; | 92% |
| With dihydrogen peroxide In neat (no solvent) at 20℃; for 0.5h; Reagent/catalyst; Green chemistry; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 90 - 95℃; for 5h; Substitution; | 91% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; Green chemistry; | 91% |
-
-
141-93-5
m-diethylbenzene

-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
82799-44-8
2,4‐diethyl‐9H‐thioxanthene‐9‐one

| Conditions | Yield |
|---|---|
| With sulfuric acid at 0 - 65℃; for 6h; Reagent/catalyst; | 90.2% |
| With aluminum oxide; iron; toluene-4-sulfonic acid at 130℃; Reagent/catalyst; | 89% |
-
-
6066-82-6
1-hydroxy-pyrrolidine-2,5-dione

-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
497262-13-2
N,N'-disuccinimidyl-2,2'-dithiosalicylate

| Conditions | Yield |
|---|---|
| With diisopropyl-carbodiimide In tetrahydrofuran; isopropyl alcohol at 20℃; for 4h; | 89% |
| With diisopropyl-carbodiimide In tetrahydrofuran; isopropyl alcohol at 25℃; for 4h; | 89% |
| With diisopropyl-carbodiimide In tetrahydrofuran; isopropyl alcohol at 25℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; Green chemistry; | 89% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 90 - 95℃; for 5h; Substitution; | 87% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; Green chemistry; | 86% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrogen iodide; hypophosphorous acid for 3h; Heating; | 85% |
| With hydrogenchloride; diphenylphosphinopolystyrene In tetrahydrofuran for 7h; Heating; | 73% |
| With tin; acetic acid weiteres Reagens: konz. Salzsaeure; |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 336000h; | 85% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; Green chemistry; | 85% |
-
-
66-71-7
1,10-Phenanthroline

-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; pH=7.2; | A 85% B 15% |


| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol; dicyclohexyl-carbodiimide In dichloromethane; N,N-dimethyl-formamide at 20℃; for 3h; | 83% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 130℃; Green chemistry; | 83% |
-
-
119-80-2
2,2'-dithiobenzoic acid

-
A
-
19602-82-5
2,2'-dithiodibenzoic acid dichloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In n-heptane | A 80% B n/a |

-
-
119-80-2
2,2'-dithiobenzoic acid

| Conditions | Yield |
|---|---|
| With N(CH2CH3)3 In water High Pressure; heating mixt. of gadolinium compd., copper compd., benzoic acid deriv., deionized water and triethylamine at 170°C for 72 h; cooling to room temp. at rate of 5°C/h, isolation of crystals, washing deionized water, elem. anal.; | 80% |

-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
122-59-8
2-phenoxyacetic acid

-
-
84434-05-9
2-[(9-oxo-9H-thioxanthen-2-yl)oxy]-acetic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 0 - 25℃; for 1h; | 76% |
-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
13515-97-4
methyl 2-aminopropanoate monohydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2,2'-dithiobenzoic acid With 1,1'-carbonyldiimidazole In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 0.666667h; Reflux; Stage #2: methyl 2-aminopropanoate monohydrochloride With triethylamine In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 48h; | 76% |
-
-
119-80-2
2,2'-dithiobenzoic acid

-
-
35190-71-7
1,2-bis(2-(hydroxymethyl)phenyl)disulfane

| Conditions | Yield |
|---|---|
| With borane-THF; boron trifluoride diethyl etherate In tetrahydrofuran at 20℃; for 18h; | 75% |
| With sodium bis(2-methoxyethoxy)aluminium dihydride In benzene |
2,2'-Dithiosalicylic acid Chemical Properties
Molecule structure of 2,2'-Dithiodibenzoic acid (CAS NO.119-80-2):
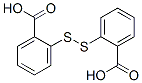
IUPAC Name: 2-(2-Carboxyphenyl)disulfanylbenzoic acid
Molecular Weight: 306.3568 g/mol
Molecular Formula: C14H10O4S2
Density: 1.53 g/cm3
Melting Point: 287-290 °C(lit.)
Boiling Point: 493.1 °C at 760 mmHg
Flash Point: 252 °C
Index of Refraction: 1.735
Molar Refractivity: 80.04 cm3
Molar Volume: 199.3 cm3
Polarizability: 31.73×10-24 cm3
Surface Tension: 83.8 dyne/cm
Enthalpy of Vaporization: 80.06 kJ/mol
Vapour Pressure: 1.54E-10 mmHg at 25 °C
Water Solubility: insoluble
XLogP3-AA: 3.1
H-Bond Donor: 2
H-Bond Acceptor: 4
Rotatable Bond Count: 5
Exact Mass: 306.00205
MonoIsotopic Mass: 306.00205
Topological Polar Surface Area: 74.6
Heavy Atom Count: 20
Complexity: 327
Canonical SMILES: C1=CC=C(C(=C1)C(=O)O)SSC2=CC=CC=C2C(=O)O
InChI: InChI=1S/C14H10O4S2/c15-13(16)9-5-1-3-7-11(9)19-20-12-8-4-2-6-10(12)14(17)18/h1-8H,(H,15,16)(H,17,18)
InChIKey: LBEMXJWGHIEXRA-UHFFFAOYSA-N
EINECS: 204-352-8
Product Categories: pharmacetical; organic acids
2,2'-Dithiosalicylic acid Uses
2,2'-Dithiodibenzoic acid (CAS NO.119-80-2) is used for intermediates of medicine, dyes, fungicides, and photoinitiators.
2,2'-Dithiosalicylic acid Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 367mg/kg (367mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 21, Pg. 284, 1971. |
2,2'-Dithiosalicylic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-24/25
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
RTECS: DG9660000
Hazard Note: Harmful / irritant
2,2'-Dithiosalicylic acid Specification
2,2'-Dithiodibenzoic acid (CAS NO.119-80-2) is also named as 2,2'-Dithiobis(benzoic acid) ; 2,2'-Dithiodibenzoesaeure ; 2,2'-Dithiodibenzoesaeure [German] ; 4-10-00-00279 (Beilstein Handbook Reference) ; AI3-15534 ; BRN 2221810 ; Bis(2-carboxyphenyl) disulfide ; Bis(o-carboxylphenyl) disulfide ; Bis(o-carboxyphenyl) disulfide ; Diphenyldisulfide-2,2'-dicarboxylic acid . 2,2'-Dithiodibenzoic acid is brown powder. 2,2'-Dithiodibenzoic acid is high toxic. It is flammable. It will produce toxic sulfur oxides fumes when buring. So the storage environment should be ventilate, low-temperature and dry. Keep 2,2'-Dithiodibenzoic acid separate from raw materials of food.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 119802-68-5
- 119807-84-0
- 1198095-99-6
- 1198096-23-9
- 1198097-53-8
- 1198103-73-9
- 1198103-89-7
- 1198106-25-0
- 1198106-53-4
- 1198107-00-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View