-
Name
2-IODOBENZALDEHYDE
- EINECS
- CAS No. 26260-02-6
- Article Data133
- CAS DataBase
- Density 1.883 g/cm3
- Solubility
- Melting Point 36-39 °C(lit.)
- Formula C7H5IO
- Boiling Point 266.3 °C at 760 mmHg
- Molecular Weight 232.021
- Flash Point 114.8 °C
- Transport Information
- Appearance white to light beige low melting crystalline
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzaldehyde,o-iodo- (6CI,7CI,8CI);2-Iodobenzaldehyde;o-Iodobenzaldehyde;
- PSA 17.07000
- LogP 2.10370
Synthetic route
-
-
5159-41-1
2-iodobenzyl alcohol

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide In chloroform at 20℃; for 70h; | 99% |
| With 1-methyl-1H-imidazole; tetrakis(acetonitrile)copper(I) trifluoromethanesulfonate; 4,4'-Dimethoxy-2,2'-bipyridin; 9-azabicyclo[3.3.1]nonane N-oxyl; oxygen In acetonitrile at 20℃; for 1h; | 98% |
| With silica gel; pyridinium chlorochromate In dichloromethane at 20℃; Inert atmosphere; | 98% |
-
-
6630-33-7
ortho-bromobenzaldehyde

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With copper(l) iodide; Trimethylenediamine; sodium iodide In 1,4-dioxane for 24h; Finkelstein Reaction; Reflux; Inert atmosphere; | 99% |
| Multi-step reaction with 3 steps 1: 83 percent / TsOH / 5 h / Heating 2: 1.) Grignard reagent, 2.) I2 / 1.), 2.) THF 3: 83 percent / aq. HCl / tetrahydrofuran / 20 h / Ambient temperature View Scheme | |
| With copper(l) iodide; sodium iodide; N,N`-dimethylethylenediamine In 1,4-dioxane at 120℃; under 1500.15 Torr; for 24h; Reagent/catalyst; Finkelstein Reaction; Autoclave; Inert atmosphere; Schlenk technique; |
-
-
80953-51-1
(2-iodophenyl)methyl acetate

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With methanol; potassium permanganate In ethyl acetate at 25℃; for 14h; | 98% |
| Multi-step reaction with 2 steps 1: aqueous KOH 2: AgNO3; Na2S2O8; water View Scheme |
-
-
35822-58-3
2-bromobenzaldehyde diethyl acetal

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromobenzaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78 - -10℃; for 1.41667h; Stage #2: With iodine In tetrahydrofuran; hexane at -78 - 20℃; for 12.6667h; | 98% |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With Iodine monochloride In dichloromethane at 20℃; for 2h; Inert atmosphere; | 94% |
-
-
88-67-5
2-Iodobenzoic acid

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; borane-THF Inert atmosphere; | 90% |
| Multi-step reaction with 2 steps 1: 40 percent / HCl / 30 h / Heating 2: 34 percent / diisobutylaluminum hydride / toluene / 2 h / -60 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 67 percent / SOCl2, DMF / 2 h / Heating 2: tert-butyl alcohol, LiAlH4 / bis-(2-methoxy-ethyl) ether / -78 - 20 °C View Scheme |
-
-
7022-46-0
2-Iodo-N-methyl-N-phenylbenzamide

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With zirconocene dichloride; lithium tri-t-butoxyaluminum hydride In tetrahydrofuran at 20℃; for 0.116667h; Inert atmosphere; | 90% |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With Iodine monochloride In dichloromethane at 20℃; for 2h; Inert atmosphere; | 90% |
| Multi-step reaction with 2 steps 1: acetonitrile / 10 h / 20 °C / pH 5 2: Iodine monochloride / dichloromethane / 2 h / 20 °C / Inert atmosphere View Scheme |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With Iodine monochloride In dichloromethane at 20℃; for 2h; Inert atmosphere; | 89% |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With Iodine monochloride In dichloromethane at 20℃; for 2h; Inert atmosphere; | 85% |
| Multi-step reaction with 2 steps 1: acetonitrile / 10 h / 20 °C / pH 5 2: Iodine monochloride / dichloromethane / 2 h / 20 °C / Inert atmosphere View Scheme |
-
-
152302-84-6
2-iodobenzaldehyde ethylene glycol ketal

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran for 20h; Ambient temperature; | 83% |
-
-
40138-16-7
2-formylbenzene boronic acid

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With perfluoroisopropyl iodide; copper; hydroquinone In N,N-dimethyl-formamide at 20℃; for 24h; | 83% |
-
-
294190-47-9
2-iodo-N-methoxy-N-methyl-benzamide

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 77% |
-
-
76464-87-4
C13H20IN

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene; sodium hydrogencarbonate In chloroform at 60℃; for 2h; Polonovski type reaction; | 75% |
-
-
380151-85-9
2-(2'-formylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With copper(I) oxide; 1,10-Phenanthroline; potassium iodide In methanol; water at 80℃; for 1h; High pressure; | 70% |
| With copper(I) oxide; 1,10-Phenanthroline; potassium iodide In methanol; water at 80℃; for 1h; Sealed tube; | 65% |
-
-
610-97-9
o-iodo-methyl-benzoic acid

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride In toluene at -60℃; for 2h; | 34% |
-
-
615-37-2
ortho-methylphenyl iodide

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With sulfuric acid bei der elektrolytischen Oxydation; | |
| Multi-step reaction with 4 steps 1: tetrachloromethane; dibenzoyl peroxide; N-bromo-succinimide 2: acetic acid 3: aqueous KOH 4: AgNO3; Na2S2O8; water View Scheme |
-
-
4387-36-4
2-iodobenzonitrile

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride; diethyl ether; tin(ll) chloride anschl. mit H2O; |
-
-
40400-13-3
2-Iodobenzyl bromide

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With chloroform; hexamethylenetetramine Erhitzen des Reaktionsprodukts mit wss. Essigsaeure; | |
| Multi-step reaction with 3 steps 1: acetic acid 2: aqueous KOH 3: AgNO3; Na2S2O8; water View Scheme |
-
-
15310-01-7
benodanil

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride; toluene at 100℃; Erwaermen des Reaktionsprodukts mit SnCl2 und aether. HCl und anschliessendes Behandeln mit Wasser; |
-
-
609-67-6
2-Iodobenzoyl chloride

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With tetrahydrogenoboratebis(triphenylphosphine)copper(I); triphenylphosphine In acetone for 1h; | 17.2 g |
| Multi-step reaction with 2 steps 1: 98 percent / sodium borohydride / dioxane / 1 h / steam-bath 2: 76.4 percent / pyridinium dichromate / CH2Cl2 / 3 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 2: toluene; PCl5 / 100 °C / Erwaermen des Reaktionsprodukts mit SnCl2 und aether. HCl und anschliessendes Behandeln mit Wasser View Scheme |
-
-
39959-51-8
2-iodobenzylamine

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With N-bromoacetamide In water at 14.9 - 34.9℃; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.); |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| In acetic acid at 19.9℃; Rate constant; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.), ΔG(excit.), var. temp.; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; tert-butyl alcohol In diethylene glycol dimethyl ether at -78 - 20℃; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
615-37-2
ortho-methylphenyl iodide

-
-
7664-93-9
sulfuric acid

-
A
-
88-67-5
2-Iodobenzoic acid

-
B
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| bei der elektrolytischen Oxydation; |


-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium iodide | |
| With potassium iodide |

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium permanganate |
-
-
121263-09-0
(2-iodo-benzylidene)-malonic acid

-
-
7732-18-5
water

-
A
-
141-82-2
malonic acid

-
B
-
124-38-9
carbon dioxide

-
C
-
26260-02-6
2-iodobenzaldehyde

-
D
-
90276-19-0
(E)-3-(2-Iodophenyl)propenoic acid

-
-
34824-58-3
2-(2-bromophenyl)-1,3-dioxolan

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) Grignard reagent, 2.) I2 / 1.), 2.) THF 2: 83 percent / aq. HCl / tetrahydrofuran / 20 h / Ambient temperature View Scheme |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
120-20-7
2-(3,4-dimethoxyphenyl)-ethylamine
-
-
26260-02-6
2-iodobenzaldehyde

-
-
100-63-0
phenylhydrazine

-
-
35713-98-5
2-iodo-benzaldehyde phenylhydrazone

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 160℃; for 0.166667h; microwave irradiation; | 100% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
4229-44-1
N-methylhydroxyamine hydrochloride

-
-
1037396-94-3
(Z)-N-(2-iodobenzylidene)methylamine N-oxide

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 20℃; for 2h; | 100% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
5680-79-5
glycine ethyl ester hydrochloride

-
-
1245604-64-1
methyl 2-[N-(2-iodobenzyl)amino]acetate

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid; triethylamine In dichloromethane at 20℃; for 19h; Inert atmosphere; | 100% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
623-33-6
glycine ethyl ester hydrochloride

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid; triethylamine In dichloromethane at 20℃; for 19h; Inert atmosphere; | 100% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
33670-32-5
methoxymethyltriphenylphosphonium bromide

| Conditions | Yield |
|---|---|
| Stage #1: methoxymethyltriphenylphosphonium bromide With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.25h; Inert atmosphere; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; | 100% |
-
-
116-11-0
2-Methoxypropene

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| With silica gel; acetic acid; tris(6,6,7,7,8,8,8-heptafluoro-2,2-dimethyl-3,5-octanedionato)ytterbium In dichloromethane at 25℃; Addition; | 99% |
| With tris(6,6,7,7,8,8,8-heptafluoro-2,2-dimethyl-3,5-octanedionato)ytterbium; silica gel; acetic acid at 25℃; | 97% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
88-67-5
2-Iodobenzoic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; tetrabutylammomium bromide; water In dichloromethane; acetone at 20℃; for 24h; | 99% |
| With perchloric acid; N-Chlorourethan; acetic acid In water at 29.9℃; for 10h; Rate constant; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.), ΔG(excit.); | |
| With perchloric acid; mercury(II) diacetate; N-bromoacetamide In acetic acid at 24.9℃; Kinetics; Mechanism; Thermodynamic data; other temperatures, ΔG(excit.), ΔH(excit.), ΔS(excit.); |

| Conditions | Yield |
|---|---|
| With potassium carbonate In water at 20℃; for 0.5h; Suzuki-Miyaura Coupling; | 99% |
| With potassium carbonate In ethanol; water at 20℃; for 1h; Suzuki-Miyaura Coupling; Green chemistry; | 94% |
| With potassium phosphate In 1,4-dioxane at 70℃; for 1.5h; Suzuki-Miyaura Coupling; | 93% |
-
-
931-53-3
Cyclohexyl isocyanide

-
-
26260-02-6
2-iodobenzaldehyde

-
-
64-19-7
acetic acid

-
-
109-73-9
N-butylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Ugi reaction; | 99% |

-
-
26260-02-6
2-iodobenzaldehyde

-
-
39687-95-1
isocyanoacetic acid methyl ester

-
-
64-19-7
acetic acid

-
-
459-73-4
GlyOEt*HCl

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Ugi reaction; | 99% |


| Conditions | Yield |
|---|---|
| Stage #1: (2-iodobenzyl)triphenylphosphonium bromide With potassium tert-butylate In tetrahydrofuran at 0℃; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran at 20℃; Wittig reaction; Further stages.; | 99% |
| With potassium hydroxide In chloroform; water at 20℃; for 2.5h; Reflux; | 92% |
-
-
1942-45-6
4-Octyne

-
-
26260-02-6
2-iodobenzaldehyde

-
-
134-32-7
1-amino-naphthalene

-
-
1099616-13-3
N-(2,3-dipropyl-1H-inden-1-yl)naphthalen-1-amine

| Conditions | Yield |
|---|---|
| With Co(dppe)Cl2; zinc In tetrahydrofuran; acetonitrile at 120℃; for 12h; Inert atmosphere; regioselective reaction; | 99% |

-
-
1942-45-6
4-Octyne

-
-
26260-02-6
2-iodobenzaldehyde

-
-
106-49-0
p-toluidine

-
-
1099615-80-1
2,3-dipropyl-N-p-tolyl-1H-inden-1-amine

| Conditions | Yield |
|---|---|
| With Co(dppe)Cl2; zinc In tetrahydrofuran; acetonitrile at 120℃; for 12h; Inert atmosphere; regioselective reaction; | 99% |

-
-
917-92-0
2,2-dimethyl-3-butyne

-
-
26260-02-6
2-iodobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 3,3-Dimethylbut-1-yne With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran at 20℃; for 3h; | 99% |
| Stage #1: 3,3-Dimethylbut-1-yne With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran; hexane at 20℃; for 4h; Stage #3: With ammonium chloride In tetrahydrofuran; hexane; water |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
100-58-3
phenylmagnesium bromide

-
-
134456-76-1
(2-iodophenyl)(phenyl)methanol

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 20℃; for 1h; Inert atmosphere; | 99% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
26260-02-6
2-iodobenzaldehyde

-
-
412272-54-9
ethyl 3-(2-iodophenyl)acrylate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran at 0 - 20℃; for 2h; | 99% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Horner-Wadsworth-Emmons Olefination; Inert atmosphere; Stage #2: 2-iodobenzaldehyde In tetrahydrofuran; mineral oil at 0 - 20℃; Horner-Wadsworth-Emmons Olefination; Inert atmosphere; | 85% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
26260-02-6
2-iodobenzaldehyde

-
-
378759-67-2
ethyl (E)-3-(2-iodophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In dichloromethane at 0℃; for 0.0833333h; Wittig-Horner reaction; Stage #2: 2-iodobenzaldehyde In dichloromethane at 0℃; for 0.5h; Wittig-Horner reaction; Further stages.; | 98.7% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran Stage #2: 2-iodobenzaldehyde In tetrahydrofuran at 20℃; | 92% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In dichloromethane; mineral oil at 0℃; for 0.0833333h; Inert atmosphere; Stage #2: 2-iodobenzaldehyde In dichloromethane; mineral oil at 0℃; for 2h; Inert atmosphere; | 89% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
1210-05-5
Biphenyl-2,2'-dicarbaldehyde

| Conditions | Yield |
|---|---|
| With copper(I) thiophene-2-carboxylate; copper In dimethyl sulfoxide at 65℃; Ullmann Condensation; | 98% |
| With indium; palladium 10% on activated carbon; tetrabutylammomium bromide; lithium chloride In water at 90℃; for 24h; Green chemistry; | 75% |
| With palladium diacetate; potassium carbonate In N,N-dimethyl-formamide at 100℃; for 24h; | 64% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
4301-14-8
acetylenemagnesium bromide

-
-
81758-22-7
1-(2-iodophenyl)prop-2-yn-1-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 2.5h; Inert atmosphere; Sealed tube; | 98% |
| In tetrahydrofuran for 2h; Ambient temperature; | 95% |
| In tetrahydrofuran at -60℃; for 14h; | 86% |
| In tetrahydrofuran at 0℃; Inert atmosphere; |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
133886-40-5
N-tosyl-N-allylpropargylamine

-
-
857026-45-0
N-allyl-N-(3-(2-formylphenyl)prop-2-yn-1-yl)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-iodobenzaldehyde With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine for 0.166667h; Inert atmosphere; Stage #2: N-tosyl-N-allylpropargylamine Inert atmosphere; | 98% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; potassium carbonate In tetrahydrofuran at 50℃; for 6h; Sonogashira coupling; | |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine Inert atmosphere; |
-
-
1942-45-6
4-Octyne

-
-
26260-02-6
2-iodobenzaldehyde

-
-
372-39-4
3,5-difluoroaniline

-
-
1099616-26-8
N-(3,5-difluorophenyl)-2,3-dipropyl-1H-inden-1-amine

| Conditions | Yield |
|---|---|
| With Co(dppe)Cl2; zinc In tetrahydrofuran; acetonitrile at 120℃; for 12h; Inert atmosphere; regioselective reaction; | 98% |

-
-
26260-02-6
2-iodobenzaldehyde

-
-
36646-78-3
3-(p-tolylamino)-5,5-dimethylcyclohex-2-enone

-
-
1196957-90-0
9-(4,4-dimethyl-6-oxo-2-(p-tolylamino)cyclohex-1-enyl)-3,3-dimethyl-10-(p-tolyl)-3,4,9,10-tetrahydroacridin-1(2H)-one

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; palladium diacetate; caesium carbonate In toluene at 110℃; for 12h; | 98% |
| With caesium carbonate In toluene at 110℃; for 24h; Inert atmosphere; | 86% |
-
-
26260-02-6
2-iodobenzaldehyde

-
-
33397-22-7
tri(naphthalen-1-yl)bismuth

-
-
142598-69-4
2-(naphthalene-1-yl)benzaldehyde

| Conditions | Yield |
|---|---|
| With potassium phosphate; copper(l) iodide; palladium diacetate; triphenylphosphine In N,N-dimethyl-formamide at 90℃; for 3h; | 98% |

| Conditions | Yield |
|---|---|
| With silver(I) acetate; copper diacetate; ethoxy(potassiosulfanyl)methanethione In N,N-dimethyl-formamide at 100℃; for 5h; | 98% |
-
-
766-98-3
1-ethynyl-4-fluorobenzene

-
-
26260-02-6
2-iodobenzaldehyde

-
-
1383549-86-7
3-(4-fluorophenyl)isoquinoline

| Conditions | Yield |
|---|---|
| With 1,2-bis(diphenylphosphino)ethane nickel(II) chloride; triethylamine In acetonitrile at 20℃; for 12h; Inert atmosphere; Schlenk technique; regioselective reaction; | 98% |
2-Iodobenzaldehyde Chemical Properties
Molecular Formula: C7H5IO
Molar mass: 232.02 g/mol
Density: 1.883 g/cm3
Flash Point: 114.8 °C
Index of Refraction: 1.668
Melting point: 36-39 °C(lit.)
Sensitive: Light Sensitive
Boiling Point: 266.3 °C at 760 mmHg
Vapour Pressure: 0.00873 mmHg at 25°C
Appearance: White to light beige low melting crystalline
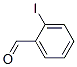
Structure of 2-Iodobenzaldehyde (26260-02-6) :
Product Categories of 2-Methyl-3-nitropyridine hydrochloride (63585-69-3) : Aldehydes;Benzaldehyde;Carbonyl Compounds;Iodine Compounds;Phenyls & Phenyl-Het;Aromatic Aldehydes & Derivatives (substituted);Adehydes, Acetals & Ketones
2-Iodobenzaldehyde Toxicity Data With Reference
Carcinogenicity of 2-Iodobenzaldehyde (26260-02-6) hasn't been listed as a carcinogen by NTP, IARC,ACGIH, or CA Prop 65. And its toxicological properties have not been fully investigated.
2-Iodobenzaldehyde Safety Profile
Hazard Codes:Xi![]()
Risk Statements:
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements:
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
2-Iodobenzaldehyde Specification
2-Iodobenzaldehyde (26260-02-6) is a compound which also can be called for benzaldehyde, 2-iodo- . 2-Iodobenzaldehyde (26260-02-6) is hazardous,so the first aid measures and others should be known .Such as: When on the skin: first,should flush skin with plenty of water immediatelyfor at least 15 minutes while removing contaminated clothing.Secondly,Get shoesmedical aid . Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.Then get medical aid soon.While ,it's Inhaled: Remove from exposure and move to fresh air immediately.Give artificial respiration While not breathing. when breathing is difficult, give oxygen.And as soon as to get medical aid.Then you have the ingesting of the product : Wash mouth out with water,and get medical aid immediately.Notes to Physician: Treat supportively and symptomatically.
In addition, 2-Iodobenzaldehyde (26260-02-6) can be stable under normal temperature and pressure conditions.It is not compatible with strong oxidizing agents and you must not take it with incompatible materials.And also prevent it to broken down into hazardous decomposition products: carbon dioxide,hydrogen iodide,carbon monoxide.
Related Products
- 2-Iodobenzaldehyde
- 262609-07-4
- 26264-02-8
- 26264-05-1
- 26264-06-2
- 26264-09-5
- 26264-14-2
- 26264-49-3
- 26264-58-4
- 26265-08-7
- 26266-57-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View