-
Name
2-Methyl-2-nitropropane
- EINECS 209-851-4
- CAS No. 594-70-7
- Article Data59
- CAS DataBase
- Density 0.95
- Solubility insoluble
- Melting Point 24 ºC
- Formula C4H9 N O2
- Boiling Point 126-127 ºC
- Molecular Weight 103.121
- Flash Point 19 ºC
- Transport Information UN 1993
- Appearance clear colourless liquid after melting
- Safety Potentially explosive when heated. When heated to decomposition it emits toxic fumes of NOx. See also NITRO COMPOUNDS.
- Risk Codes R11;R36/37/38
-
Molecular Structure
-
Hazard Symbols


- Synonyms 1,1-Dimethyl-1-nitroethane;2-Methyl-2-nitropropane; 2-Nitro-2-methylpropane; 2-Nitroisobutane; NSC 3651;Trimethylnitromethane
- PSA 45.82000
- LogP 1.58480
Synthetic route

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Na12[WZn3(H2O)2(ZnW9O34)2] at 75℃; for 7h; | 100% |
| With potassium permanganate In water 1.) 11 deg C -> room temperature, 8 h, 2.) 55 deg C, 18 h; | 78% |
| With sodium permanganate In hexane at 69℃; for 24h; | 76% |

| Conditions | Yield |
|---|---|
| With nitronium tetrafluoborate; silver carbonate In N,N-dimethyl acetamide at 90℃; for 12h; Inert atmosphere; Schlenk technique; regioselective reaction; | 79% |

| Conditions | Yield |
|---|---|
| With iodosylbenzene; meso-tetraphenylporphyrin iron(III) chloride In dichloromethane at 0℃; | 75% |
-
-
38442-51-2
tert-butylmercury chloride

-
-
594-70-7
2-Methyl-2-nitropropane

| Conditions | Yield |
|---|---|
| With potassium nitrite; 18-crown-6 ether In various solvent(s) for 2h; Irradiation; | 70% |
| With potassium nitrite; 18-crown-6 ether In dimethyl sulfoxide at 35℃; Irradiation; | 71 % Spectr. |
-
-
917-95-3
t-butylnitrite

-
-
73876-87-6
1-(trimethylsilyl)-2-methyl-1-propenyl triflate

-
A
-
933-52-8
2,2,4,4-tetramethyl-1,3-cyclobutanedione

-
B
-
503-17-3
dimethylacetylene

-
C
-
594-70-7
2-Methyl-2-nitropropane

-
D
-
6554-73-0
N-tert-butylmethacrylamide

-
E
-
7472-49-3
N-tert-butylisobutyramide

-
F
-
13947-81-4
N-tert-butyl-2-hydroxy-2-methylpropanamide

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In 1,2-dimethoxyethane at -20℃; for 0.0333333h; Mechanism; | A 8.3% B 70% C n/a D 6.4% E 8.3% F 5.3% |

-
-
13686-33-4
t-butyl azide

-
-
594-70-7
2-Methyl-2-nitropropane

| Conditions | Yield |
|---|---|
| With HOF* CH3CN In chloroform at -78℃; | 70% |

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; air; Nitrogen dioxide In various solvent(s) at 70℃; under 1520.1 Torr; for 14h; | 61% |
| With N-hydroxyphthalimide; air; Nitrogen dioxide In various solvent(s) at 70℃; under 760 Torr; for 14h; | 46% |
| With nitric acid at 150℃; |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Solvent; | A 0.5% B 60% |

-
-
24163-39-1
sodium 2-nitropropane

-
-
2355-56-8
1N-methyl-2,4,6-triphenylpyridinium tetrafluoroborate

-
A
-
594-70-7
2-Methyl-2-nitropropane

-
B
-
580-35-8
2,4,6-triphenylpyridine

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 80℃; | A 57% B n/a |
| In dimethyl sulfoxide at 25℃; for 2h; Rate constant; Kinetics; |

-
-
16649-50-6
N-tert-Butylhydroxylamine

-
-
594-70-7
2-Methyl-2-nitropropane

| Conditions | Yield |
|---|---|
| With potassium permanganate In water at 45 - 55℃; | 52% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Solvent; | A n/a B 0.65% |


| Conditions | Yield |
|---|---|
| With nitric acid |

| Conditions | Yield |
|---|---|
| With silver(I) nitrite; diethyl ether |

| Conditions | Yield |
|---|---|
| With silver(I) nitrite unter Kuehlung; |
-
-
7119-88-2
1,1-dibromo-1-nitro-ethane

-
-
544-97-8
dimethyl zinc(II)

-
A
-
79-24-3
Nitroethane

-
B
-
79-46-9
2-nitropropane

-
C
-
594-70-7
2-Methyl-2-nitropropane

-
-
113548-13-3
N-(benzenesulfonyl)-3-phenyloxaziridine

-
-
75-64-9
tert-butylamine

-
A
-
917-95-3
t-butylnitrite

-
B
-
594-70-7
2-Methyl-2-nitropropane

-
C
-
6852-58-0
N-benzylidene tert-butylamine

-
D
-
3376-24-7
N-tert-butyl-α-phenylnitrone

| Conditions | Yield |
|---|---|
| In chloroform-d1 for 1h; Product distribution; other primary amines; | A 30 % Spectr. B n/a C 65 % Spectr. D n/a |

-
-
75-64-9
tert-butylamine

-
A
-
31107-20-7, 117894-51-6
tert-butylnitroso dimer

-
B
-
917-95-3
t-butylnitrite

-
C
-
594-70-7
2-Methyl-2-nitropropane

| Conditions | Yield |
|---|---|
| With oxaziridinium tetrafluoroborate derived from N-methyl-1,2,3,4-tetrahydroisoquinoline In dichloromethane at 0℃; for 48h; | A 50 % Spectr. B 30 % Spectr. C 20 % Spectr. |
| With oxaziridinium tetrafluoroborate derived from N-methyl-1,2,3,4-tetrahydroisoquinoline In dichloromethane for 2h; | A 5 % Spectr. B 65 % Spectr. C 25 % Spectr. |



| Conditions | Yield |
|---|---|
| at 150℃; |

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
60-29-7
diethyl ether

-
-
507-20-0
tertiary butyl chloride

-
A
-
540-80-7
tert.-butylnitrite

-
B
-
594-70-7
2-Methyl-2-nitropropane

-
-
463-82-1
2,2-dimethylpropane

-
-
7697-37-2
nitric acid

-
A
-
75-52-5
nitromethane

-
B
-
594-70-7
2-Methyl-2-nitropropane

-
C
-
34715-98-5
2,2-dimethyl-1-nitro-propane

| Conditions | Yield |
|---|---|
| at 410℃; |

-
-
75-28-5
Isobutane

-
-
7697-37-2
nitric acid

-
A
-
79-46-9
2-nitropropane

-
B
-
625-74-1
1-nitroisobutane

-
C
-
594-70-7
2-Methyl-2-nitropropane

-
D
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| at 420℃; Produkt 5:Nitromethan; |


| Conditions | Yield |
|---|---|
| In tetralin at 154℃; Mechanism; Kinetics; Ea, log A, other solvent, other temperatures; |
-
-
926-42-1
2,2-dimethylpropyl nitrate

-
A
-
50-00-0
formaldehyd

-
B
-
540-80-7
tert.-butylnitrite

-
C
-
594-82-1
tetramethyl-2,2,3,3 butane

-
D
-
21823-36-9
2-methyl-2-butanol nitrate

-
E
-
594-70-7
2-Methyl-2-nitropropane

-
F
-
630-19-3
pivalaldehyde

| Conditions | Yield |
|---|---|
| In cyclohexane at 175℃; Mechanism; gas phase; |

-
-
594-70-7
2-Methyl-2-nitropropane

-
-
16649-50-6
N-tert-Butylhydroxylamine

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 10 - 20℃; for 3h; | 100% |
| With ammonia borane; gold on titanium oxide In ethanol at 20℃; for 0.666667h; Inert atmosphere; | 85% |
| Reduction; | 82% |
-
-
15133-82-1
tetrakis(triphenylphosphine)nickel(0)

-
-
594-70-7
2-Methyl-2-nitropropane

-
-
71928-64-8
C4H9NONi(P(C6H5)3)2

| Conditions | Yield |
|---|---|
| In benzene Kinetics; byproducts: P(C6H5)3, OP(C6H5)3; the soln. of Ni(PPh3)4/benzene was added to the soln. of t-BuNO2/benzene with stirring at under Ar, stirred for 1 h; evapd. in vac., washed with hot hexane, dried in vac.; | 100% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
51320-65-1
tetrakis(triethylphosphine)nickel(0)

-
-
71928-82-0
C4H9NONi(P(C2H5)3)2

| Conditions | Yield |
|---|---|
| In pentane Kinetics; byproducts: P(C2H5)3, OP(C2H5)3; to the soln. of Ni(PEt3)4/butane the soln. of t-BuNO2/pentane was addedwith stirring at -6°C under Ar, stirred for 1 h, warmed to 25°C; butane was added, cooled to -20°C, the liquid was remuved, partially evapd., this procedure repeated five times, crystd. at -78°C; | 100% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
10017-37-5
tert-butylamine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methyl-2-nitropropane With iron(III)-acetylacetonate; hydrazine hydrate In methanol at 150℃; for 0.0333333h; Microwave irradiation; Stage #2: With hydrogenchloride In water; ethyl acetate chemoselective reaction; | 99% |
| Stage #1: 2-Methyl-2-nitropropane With C36H56Cl3CrN2O; magnesium; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In tetrahydrofuran at 60℃; for 24h; Inert atmosphere; Schlenk technique; Stage #2: With hydrogenchloride In tetrahydrofuran; water; ethyl acetate at 20℃; | 99% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
1435482-51-1
(1R,3aR,4S,7aR)-octahydro-1-[(1’R,2’E)-4’-acetoxy-1’-methyl-but-2’-en-1’-yl]-7a-methyl-4-triethylsilyloxy-1H-indene

-
-
1435482-53-3
(1R,3aR,4S,7aR)-octahydro-1-[(1’R,2’E)-1’,5’-dimethyl-5’-nitro-hex-2’-en-1’-yl]-7a-methyl-4-triethylsilyloxy-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In dimethyl sulfoxide at 20℃; for 16h; | 99% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
135-02-4
ortho-anisaldehyde

-
-
130995-65-2
C-(2-methoxyphenyl)-N-tert-butylnitrone

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 97% |

| Conditions | Yield |
|---|---|
| With [Zn(BH4)2(py)] In tetrahydrofuran for 0.67h; Heating; | 96% |
| Stage #1: 2-Methyl-2-nitropropane In water; acetonitrile at 20℃; for 0.0833333h; Stage #2: With sodium tetrahydroborate In water; acetonitrile at 20℃; for 0.5h; | 90% |
| With palladium on activated charcoal; tetrabutylammomium bromide; water; sodium hydroxide; silicon at 100℃; for 6h; | 86% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 20℃; for 66h; | 96% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 20℃; for 20h; | 95% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
454-89-7
3-Trifluoromethylbenzaldehyde

-
-
139607-33-3
C-<3-(α,α,α-trifluoromethyl)phenyl>-N-tert-butylnitrone

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 93% |
| With acetic acid; zinc In ethanol at 0 - 20℃; for 16h; | 39% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
135-02-4
ortho-anisaldehyde

-
-
130995-65-2
N-tert-butyl-α-(2-methoxy)phenylnitrone

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 0 - 60℃; for 22.5h; Inert atmosphere; Darkness; Molecular sieve; | 92% |
| With zinc diacetate; acetic acid In ethanol | 877.2 mg(93%) |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel(II) chloride hexahydrate In water at 20℃; for 3h; Inert atmosphere; Green chemistry; | 92% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
34036-07-2
3,4 difluorobenzaldehyde

-
-
204707-23-3
3,4-difluorophenyl t-butyl nitrone

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol | 91% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
2646-90-4
2,5-difluorobenzaldehyde

-
-
204707-24-4
N-tert-butyl-α-(2,5-difluorophenyl)nitrone

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methyl-2-nitropropane With acetic acid; zinc In ethanol at 20℃; for 3h; Cooling with ice; Stage #2: C16H15NO2 In ethanol at 20℃; for 10h; | 90% |
| Stage #1: 2-Methyl-2-nitropropane With acetic acid; zinc In ethanol at 20℃; for 3h; Cooling with ice; Stage #2: C16H15NO2 In ethanol at 20℃; for 10h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methyl-2-nitropropane With acetic acid; zinc In ethanol at 20℃; for 3h; Cooling with ice; Stage #2: C12H13NO2 In ethanol at 20℃; for 16h; | 89% |
| Stage #1: 2-Methyl-2-nitropropane With acetic acid; zinc In ethanol at 20℃; for 3h; Cooling with ice; Stage #2: C12H13NO2 In ethanol at 20℃; for 16h; | 89% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 20℃; for 29h; | 88% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
55830-07-4
N-ethylidene-1,1-dimethylethanamine N-oxide

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium(II)chloride hexahydrate In acetonitrile at 50℃; for 9h; Irradiation; | 88% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
123-72-8
butyraldehyde

-
-
72552-75-1
N-butylidene-1,1-dimethylethanamine N-Oxide

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium(II)chloride hexahydrate; N,N-bis(1-methylethyl)-2-methyl-1-propanamine In acetonitrile at 50℃; for 9h; Irradiation; | 88% |
-
-
455-19-6
4-Trifluoromethylbenzaldehyde

-
-
594-70-7
2-Methyl-2-nitropropane

-
-
122778-03-4, 137957-46-1
C-<4-(α,α,α-trifluoromethyl)phenyl>-N-tert-butylnitrone

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 86% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
80670-36-6
3-diazo-5-phenyl-3H-1,2,4-triazole

-
A
-
917-95-3
t-butylnitrite

-
B
-
100-47-0
benzonitrile

| Conditions | Yield |
|---|---|
| at 80℃; Yields of byproduct given; | A 86% B n/a |

-
-
594-70-7
2-Methyl-2-nitropropane

-
-
100-52-7
benzaldehyde

-
-
3376-24-7, 52392-70-8, 85225-53-2
(Z)-N-benzylidene-2-methylpropan-2-amine oxide

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 20℃; for 21h; | 85% |
| With tin(II) chloride dihdyrate; sodium carbonate In tetrahydrofuran for 32h; Reflux; | 82% |
| With acetic acid; zinc In ethanol 1.) 10 deg C, 2 h, 2.) 6 deg C, 48 h; | 78% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 20℃; for 19h; | 85% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
44976-81-0
N,N-bis(1-methylethyl)-2-methyl-1-propanamine

-
-
85664-55-7
(Z)-2-methyl-N-(2-methylpropylidene)propan-2-amine N-oxide

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium(II)chloride hexahydrate In acetonitrile at 50℃; for 9h; Irradiation; | 84% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
139607-30-0
(Z)-N-3-fluorobenzylidene-t-butanamine oxide

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 83% |
| With acetic acid; zinc In ethanol at 0 - 20℃; |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
591-31-1
3-methoxy-benzaldehyde

-
-
115995-22-7
N-tert-butyl-α-(3-methoxy)phenylnitrone

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol at 0 - 60℃; for 24h; Inert atmosphere; Darkness; Molecular sieve; | 83% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
66-99-9
β-naphthaldehyde

-
-
120350-90-5, 1251855-74-9, 1590374-51-8
(Z)‑N‑(tert‑butyl)‑1‑(naphthalen‑2‑yl)methanimine oxide

| Conditions | Yield |
|---|---|
| With tin(II) chloride dihdyrate; sodium carbonate In tetrahydrofuran for 32h; Reflux; | 81% |
| With acetic acid; zinc In ethanol at 0 - 20℃; |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
104-88-1
4-chlorobenzaldehyde

-
-
40117-30-4, 137957-45-0
(Z)‑N‑(tert‑butyl)‑1‑(4‑chlorophenyl)methanimine oxide

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 80% |
| With tin(II) chloride dihdyrate; sodium carbonate In tetrahydrofuran for 32h; Reflux; | 80% |
-
-
594-70-7
2-Methyl-2-nitropropane

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
76790-28-8, 127896-05-3
N-<(4-Bromophenyl)methylene>-2-methyl-2-propanamine N-oxide

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In ethanol for 2h; Ambient temperature; | 80% |
2-Methyl-2-nitropropane Chemical Properties
IUPAC Name: 2-Methyl-2-nitro-propane
Synonyms: 1, 1-Dimethyl-1-nitroethane ; 1,1-Dimethyl-1-nitroethane ; 2-Methyl-2-nitropropane ; 2-Nitro-2-methylpropane
Product Categories: Nitro Compounds;Nitrogen Compounds;Organic Building Blocks Molecular Structure :
Molecular Formula of tert-Nitrobutane (CAS NO.594-70-7) : C4H9NO2
Molecular Weight of tert-Nitrobutane (CAS NO.594-70-7) : 103.12
Molecular Structure of tert-Nitrobutane (CAS NO.594-70-7) :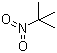
CAS NO: 594-70-7
EINECS: 209-851-4
Index of Refraction: 1.409
Surface Tension: 27 dyne/cm
Density: 0.96 g/cm3
Flash Point: 19.4 °C
Enthalpy of Vaporization: 36.37 kJ/mol
Boiling Point: 125.9 °C at 760 mmHg
Vapour Pressure: 11.9 mmHg at 25°C
Melting point 24 ºC
Storage temp: Flammables area
Water solubility: insoluble
Appearance:clear colourless liquid after melting
2-Methyl-2-nitropropane Safety Profile
Potentially explosive when heated. When heated to decomposition it emits toxic fumes of NOx. See also NITRO COMPOUNDS.
Hazard Codes  F,
F, Xi
Xi
Risk Statements 11-36/37/38
R11:Highly flammable.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements 16-26-36/37/39
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
RIDADR UN 1993 3/PG 2
WGK Germany 3
HazardClass 4.1
PackingGroup II
Related Products
- 2-Methyl-2-nitropropane
- 59-47-2
- 594-72-9
- 59473-45-9
- 5947-49-9
- 59481-23-1
- 594813-32-8
- 594-82-1
- 594823-69-5
- 59-48-3
- 59483-54-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View