-
Name
3-Nitrophenylboronic acid
- EINECS
- CAS No. 13331-27-6
- Article Data15
- CAS DataBase
- Density 1.4 g/cm3
- Solubility soluble in hot water
- Melting Point 289-291 °C
- Formula C6H6BNO4
- Boiling Point 363.3 °C at 760 mmHg
- Molecular Weight 166.929
- Flash Point 173.5 °C
- Transport Information
- Appearance off-white powder
- Safety 26-37/39-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzeneboronicacid, m-nitro- (6CI,7CI,8CI);Boronic acid, (3-nitrophenyl)- (9CI);3-Nitrobenzeneboronic acid;NSC 401539;NSC 59739;m-Nitrobenzeneboronic acid;m-Nitrophenylboronic acid;
- PSA 86.28000
- LogP -0.20220
Synthetic route
-
-
68716-48-3
4,4,5,5-tetramethyl-2-(3-nitrophenyl)-1,3,2-dioxaborolane

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; polystyrene boronic acid In acetonitrile at 20℃; for 18h; | 88% |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With water; silica gel at 20℃; for 24h; Inert atmosphere; | 86% |
| With water In tetrahydrofuran at 55℃; Kinetics; Reagent/catalyst; |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In dichloromethane; water at 50℃; for 24h; | 65% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-nitro-aniline With hydrogenchloride; sodium nitrite In water at 0℃; for 0.25h; Stage #2: tetrahydroxydiboron With sodium acetate In water at 20℃; for 0.333333h; | 64% |
-
-
98-80-6
phenylboronic acid

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| nitration according to W. Seaman and J. R. Johnson, J. Am. Chem. Soc., 1931, 53, 711; | 47% |
| With nitric acid at -15 - -9℃; | 28% |
-
-
13675-18-8
tetrahydroxydiboron

-
-
586-36-7
3-nitrophenyldiazonium tetrafluoroborate

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 0.25h; | 38% |
-
-
98-80-6
phenylboronic acid

-
A
-
13331-27-6
m-nitrobenzene boronic acid

-
B
-
5570-19-4
2-nitrophenylboronic acid

| Conditions | Yield |
|---|---|
| With nitric acid; urea | |
| With sulfuric acid; nitric acid |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With Trimethyl borate; hydrogen cation |
-
-
585-79-5
3-Bromonitrobenzene

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium; Triisopropyl borate In tetrahydrofuran; hexane; toluene at -70 - -20℃; | 13 % Chromat. |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| In phosphate buffer pH=6.9 - 7.0; Kinetics; |

| Conditions | Yield |
|---|---|
| With glass or Cs2CO3 In tetrahydrofuran; water Kinetics; hydrolysis at 55°C in THF/H2O in the presence of glass powder or Cs2CO3; not isolated; |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With water; acetic acid In tetrahydrofuran at 20℃; for 2h; |
-
-
99-09-2
3-nitro-aniline

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: water / 0.03 h / 20 °C 1.2: 0.25 h / 0 °C 2.1: N,N-dimethyl-formamide / 0.25 h / 20 °C View Scheme |
-
-
3262-89-3
triphenylboroxine

-
-
80283-99-4
2-nitrobenzo[d]isothiazol-3(2H)-one 1,1-dioxide

-
A
-
13331-27-6
m-nitrobenzene boronic acid

-
B
-
98-95-3
nitrobenzene

| Conditions | Yield |
|---|---|
| With 1,1,1,3',3',3'-hexafluoro-propanol; magnesium sulfate at 60℃; for 17h; Reagent/catalyst; | A 24 %Chromat. B 38 %Chromat. |

-
-
619-42-1
4-methoxycarbonylphenyl bromide

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
89900-93-6
3'-nitro-[1,1'-biphenyl]-4-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With sodium carbonate; bis(dibenzylideneacetone)-palladium(0); C78H99N6P(6+)*6Br(1-) In methanol; water at 65℃; for 3h; Suzuki-Miyaura cross-coupling; Inert atmosphere; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; toluene at 20 - 80℃; for 5h; Inert atmosphere; | 21% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,4-dioxane at 85℃; Suzuki cross-coupling; | 16% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
223797-51-1
1-tert-butoxycarbonyl-4-(5-trifluoromethanesulfonyloxy-3-pyridyl)homopiperazine

-
-
223797-57-7
1-[5-(3-nitrophenyl)-pyridin-3-yl]-4-tert-butoxycarbonylhomopiperazine

| Conditions | Yield |
|---|---|
| With potassium carbonate; lithium chloride; trimethyleneglycol; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane for 2h; Heating / reflux; | 100% |

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate; Pd(PPh3)4 In ethanol; toluene | 100% |
| With sodium carbonate; Pd(PPh3)4 In ethanol; toluene | 1.22% |
-
-
913067-90-0
N-(3-iodo-4-methyl-phenyl)-3-(trifluoromethyl)benzamide

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
1036376-84-7
N-(6-methyl-3'-nitrobiphenyl-3-yl)-3-(trifluoromethyl)benzamide

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water; toluene at 90℃; for 16h; Heating / reflux; | 100% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
554-84-7
meta-nitrophenol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water at 30℃; Green chemistry; | 99% |
| With 2,5-dimethylfuran; zinc(II) phthalocyanine; oxygen In tetrahydrofuran at 25℃; under 760.051 Torr; for 1.5h; Irradiation; Sealed tube; Schlenk technique; | 98% |
| With water; 3-chloro-benzenecarboperoxoic acid In ethanol at 20℃; for 6h; | 97% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
99-90-1
para-bromoacetophenone

-
-
5002-11-9
1-(3'-nitrobiphenyl-4-yl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol; water at 70℃; for 14h; Suzuki-Miyaura reaction; | 99% |
| With lithium niobate; palladium diacetate; potassium carbonate In ethanol; water; paraffin oil at 87 - 88℃; for 0.2h; Suzuki Coupling; | 99% |
| With tetrabutylammomium bromide; palladium diacetate; potassium carbonate In water at 70℃; for 3.33h; Suzuki Coupling; Inert atmosphere; | 96.5% |
-
-
610-94-6
2-bromobenzoic acid methyl ester

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
83527-96-2
methyl 3’-nitro-[1,1’-biphenyl]-2-carboxylate

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,4-dioxane at 85℃; Suzuki cross-coupling; | 99% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; toluene at 20 - 80℃; for 6h; Inert atmosphere; | 17% |
| With tetra-butylammonium acetate; Pd EnCat-30TM In ethanol Suzuki cross-coupling; microwave irradiation; |
-
-
591-50-4
iodobenzene

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine) palladium(0); tri-tert-butyl phosphine In 1,1,1,2,3,3,3-Heptafluoropropane; ethanol; 1,1,1,3,3-pentafluorobutane at 20℃; for 24h; Suzuki-Miyaura cross-coupling reaction; Inert atmosphere; | 99% |
| With C22H24Cl2N6O2Pd2; potassium carbonate In ethanol; water for 4h; Catalytic behavior; Suzuki-Miyaura Coupling; Reflux; | 99% |
| With potassium carbonate In water at 20℃; for 0.5h; Suzuki-Miyaura Coupling; | 97% |
-
-
942227-99-8
4-bromo-3,5-diphenylisothiazole

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 20 - 110℃; for 0.916667h; | 99% |
-
-
425379-37-9
2-bromo-3,5-difluorobenzonitrile

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
731817-91-7
4,6-difluoro-3'-nitrobiphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine In tetrahydrofuran; hexane; water at 50℃; for 4h; | 99% |
-
-
1262431-28-6
(E)-2-fluorovinyl 4-methylbenzenesulfonate

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
1262431-33-3
(E)-1-(2-fluorovinyl)-3-nitrobenzene

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; potassium phosphate; tricyclohexylphosphine In 1,4-dioxane at 100℃; for 14h; Suzuki-Miyaura reaction; Inert atmosphere; stereoselective reaction; | 99% |
-
-
7633-38-7
di-tert-butyl fumarate

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
1378314-51-2
(S)-di-tert-butyl-2-(3-nitrophenyl)succinate

| Conditions | Yield |
|---|---|
| Stage #1: m-nitrobenzene boronic acid With chlorobis(ethylene)rhodium(I) dimer; (S,S)-1,7,7-trimethyl-2,5-diphenylbicyclo[2.2.1]hepta-2,5-diene; potassium tert-butylate In methanol; dichloromethane at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: di-tert-butyl fumarate In methanol; dichloromethane; isopropyl alcohol at 60℃; for 1h; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); tri tert-butylphosphoniumtetrafluoroborate In water; toluene at 90℃; for 14h; Heck-Suzuki cascade reaction; optical yield given as %de; diastereoselective reaction; | 99% |
-
-
5159-41-1
2-iodobenzyl alcohol

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
893743-35-6
(3'-nitro-[1,1'-biphenyl]-2-yl)methanol

| Conditions | Yield |
|---|---|
| With C22H24Cl2N6O2Pd2; potassium carbonate In ethanol; water for 4h; Catalytic behavior; Suzuki-Miyaura Coupling; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; 1,10-Phenanthroline; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; 1,1,1,2,2,2-hexamethyldisilane; oxygen; copper diacetate; diisopropylamine at 20 - 150℃; Schlenk technique; | 99% |
-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium hydrogencarbonate In water; N,N-dimethyl-formamide at 110℃; for 4h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
56990-02-4
3,5-dibromobenzaldehyde

-
-
13331-27-6
m-nitrobenzene boronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 90℃; for 24h; Schlenk technique; Inert atmosphere; | 99% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 90℃; for 24h; Inert atmosphere; | 80% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
958-96-3
3,3'-dinitro-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With air In N,N-dimethyl-formamide at 20℃; for 16h; Green chemistry; | 98% |
| With potassium permanganate; potassium carbonate In methanol; water at 20℃; Catalytic behavior; Reagent/catalyst; | 97% |
| With [(μ4-Te)Fe3(CO)9Cu2(Me2Im)2]; oxygen In methanol at 25℃; for 24h; Kinetics; | 97% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
585-79-5
3-Bromonitrobenzene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; copper(I) bromide In acetonitrile at 80℃; for 1h; | 98% |
| With N-Bromosuccinimide In acetonitrile at 80℃; for 12h; | 98% |
| With tetrabutylammomium bromide; copper(ll) bromide In water at 100℃; Sealed tube; | 80% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
76757-90-9
N-(tert-butoxycarbonyl)-D-tyrosine methyl ester

| Conditions | Yield |
|---|---|
| With pyridine; copper diacetate; 4 A molecular sieve In dichloromethane at 25℃; for 18h; | 98% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
121-73-3
3-Chloronitrobenzene

| Conditions | Yield |
|---|---|
| With 1,3-dichloro-5,5-dimethylhydantoin; copper(l) chloride In acetonitrile at 80℃; for 2h; | 98% |
| With N-chloro-succinimide In acetonitrile at 80℃; for 12h; | 97% |
| With 1,3-dichloro-5,5-dimethylhydantoin; sodium methylate In methanol; water; acetonitrile at 60℃; | 43 % Chromat. |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
870-50-8
di-tert-butyl (E)-azodicarboxylate

| Conditions | Yield |
|---|---|
| With copper diacetate In tetrahydrofuran at 20℃; for 20h; | 98% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
4330-20-5
N-3-benzoylthymine

| Conditions | Yield |
|---|---|
| With pyridine; air; copper diacetate In dichloromethane at 20℃; for 75h; Chan-Lam-Evans-modified Ullmann condensation; | 98% |
-
-
696-62-8
para-iodoanisole

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
53059-31-7
4-methoxy-3'-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With potassium carbonate In water at 50℃; for 2h; Suzuki Coupling; | 98% |
| With magnesium hydroxide; potassium hydroxide; barium(II) hydroxide; calcium hydroxide In water at 20℃; for 2h; Suzuki Coupling; | 98% |
| With 0.1 % Cu/C; potassium carbonate In water at 50℃; for 3h; Suzuki Coupling; Green chemistry; | 95% |
-
-
395-44-8
o-trifluoromethylbenzyl bromide

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
886211-35-4
1-(3-nitrobenzyl)-2-(trifluoromethyl)benzene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane; water at 50℃; for 16h; | 98% |
-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
68716-48-3
4,4,5,5-tetramethyl-2-(3-nitrophenyl)-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With sodium sulfate In tetrahydrofuran at 20℃; for 24h; Schlenk technique; Inert atmosphere; | 98% |
| In toluene at 120℃; for 1h; | 92.1% |
| In benzene for 1h; Inert atmosphere; Reflux; | 75% |
| With magnesium sulfate In tetrahydrofuran at 20℃; for 16h; | 70% |
| In diethyl ether at 20℃; for 18h; | 60% |
-
-
1666-13-3
diphenyl diselenide

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
301329-93-1
(3-nitrophenyl)(phenyl)selane

| Conditions | Yield |
|---|---|
| With copper(II) oxide In dimethyl sulfoxide at 100℃; for 24h; Air atmosphere; | 98% |
| With potassium carbonate In water at 70℃; for 9h; | 88% |
| With iodine; dimethyl sulfoxide In neat (no solvent) at 100℃; for 0.166667h; Sealed tube; Microwave irradiation; Green chemistry; | 86% |
| With potassium carbonate at 80℃; for 10h; Green chemistry; | 85% |
| With sodium tetrahydroborate; 1,10-Phenanthroline; copper(ll) sulfate pentahydrate In ethanol at 20℃; for 8h; | 79% |
-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
99-02-5
3'-Chloroacetophenone

-
-
371157-19-6
1-(3'-nitrobiphenyl-3-yl)ethanone

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate monohydrate; 2-(2-methoxyphenyl)-1-methyl-3-(diphenylphosphino)-1H-indole In 1,4-dioxane at 90℃; for 24h; Suzuki-Miyaura coupling; Inert atmosphere; | 98% |
-
-
1226548-51-1
(2S)-2-[(5-bromo-6-phenylfuro[2,3-d]pyrimidin-4-yl)amino]-2-phenylethanol

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
1226549-62-7
(2S)-2-{[5-(3-nitrophenyl)-6-phenylfuro[2,3-d]pyrimidin-4-yl]amino}-2-phenylethanol

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In 1,4-dioxane; water at 100℃; for 2h; Inert atmosphere; | 98% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In 1,4-dioxane; water at 95℃; for 16h; Inert atmosphere; | 75% |
| With palladium bis[bis(diphenylphosphino)ferrocene] dichloride; sodium carbonate In 1,4-dioxane; water Suzuki Coupling; Inert atmosphere; Reflux; | 43% |
-
-
15249-31-7
N-phenyl-N-methyl-2-propynamide

-
-
13331-27-6
m-nitrobenzene boronic acid

-
-
1238625-89-2
N-methyl-3-(3-nitrophenyl)-N-phenylpropiolamide

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; silver(l) oxide In acetonitrile at 70℃; for 12h; Sonogashira coupling; | 98% |
3-Nitrophenylboronic acid Chemical Properties
IUPAC Name: (3-Nitrophenyl)boronic acid
Molecular Formula: C6H6BNO4
Molecular Weight: 166.94g/mol
Melting Point: 289-291 ºC
Freely Rotating Bonds: 4
Polar Surface Area: 64.28 Å2
Index of Refraction: 1.573
Molar Refractivity: 39.32 cm3
Molar Volume: 119.2 cm3
Polarizability: 15.58× 10-24cm3
Surface Tension: 57.8 dyne/cm
Density:1.4 g/cm3
Flash Point: 173.5 °C
Enthalpy of Vaporization: 64.29 kJ/mol
Boiling Point: 363.3 °C at 760 mmHg
Vapour Pressure: 6.5E-06 mmHg at 25°C
The Cas Register Number of 3-Nitrophenylboronic acid is 13331-27-6 .The chemical synonyms of 3-Nitrophenylboronic acid (CAS No.13331-27-6) are (3-Nitrophenyl)-boronicaci ; M-Nitro-benzeneboronicaci ; M-Nitrobenzeneboronicacid ; 3-Nitrophenylboric acid ; 3-Nitrophenylboronic acid ; 3-Nitrobenzeneboronic acid ; Akos brn-0128 ; M-Nitrophenylboronic acid .Product categories of 3-Nitrophenylboronic acid (CAS No.13331-27-6) are Blocks ; Boronicacids ; Boronic acid series ; Substituted boronic acids ; Organometallics ; Boronic acids ; Boronic acid ; Aryl ; Organoborons ; B (classes of boron compounds) .The molecular structure of 3-Nitrophenylboronic acid (CAS No.13331-27-6) is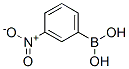 .
.
3-Nitrophenylboronic acid Toxicity Data With Reference
| 1. | mouse LD50:180mg/kg | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#01859, |
3-Nitrophenylboronic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 26-37/39-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39: Wear suitable gloves and eye/face protection.
S36: Wear suitable protective clothing.
WGK Germany: 3
RTECS: CY8980000
TSCA: T
HS Code: 29310095
Related Products
- 3-Nitrophenylboronic acid
- 3-Nitrophenylboronic acid pinacol ester
- 13331-31-2
- 13331-52-7
- 13331-93-6
- 1333-22-8
- 133-32-4
- 133331-77-8
- 13333-86-3
- 1333-39-7
- 13333-97-6
- 13334-00-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View