-
Name
3-Penten-2-one
- EINECS 210-888-3
- CAS No. 625-33-2
- Article Data95
- CAS DataBase
- Density 0.862 g/mL at 25 °C(lit.)
- Solubility slightly soluble in Water
- Melting Point
- Formula C5H8 O
- Boiling Point 121-124 °C(lit.)
- Molecular Weight 84.1179
- Flash Point 70 °c
- Transport Information UN 1992
- Appearance colourless liquid with a pungent odour
- Safety Moderately toxic by ingestion, inhalation, and skin contact. A skin and eye irritant. Flammable when exposed to heat or flame; can react with oxidizing materials. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also KETONES.
- Risk Codes 11-21-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F
F Xn
Xn
- Synonyms 2-Oxo-3-pentene;2-Penten-4-one; Ethylideneacetone; Methyl 1-propenyl ketone; Methyl propenylketone; NSC 61468
- PSA 17.07000
- LogP 1.15150
Synthetic route

| Conditions | Yield |
|---|---|
| With sulfur tetrafluoride; sodium fluoride at 20℃; for 3h; autoclave; | A 80% B 20% |
| With sulfur tetrafluoride; sodium fluoride at 20℃; for 24h; autoclave; | A 45% B 55% |
-
-
111957-98-3, 625-31-0
Pent-4-en-2-ol

-
A
-
13891-87-7
pent-4-en-2-one

-
B
-
625-33-2
3-penten-2-one

-
C
-
34218-21-8, 34218-22-9, 5729-47-5
2-pentene-1,4-dione

| Conditions | Yield |
|---|---|
| With tetrapropylammonium perruthennate In dichloromethane at 20℃; for 8h; Molecular sieve; | A 74% B 10% C 16% |


| Conditions | Yield |
|---|---|
| With potassium phosphate; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; Cu2(phenanthroline)2(μ-Cl)2Cl2; oxygen In acetonitrile at 20℃; for 5h; | 71% |
| With (1R,2R)-1,2-di(naphthalen-1-yl)ethane-1,2-diamine; tert.-butylhydroperoxide; potassium carbonate In water at 95℃; for 16h; | 21% |
| With oxygen; aluminum oxide; ruthenium In various solvent(s) at 82.84℃; for 6h; |
-
-
33177-29-6
5,6-dihydro-4-hydroxy-6-methyl-2H-pyran-2-one

-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| In water at 179.84℃; under 25858.1 Torr; Product distribution / selectivity; Inert atmosphere; | 59.7% |
| With Amberlyst 70 In water at 99.84℃; under 15751.6 Torr; for 4h; Solvent; Time; Inert atmosphere; Flow reactor; |

| Conditions | Yield |
|---|---|
| With dmap; acetic anhydride; triethylamine Ambient temperature; | A 28% B 53% |
| With dmap; triethylamine for 2h; Ambient temperature; | A 28% B 53% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In diethyl ether at 0℃; for 5h; | 9.6% |
| With sodium hydroxide; diethyl ether und Destillation des Reaktionsprodukts mit wasserfreier Oxalsaeure; | |
| With caesium carbonate In methanol at 20℃; for 5.5h; | |
| With sodium hydroxide In water at 40℃; | |
| With sodium hydroxide In water at 25℃; for 6h; |

| Conditions | Yield |
|---|---|
| at 525℃; Erhitzen; |
-
-
13891-87-7
pent-4-en-2-one

-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| durch verschiedene Agenzien; | |
| With sodium ethanolate | |
| With piperidine | |
| With mineral acid | |
| With hydrogenchloride In gas |

| Conditions | Yield |
|---|---|
| at 25 - 170℃; Quantum yield; Photolysis; |

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide; hexane | |
| With carbon disulfide; aluminum tri-bromide |

| Conditions | Yield |
|---|---|
| With iodine at 155℃; | |
| With acetic anhydride; zinc(II) chloride at 150 - 160℃; | |
| at 250℃; |
-
-
113704-77-1
4-bromo-pentan-2-one

-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate |

| Conditions | Yield |
|---|---|
| With sulfuric acid |


| Conditions | Yield |
|---|---|
| at 25℃; ohne Katalysator; | |
| at 25℃; analog verlaeuft die Einw. auf Propionaldehyd und Butyraldehyd; |

| Conditions | Yield |
|---|---|
| durch Saettigen des Gemisches mit Chlorwasserstoff und Destillieren des Reaktionsproduktes mit Diaethylanilin; |
-
-
17615-19-9
3,4-pentadien-2-ol

-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| With water; mercury(II) sulfate for 1.5h; Heating; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; zinc(II) chloride 1.) ethyl acetate, room temp., 18 h; 2.) 80 deg C, 2 h; Yield given. Multistep reaction; |
-
-
105-57-7
diethyl acetal

-
-
6651-39-4
trimethyl[(1-methylpropenyl)oxy]-silane

-
A
-
625-33-2
3-penten-2-one

-
B
-
33330-50-6
4-ethoxypentan-2-one

| Conditions | Yield |
|---|---|
| zinc(II) chloride In ethyl acetate Ambient temperature; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
623-43-8, 4358-59-2, 18707-60-3, 130981-57-6
methyl crotonate

-
-
107-31-3
Methyl formate

-
-
16812-43-4
bis(trimethylstannyl)methane

-
A
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction; |

-
-
1071-73-4
5-Hydroxy-2-pentanone

-
A
-
1487-15-6
2-Methyl-4,5-dihydrofuran

-
B
-
625-33-2
3-penten-2-one

-
C
-
765-43-5
Cyclopropyl methyl ketone

-
D
-
75-07-0
acetaldehyde

-
E
-
107-87-9
2-Pentanone

-
F
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| at 500℃; for 0.005h; Product distribution; other temperatures from 430 to 530 deg C, other reaction time, degree of conversion; |

-
-
5185-97-7
4-oxopentyl acetate

-
A
-
13891-87-7
pent-4-en-2-one

-
B
-
625-33-2
3-penten-2-one

-
C
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| at 380.4℃; Kinetics; Product distribution; Mechanism; other temperatures, various pressures, effect of propene inhibitors; Ea. log A; |

-
-
78080-39-4
4-(butylthio)pent-3-en-2-one

-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether 1.) room temperature, 2 h, 2.) 30 min, reflux; | 27.5 % Chromat. |
| With lithium aluminium tetrahydride In diethyl ether Product distribution; other reagent, solvent; | 27.5 % Chromat. |

| Conditions | Yield |
|---|---|
| With potassium hydroxide Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether 1.) room temperature, 2 h, 2.) 30 min, reflux; | A 62.5 % Chromat. B 6.5 % Chromat. |
| With lithium aluminium tetrahydride In diethyl ether Product distribution; other reagent, solvent; | A 62.5 % Chromat. B 6.5 % Chromat. |
-
-
74-85-1
ethene

-
-
67-64-1
acetone

-
A
-
13891-87-7
pent-4-en-2-one

-
B
-
625-33-2
3-penten-2-one

-
C
-
21889-88-3
hept-6-en-2-one

-
D
-
5185-97-7
4-oxopentyl acetate

-
E
-
107-87-9
2-Pentanone

| Conditions | Yield |
|---|---|
| With copper diacetate; manganese triacetate In acetic acid at 85℃; under 38000 Torr; for 4h; Product distribution; Mechanism; other solvents, var. conc. of reagents, var. pressure, var. reagent (MnO2); | A 30 % Turnov. B 18 % Turnov. C 28 % Turnov. D 14 % Turnov. E 10 % Turnov. |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; acetaldehyde Yield given; |

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane In acetone at 0 - 20℃; Product distribution; other solvent; |


-
A
-
625-86-5
2,5-dimethylfuran

-
B
-
620-02-0
5-Methylfurfural

-
C
-
625-33-2
3-penten-2-one

-
D
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| With air at 400 - 550℃; Oxidation; Formation of xenobiotics; |


| Conditions | Yield |
|---|---|
| With limonene.; palladium on activated charcoal for 0.25h; Heating; | 100% |
| With potassium bis(trimethylsilyl)amide In tetrahydrofuran at 0℃; for 6h; | 94.2% |
| With hydrogen; 1,5-hexadienerhodium(I)-chloride dimer In hexane for 3h; Ambient temperature; pH=7.6; | 71% |

| Conditions | Yield |
|---|---|
| With [Cu((S,S)-(C6H5CHCH2OCN)2C(CH3)2)](CF3SO3)2; 1,1,1,3',3',3'-hexafluoro-propanol In toluene at 20℃; for 5h; | 100% |
-
-
696-63-9
4-Methoxybenzenethiol

-
-
625-33-2
3-penten-2-one

-
-
1233849-11-0
4-(4-methoxyphenylthio) pentan-2-one

| Conditions | Yield |
|---|---|
| With 1-methyl-imidazolium p-toluenesulfonic acid at 20℃; for 4h; Michael addition; neat (no solvent); | 99% |
| With [Ni(2,6-bis[[(S)-2-(methyloxycarbonyl)-1-pyrrolidinyl]methyl]-pyridine)(CH3CN)](ClO4)2*H2O In acetonitrile at 20℃; for 2h; Michael Addition; Inert atmosphere; Schlenk technique; | 96% |
| With C39H47N7Ni2O5(3+)*3ClO4(1-) In acetonitrile at 20℃; for 2h; thia-Michael addition; | 41% |

| Conditions | Yield |
|---|---|
| With 9-epi-9-amino-9-deoxyquinine; N,N,N',N'-tetramethyl-1,8-diaminonaphthalene; benzoic acid at 40℃; for 144h; Michael Addition; enantioselective reaction; | 99% |
| With (S)-N1-cyclohexyl-3,3-dimethylbutane-1,2-diamine; benzoic acid In dichloromethane at 30℃; for 48h; Michael Addition; enantioselective reaction; | 80% |

| Conditions | Yield |
|---|---|
| With chlorobis(ethylene)rhodium(I) dimer; [(η5-1-bis(3,5-dimethylphenyl)phosphino-2-(3-diphenylphosphino-2-methylpropenyl)cyclopentadienyl-P)]manganese(I) dicarbonyl; potassium hydroxide In 1,4-dioxane; water at 50℃; for 9h; Inert atmosphere; enantioselective reaction; | 99% |
| With chlorobis(ethylene)rhodium(I) dimer; C41H24F12O2P2; potassium hydroxide In water; toluene at 20℃; for 4h; Inert atmosphere; Schlenk technique; enantioselective reaction; | 95% |
| With chlorobis(ethylene)rhodium(I) dimer; C41H24F12O2P2; potassium hydroxide In water; toluene at 20℃; for 0.5h; Inert atmosphere; Schlenk technique; | 95% |
| With C17H27NO In water; toluene at 100℃; for 12h; Inert atmosphere; | 91% |
| With heterogeneous Rh/Ag bimetallic nanoparticle catalyst immobilized on chiral polymer In water; toluene at 100℃; for 24h; Inert atmosphere; enantioselective reaction; | 90% |
-
-
625-33-2
3-penten-2-one

-
-
5720-05-8
4-methylphenylboronic acid

-
-
69657-27-8
(S)-4-(4-methylphenyl)-2-pentanone

| Conditions | Yield |
|---|---|
| With chlorobis(ethylene)rhodium(I) dimer; C41H24F12O2P2; potassium hydroxide In water; toluene at 20℃; for 4h; Inert atmosphere; Schlenk technique; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In diethyl ether Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With 1-methyl-imidazolium p-toluenesulfonic acid at 20℃; for 16h; Michael addition; neat (no solvent); | 98% |
-
-
625-33-2
3-penten-2-one

-
-
145865-96-9
4,9-dihydro-9-(p-toluenesulfonyl)-3H-pyrido-<3,4-b>indole

-
-
1421697-76-8
(4R,12bS)-4-methyl-12-tosyl-1,3,4,6,7,12b-hexahydroindolo[2,3-a]quinolizin-2(12H)-one

| Conditions | Yield |
|---|---|
| With (S)-2-(3-((1R,2R)-2-aminocyclohexyl)thioureido)-N-benzhydryl-N,3,3-trimethylbutanamide; acetic acid In toluene at 4℃; for 48h; Diels-Alder Cycloaddition; Inert atmosphere; enantioselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| With C38H54ClFeN2O2 In 1,2-dichloro-ethane at -5℃; for 32h; enantioselective reaction; | 97% |
-
-
625-33-2
3-penten-2-one

-
-
64420-86-6
O-p-toluenesulfonyl benzyloxyhydroxamate

-
-
1096017-08-1
2-acetyl-3-methylaziridine-1-carboxylic acid benzyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 3-penten-2-one With 1.5C13H17NO4*C20H27N3O In chloroform at 23℃; for 0.166667h; Stage #2: O-p-toluenesulfonyl benzyloxyhydroxamate With sodium hydrogencarbonate In chloroform at 23℃; for 16h; optical yield given as %ee; enantioselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With 4-n-butyl-4-(3-sulfopropyl)thiomorpholinium 1,1-dioxide trifluoromethane sulfonate at 70℃; for 0.5h; Michael Addition; Green chemistry; | 96% |

| Conditions | Yield |
|---|---|
| With Boc-D-Phg-OH; (R,R)-1,2-diphenylethylenediamine In toluene at 20℃; for 24h; Michael Addition; stereoselective reaction; | A n/a B 96% |


| Conditions | Yield |
|---|---|
| With hydrogen; In diethylene glycol dimethyl ether; water at 30℃; under 735.5 Torr; for 5h; Kinetics; reaction profiles reactions of hydrogenation of some unsatureted ketones; | A 95.3% B 4.7% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
625-33-2
3-penten-2-one

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
92803-27-5
4-methyl-5-(N-tert-butylimino)-2-nonanone

| Conditions | Yield |
|---|---|
| With copper(l) iodide; boron trifluoride diethyl etherate In diethyl ether at -78 - 0℃; for 0.5h; Product distribution; reactions of Cu(I) aldimines with α,β-unsaturated carbonyl compounds; | 95% |
| With copper(l) iodide; boron trifluoride diethyl etherate; sodium carbonate In diethyl ether -78 degC, 30 min, than 0 degC; | 95% |


| Conditions | Yield |
|---|---|
| With aluminium(III) triflate at 80℃; for 0.133333h; Time; Wavelength; Microwave irradiation; regioselective reaction; | 95% |
| With N-[(perfluorobutyl)sulfonyl]-3-{N-[(perfluorobutyl)sulfonyl]sulfamoyl}benzamide In water at 30℃; for 3h; Temperature; Michael Addition; | 91% |
| With ionic liquid immobilized on silica-SO2Cl In diethyl ether at 20℃; for 5h; | 90% |

| Conditions | Yield |
|---|---|
| With BF4(1-)*C68H44N2O4P2Rh(1+); water; potassium hydroxide In 1,4-dioxane at 60℃; for 1h; Anaerobic conditions; Combinatorial reaction / High throughput screening (HTS); optical yield given as %ee; enantioselective reaction; | 95% |
| With chlorobis(ethylene)rhodium(I) dimer; (RS)-N-(cinnamyl)-2-methylpropane-2-sulfinamide; potassium hydroxide In methanol; water at 40℃; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 78% |
| With chlorobis(ethylene)rhodium(I) dimer; [η5-1-bis(3,5-di(trifluormethylphenyl)phosphino-2-(3-diphenylphosphino-2-methylpropenyl)cyclopentadienyl-P)]manganese(I) dicarbonyl; potassium hydroxide In 1,4-dioxane; water at 50℃; for 9h; Reagent/catalyst; Inert atmosphere; enantioselective reaction; | 60% |
-
-
625-33-2
3-penten-2-one

-
-
18039-42-4
5-phenyl-2H-1,2,3,4-tetrazole

-
-
1353002-70-6
4-(5-phenyl-2H-tetrazol-2-yl) pentan-2-one

| Conditions | Yield |
|---|---|
| With 1-methyl-imidazolium p-toluenesulfonic acid In dichloromethane at 20℃; for 1h; Michael addition; | 95% |
-
-
625-33-2
3-penten-2-one

-
-
20361-10-8
2,3-dihydro-3-oxo-1H-isoindole-1-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; for 8h; Michael Addition; | 95% |

| Conditions | Yield |
|---|---|
| With C38H54ClFeN2O2 In 1,2-dichloro-ethane at -5℃; for 31h; enantioselective reaction; | 95% |
| Stage #1: 3-penten-2-one With nanoparticles of Iron(II)-folded copolymer from N-isopropylacrylamide and 4-benzyl-2-vinyloxazoline In water for 0.0833333h; Michael Addition; Stage #2: (4-chlorophenyl)methanethiol In water at 25℃; for 48h; Michael Addition; enantioselective reaction; | 94% |

| Conditions | Yield |
|---|---|
| With phosphomolybdic acid; sodium dodecyl-sulfate In water; toluene at 80℃; for 0.833333h; | 94% |

| Conditions | Yield |
|---|---|
| With sodium ligninsulfonate-immobilized Sc(OTf)3 In ethanol Michael Addition; | 94% |
-
-
625-33-2
3-penten-2-one

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 1h; Reagent/catalyst; Michael Addition; | 94% |
-
-
873-73-4
4-n-chlorophenylacetylene

-
-
625-33-2
3-penten-2-one

-
-
58458-85-8
trimethoxy(phenylethynyl)silane

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; methanol; zinc In N,N-dimethyl-formamide at 20℃; for 18h; Schlenk technique; Sealed tube; Inert atmosphere; stereoselective reaction; | 94% |

-
-
625-33-2
3-penten-2-one

-
-
39748-15-7, 33115-74-1
methyl 2-isocyanopropanoate

-
-
125294-54-4, 125294-55-5
2-Isocyano-2,3-dimethyl-5-oxo-hexanoic acid methyl ester

| Conditions | Yield |
|---|---|
| tetrabutyl ammonium fluoride In tetrahydrofuran for 3h; Ambient temperature; | 93% |
-
-
10075-50-0
5-bromo-1H-indole

-
-
625-33-2
3-penten-2-one

-
-
1018636-36-6
4-(5-bromo-1H-indol-3-yl)pentan-2-one

| Conditions | Yield |
|---|---|
| With N-[(perfluorobutyl)sulfonyl]-3-{N-[(perfluorobutyl)sulfonyl]sulfamoyl}benzamide In water at 30℃; for 5h; Michael Addition; | 93% |
| With zirconium(IV) chloride In dichloromethane at 20℃; | 72% |
-
-
625-33-2
3-penten-2-one

-
-
41795-35-1
(5-hexen-1-ylsulfonyl)-benzene

-
-
1235835-70-7
8-(phenylsulfonyl)-3-octen-2-one

| Conditions | Yield |
|---|---|
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In dichloromethane at 20℃; for 13h; | 93% |
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In dichloromethane at 20℃; for 13h; | 93% |

| Conditions | Yield |
|---|---|
| With cadmium tetrahydroborate In acetonitrile for 24h; Mechanism; Ambient temperature; | 92% |
| With cadmium tetrahydroborate In acetonitrile for 24h; Ambient temperature; | 92% |
| With sodium tetrahydroborate In tetrahydrofuran; methanol at 0℃; for 0.333333h; | 70% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
625-33-2
3-penten-2-one

-
-
1340332-19-5
4-(1H-benzo[d][1,2,3]triazol-1-yl)pentan-2-one

| Conditions | Yield |
|---|---|
| With 1-methyl-imidazolium p-toluenesulfonic acid In dichloromethane at 20℃; for 3h; Michael addition; | 92% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; acetic acid at 25℃; for 30h; Inert atmosphere; Darkness; | 92% |
3-Penten-2-one Chemical Properties
Molecular Structure of 3-Penten-2-one (625-33-2):
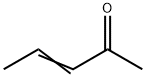
EINECS: 210-888-3
IUPAC Name: (E)-Pent-3-en-2-one
Molecular Formula: C5H8O
Molecular Weight: 84.116420 g/mol
XLogP3: 0.5
H-Bond Donor: 0
H-Bond Acceptor: 1
Canonical SMILES: CC=CC(=O)C
Isomeric SMILES: C/C=C/C(=O)C
InChI: InChI=1S/C5H8O/c1-3-4-5(2)6/h3-4H,1-2H3/b4-3+
InChIKey: LABTWGUMFABVFG-ONEGZZNKSA-N
Index of Refraction: 1.411
Molar Refractivity: 25.3 cm3
Molar Volume: 101.8 cm3
Surface Tension: 23.1 dyne/cm
Density: 0.826 g/cm3
Flash Point: 19.8 °C
Enthalpy of Vaporization: 36 kJ/mol
Boiling Point: 122 °C at 760 mmHg
Vapour Pressure: 14.2 mmHg at 25 °C
Water Solubility: 4.618e+004 mg/L at 25 °C
Refractive Index: n20/D 1.437(lit.)
Storage Temp.: 2-8 °C
BRN: 1633505
3-Penten-2-one Toxicity Data With Reference
| 1. | skn-rbt 10 mg/24H | JIHTAB Journal of Industrial Hygiene and Toxicology. 30 (1948),63. | ||
| 2. | eye-rbt 500 mg | JIHTAB Journal of Industrial Hygiene and Toxicology. 30 (1948),63. | ||
| 3. | orl-rat LD50:3200 mg/kg | JIDHAN Journal of Industrial Hygiene. 30 (1948),63. | ||
| 4. | ihl-rat LCLo:250 ppm/4H | JIHTAB Journal of Industrial Hygiene and Toxicology. 31 (1949),343. | ||
| 5. | skn-rbt LD50:500 mg/kg | JIHTAB Journal of Industrial Hygiene and Toxicology. 30 (1948),63. |
3-Penten-2-one Consensus Reports
Community Right-To-Know List. Reported in EPA TSCA Inventory.
3-Penten-2-one Safety Profile
Safety Information of 3-Penten-2-one (625-33-2):
Hazard Codes: F ![]() ,Xn
,Xn ![]()
Risk Statements: 11-21-36/37/38
11: Highly Flammable
21: Harmful in contact with skin
36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 16-26-33-36/37/39
16: Keep away from sources of ignition - No smoking
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
33: Take precautionary measures against static discharges
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
RIDADR: UN 1992 3/PG 2
WGK Germany: 3
RTECS: SB3850000
F: 10-19
HazardClass: 3
PackingGroup: II
Moderately toxic by ingestion, inhalation, and skin contact. A skin and eye irritant. Flammable when exposed to heat or flame; can react with oxidizing materials. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also KETONES.
3-Penten-2-one Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
3-Penten-2-one Specification
3-Penten-2-one (625-33-2) is colourless liquid with a pungent odour. It is also called for 2-Oxo-3-pentene ; 3-01-00-02985 (Beilstein Handbook Reference) ; AI3-37794 ; Ethylidene acetone ; FEMA No. 3417 ; Methyl 1-propenyl ketone ; Methyl propenyl ketone ; NSC 61468 .
Related Products
- 3-Penten-2-one
- 3-Penten-2-one,1,1,1-trifluoro-4-methyl-
- 3-Penten-2-one,4-(1-pyrrolidinyl)-
- 3-Penten-2-one,4-[(trimethylsilyl)oxy]-
- 3-Penten-2-one,4-[[(1,1-dimethylethyl)dimethylsilyl]oxy]-
- 62534-43-4
- 625-35-4
- 625-36-5
- 625-37-6
- 62538-21-0
- 625-38-7
- 625-40-1
- 62-54-4
- 625446-22-2
- 6254-48-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View