-
Name
4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride
- EINECS 249-220-0
- CAS No. 28783-41-7
- Article Data12
- CAS DataBase
- Density 1.143g/cm3
- Solubility
- Melting Point 212-215°C
- Formula C7H10ClNS
- Boiling Point 242.9 °C at 760 mmHg
- Molecular Weight 175.682
- Flash Point 100.7 °C
- Transport Information
- Appearance Off-White Solid
- Safety 26-36/37/39-37
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Thieno[3,2-c]pyridine,4,5,6,7-tetrahydro-, hydrochloride (8CI,9CI);
- PSA 40.27000
- LogP 2.52460
Synthetic route
-
-
30433-91-1
[2-(2-thyenyl)ethyl]amine

-
-
50-00-0
formaldehyd

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: [2-(2-thyenyl)ethyl]amine; formaldehyd In dichloromethane at 40 - 45℃; Stage #2: With hydrogenchloride In dichloromethane; water; N,N-dimethyl-formamide at 20 - 70℃; | 99% |
| Stage #1: [2-(2-thyenyl)ethyl]amine; formaldehyd In toluene for 1.5h; Reflux; Inert atmosphere; Dean-Stark; Stage #2: With hydrogenchloride In 1,4-dioxane; toluene at 60℃; for 0.5h; Inert atmosphere; | 98% |
| Stage #1: [2-(2-thyenyl)ethyl]amine; formaldehyd In 1,2-dichloro-ethane for 4h; Heating / reflux; Stage #2: With hydrogenchloride In 1,2-dichloro-ethane; N,N-dimethyl-formamide at 70℃; for 4h; | 90% |
-
-
68559-49-9
N-methoxycarbonyl-4,5,6,7-tetrahydrothieno[2,3-c]pyridine

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: N-methoxycarbonyl-4,5,6,7-tetrahydrothieno[2,3-c]pyridine With sodium hydroxide In methanol for 12h; Reflux; Stage #2: With hydrogenchloride In 1,4-dioxane pH=1; | 82% |
-
-
111954-31-5
N-methylene-2-(thien-2-yl)ethanamine

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In N,N-dimethyl-formamide | |
| With hydrogenchloride In isopropyl alcohol at 60℃; for 5h; |
-
-
107112-93-6
dihydro-6,7 thieno<3,2-c>pyridine

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: dihydro-6,7 thieno<3,2-c>pyridine With methanol; sodium tetrahydroborate at 20℃; for 2h; Stage #2: With hydrogenchloride |
-
-
5402-55-1
2-thiophenethanol

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: triethylamine / dichloromethane / 2 h / -5 - 20 °C 2: formic acid / 8.5 h / 80 °C / Inert atmosphere 3: trifluoroacetic acid / 20 °C 4: sodium tetrahydroborate; methanol / 2 h / 20 °C View Scheme |
-
-
40412-06-4
2-(2-thienyl)ethyl tosylate

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: formic acid / 8.5 h / 80 °C / Inert atmosphere 2: trifluoroacetic acid / 20 °C 3: sodium tetrahydroborate; methanol / 2 h / 20 °C View Scheme |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
230301-73-2
6,7-dihydro-4H-thieno[3,2-c]-pyridine-5-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; | 100% |
| With triethylamine In dichloromethane at 20℃; for 4.5h; | 100% |
| With triethylamine In dichloromethane at 20℃; for 6h; | 92.3% |
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With potassium carbonate In 1,4-dioxane at 80℃; for 0.5h; Stage #2: di-tert-butyl dicarbonate In 1,4-dioxane for 1h; | 16.8 g |
-
-
76-83-5
trityl chloride

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
109904-25-8
5-trityl-4,5,6,7-tetrahydro-tieno[3,2-c]pyridine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | 100% |
| With triethylamine In dichloromethane at 12 - 28℃; | 99% |
| With N-ethyl-N,N-diisopropylamine In acetonitrile for 3h; | 94% |

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
113665-84-2
(S)-(+)-clopidogrel

| Conditions | Yield |
|---|---|
| With sodium carbonate In dichloromethane at 25 - 40℃; for 24h; Temperature; | 97.02% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
100-44-7
benzyl chloride

-
-
55142-78-4
5-benzyl-4,5,6,7-tetrahydro-thieno[3,2-c]-pyridine

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate; potassium carbonate In acetonitrile at 70℃; | 97% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
54903-50-3
4,5,6,7-tetrahydrothieno[3,2-c]pyridine

| Conditions | Yield |
|---|---|
| With water; potassium hydroxide In toluene at 25 - 30℃; | 96.5% |
| With sodium hydroxide In dichloromethane at 20℃; for 3.5h; | 3.12 g |
-
-
223123-46-4
methyl (R)-2-(2-chlorophenyl)-2-benzenesulfonyloxyacetate

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
120202-66-6
(S)-(+)-clopidogrel bisulfate

| Conditions | Yield |
|---|---|
| Stage #1: methyl (R)-2-(2-chlorophenyl)-2-benzenesulfonyloxyacetate; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With sodium hydrogencarbonate In acetone for 6h; Reflux; Stage #2: With sulfuric acid In acetone | 95.2% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
29079-79-6
4,5,6,7-tetrahydrothieno[3,2-c]pyridine-5-carbaldehyde

| Conditions | Yield |
|---|---|
| With molybdenum (IV) sulfide at 150℃; for 18h; | 95% |
| at 150℃; for 96h; Inert atmosphere; Sealed tube; | 89% |
| With graphene oxide at 150℃; for 18h; Sealed tube; | 81% |

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
120202-66-6
(S)-(+)-clopidogrel bisulfate

| Conditions | Yield |
|---|---|
| Stage #1: (R)-2-(2-chlorophenyl)-2-(4-nitrobenzenesulfonyloxy)acetic acid methyl ester; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With potassium carbonate In acetone at 20℃; for 24h; Stage #2: With sulfuric acid In acetone | 93.1% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| With manganese(II) chloride tetrahydrate at 150℃; for 10h; Inert atmosphere; Sealed tube; | 93% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
135270-13-2
1-(((2R,3R)-2-(2,4-difluorophenyl)-3-methyloxiran-2-yl)-methyl)-1H-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With magnesium 2-methylpropan-2-olate In acetonitrile for 16h; Inert atmosphere; Reflux; | 93% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
4023-34-1
cyclopropanecarboxylic acid chloride

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0℃; for 1h; | 93% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
85259-19-4
methyl 2-bromo-2-(2-chlorophenyl)acetate

-
-
135046-48-9
clopidogrel bisulfate

| Conditions | Yield |
|---|---|
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With sodium carbonate In water at 25℃; for 0.5h; Stage #2: methyl 2-bromo-2-(2-chlorophenyl)acetate In water; toluene at 20℃; for 12h; Stage #3: With sulfuric acid In ethyl acetate for 1h; Reagent/catalyst; Solvent; | 90% |
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride; methyl 2-bromo-2-(2-chlorophenyl)acetate With potassium carbonate In acetone Stage #2: With sulfuric acid |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
949922-62-7
2-bromo-6,7-dihydrothieno[3,2-c]pyridine-5(4H)-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: di-tert-butyl dicarbonate; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With tert-butyl methyl ether; sodium hydroxide In water at 0℃; Stage #2: With bromine In water at 0 - 20℃; for 1.5h; | 88.32% |
-
-
29270-30-2
α-bromo-2-chlorophenyl acetic acid

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
90055-55-3
2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridine-5(4H)-yl)acetic acid

| Conditions | Yield |
|---|---|
| Stage #1: α-bromo-2-chlorophenyl acetic acid; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With potassium hydroxide In methanol; water at 10 - 40℃; for 3h; Stage #2: With hydrogenchloride In methanol; water at 5 - 20℃; for 4h; pH=3.5; | 88% |
| Stage #1: α-bromo-2-chlorophenyl acetic acid; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With potassium hydroxide In methanol; water at 15 - 40℃; for 3h; Stage #2: With hydrogenchloride In methanol; water at 20℃; for 2h; pH=3.5; | 88% |
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With sodium hydroxide In water at 20 - 35℃; Stage #2: α-bromo-2-chlorophenyl acetic acid In water at 20 - 27℃; Stage #3: With hydrogenchloride In water; ethyl acetate at 20 - 30℃; pH=3.5 - 4.5; | |
| With potassium hydroxide In water at 0 - 20℃; for 24h; | 12 g |
-
-
143-33-9
sodium cyanide

-
-
89-98-5
2-chloro-benzaldehyde

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
444728-11-4
2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridine-5(4H)-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Stage #1: sodium cyanide; 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride In methanol; water for 0.666667h; Stage #2: 2-chloro-benzaldehyde In methanol; water at 23 - 50℃; for 6h; Stage #3: With sodium metabisulfite In methanol; water at 25 - 30℃; for 2h; | 87.4% |

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
5829-89-0
(+/-)α-chloro-2-(2-chlorophenyl)acetonitrile

-
-
444728-11-4
2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridine-5(4H)-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In butan-1-ol for 3h; Product distribution / selectivity; Heating / reflux; | 86% |
-
-
444891-19-4
2-bromo-2-(2-chlorophenyl)acetonitrile

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
444728-11-4
2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridine-5(4H)-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In methanol for 3h; Product distribution / selectivity; Heating / reflux; | 85% |
| With sodium hydrogencarbonate In methanol for 3h; Heating / reflux; | 85% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
121-60-8
p-acetylaminobenzenesulfonyl chloride

-
-
949779-15-1
N-(4-{6,7-dihydrothieno[3,2-c]pyridin-5(4H)-ylsulfonyl}phenyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 5h; Reflux; | 84.5% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
272-14-0
thieno[3,2-c]pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With water; sodium hydroxide In dichloromethane at 20℃; Stage #2: With manganese(IV) oxide In toluene at 140℃; for 24h; | 84.42% |
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With sodium hydroxide In water for 2h; Stage #2: With manganese(IV) oxide In toluene at 130℃; | 650 mg |
-
-
1121-57-9
1-isocyanocyclohexene

-
-
89-98-5
2-chloro-benzaldehyde

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
1130832-62-0
2-(2-chlorophenyl)-N-cyclohex-2-enyl-2-(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-yl)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With triethylamine In methanol at 20℃; for 0.0833333h; Stage #2: 2-chloro-benzaldehyde In methanol at 20℃; for 1h; Ugi reaction; Stage #3: 1-isocyanocyclohexene With formic acid In methanol at 60℃; for 1h; Ugi reaction; Microwave irradiation; | 83% |

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
4392-24-9
Cinnamyl bromide

-
-
1581720-50-4
5-cinnamyl-4,5,6,7-tetrahydrothieno[3,2-c]pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With sodium hydroxide In water at 0 - 25℃; for 0.166667h; Inert atmosphere; Stage #2: Cinnamyl bromide With triethylamine In tetrahydrofuran; water for 0.333333h; Inert atmosphere; | 76% |
-
-
50-00-0
formaldehyd

-
-
568-72-9
tanshinone IIA

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

| Conditions | Yield |
|---|---|
| With copper diacetate; acetic acid In dimethyl sulfoxide at 60℃; | 76% |


-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
1093351-48-4
(S)-2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridine-5(4H)-yl)acetic acid methyl ester-d3

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In acetonitrile at 20℃; for 24h; Inert atmosphere; | 73.6% |
-
-
1089210-23-0
5-bromo-2,3-dihydro-2,2-dimethyl-7-(4-bromo-butoxy)-benzofuran

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
1089209-66-4
1-[(5-bromo-2,3-dihydro-2,2-dimethyl-benzofuran-7-yl)-oxy]-4-(4,5,6,7-tetrahydro-thieno[3,2-c]pyridin-5-yl)-butane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; triethylbutylammonium chloride In chloroform; water at 20 - 60℃; for 28h; | 73.3% |
-
-
204205-33-4
2-bromo-1-cyclopropyl-2-(2-fluorophenyl)ethanone

-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
1287752-37-7
1-cyclopropyl-2-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)-2-(2-fluorophenyl)ethanone hydrobromide salt

| Conditions | Yield |
|---|---|
| Stage #1: 6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride With potassium hydrogencarbonate at 20℃; for 0.5h; Stage #2: 2-bromo-1-cyclopropyl-2-(2-fluorophenyl)ethanone In acetonitrile at 20℃; for 25h; Stage #3: With hydrogen bromide In water; acetone at 10 - 20℃; for 3h; Reagent/catalyst; | 73.1% |
| Multi-step reaction with 2 steps 1: potassium carbonate / acetonitrile / 5 h / 25 - 35 °C 2: hydrogen bromide / water; acetone / 5.25 h / 0 - 30 °C View Scheme |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
938904-49-5
4'-(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-ylmethyl)biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol | 72% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
106-96-7
propargyl bromide

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 16h; | 72% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 16h; | 72% |
-
-
28783-41-7
6,7-dihydro-4H-thieno[3,2-c]pyridine hydrochloride

-
-
89151-46-2
tert-butyl 4-(2-iodoethyl)piperidine-1-carboxylate

-
-
158149-68-9
5-<2-(N-Boc-4-piperidinyl)ethyl>-4,5,6,7-tetrahydrothieno<3,2-c>pyridine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Aliquat 336 In dichloromethane for 20h; | 70% |
| With sodium hydroxide In dichloromethane |
4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride Chemical Properties
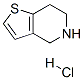
IUPAC Name:4,5,6,7-tetrahydrothieno[3,2-c]pyridin-1-ium chloride
Molecular Formula:C7H10ClNS
Molecular Weight:175.679000 g/mol
Melting Point:212-215°C
CAS DataBase Reference:28783-41-7(CAS DataBase Reference)
Synonyms of 4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride(28783-41-7):
4,5,6,7-TETRAHYDROTHIENO[3,2,C] PYRIDINE HCL;4,5,6,7-TETRAHYDROTHIENO[3,2-C]PYRIDINE HYDROCHLORIDE;4,5,6,7-tetrahydrotieno[3,2-c]pyridinium chloride;4,5,6,7-TETRAHYDROTHIENO[3,2-C]PYRIDINE HCl(TTP);4,5,6,7-tetrahydrothieno[3,2-c]pyridinium chloride;Tetrahydrothieno pyridine HCl;4,5,6,7-Tetrahydrothieon[3,2-C]pyridine,HCl;4,5,6,7-Tetrahydrothieon[3,2-C]pyridine,hydrochloride
Categories of 4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride(28783-41-7):
Heterocyclic Compounds;Bases & Related Reagents;Heterocycles;Nucleotides;Sulfur & Selenium Compounds
4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride Uses
4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride Safety Profile
Safety Information of 4,5,6,7-Tetrahydrothieno[3,2,c] pyridine hydrochloride(28783-41-7):
Risk Statements:36/37/38
36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements:26-36/37/39
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39:Wear suitable protective clothing, gloves and eye/face protection
Related Products
- 4-06-00-02342 (Beilstein Handbook Reference)
- 4,10-Ace-1,2-benzanthracene
- 4,10-Dioxatricyclo[5.2.1.0(2,6)]decan-8-en-3-one
- 4-(1,1,2,2-Tetrafluoroethoxy)benzoicacid
- 4-(1,1,2,2-Tetrafluoroethoxy)chlorobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)nitrobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)toluene
- 4-(1,1-Difluoropropan-2-yl)benzene-1-sulfonyl chloride
- 4-(1,1-Dioxothiazolidin-2-yl)benzoate
- 4′-(1,2,3,4-TETRAHYDRO-4-(4-HYDROXY-2-OXO-2H-1-BENZOPYRAN-3-YL)-2-NAPHTHALENYL)(1,1′-BIPHENYL)-4-CARBONITRILE, cis-
- 28783-51-9
- 28787-22-6
- 28787-36-2
- 28788-62-7
- 28788-68-3
- 28789-35-7
- 28791-26-6
- 2879-15-4
- 287917-96-8
- 287917-97-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View