-
Name
4-Methoxyindole
- EINECS -0
- CAS No. 4837-90-5
- Article Data45
- CAS DataBase
- Density 1.169 g/cm3
- Solubility
- Melting Point 69-70 °C(lit.)
- Formula C9H9NO
- Boiling Point 306.9 °C at 760 mmHg
- Molecular Weight 147.177
- Flash Point 109.2 °C
- Transport Information
- Appearance light brown to grey crystalline powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 4-methoxy-1H-indole;1H-Indole, 4-methoxy-;4-Methoxy Indole;4-Methoxy-indole;
- PSA 25.02000
- LogP 2.17650
Synthetic route
-
-
103260-65-7
4-methoxy-1H-indole-2-carboxylic acid

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| In quinoline for 0.2h; microwave; | 97% |
| With quinoline; copper for 2h; Heating; | 94% |
| copper In quinoline for 2h; Heating / reflux; | 94% |
| With quinoline; copper oxide-chromium oxide at 200 - 210℃; |
-
-
20876-27-1
(6-methoxy-2-nitrophenyl)acetonitrile

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With hydrogen; acetic acid; 10% palladium on active carbon In ethanol under 2280 Torr; for 2h; Ambient temperature; | 96% |
| With palladium 10% on activated carbon; hydrogen; acetic acid at 20℃; for 12h; |
-
-
112970-67-9
4-methoxy-1-<(4-methylphenyl)sulfonyl>indole

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 12h; Heating; | 96% |
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 3h; Inert atmosphere; | 74% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol; butanone for 6h; Heating; | 95% |
| Stage #1: 1H-indol-4-ol With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 0.5h; Stage #2: methyl iodide In tetrahydrofuran; mineral oil at 20℃; for 1h; | 92% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 18h; Inert atmosphere; | 91% |

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With carbon monoxide; triphenylphosphine; palladium diacetate In acetonitrile under 3040 Torr; for 20h; Heating; | 89% |
-
-
34158-98-0
acetaldehyde 3-methoxyphenylhydrazone

-
A
-
3189-13-7
6-methoxylindole

-
B
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| GIPKh-115 In benzene at 260 - 280℃; (g) phase; | A n/a B 85% |
| GIPKh-115 In benzene at 180 - 320℃; (g) phase. Object of study: effect of electronic factors; | A n/a B 85% |
| GIPKh-115 at 260 - 280℃; Yield given. Yields of byproduct given; |

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol under 3102.9 Torr; for 6h; | 82% |

| Conditions | Yield |
|---|---|
| With C55H49N4OP2Ru In o-xylene at 140℃; under 750.075 Torr; for 48h; Inert atmosphere; Schlenk technique; Green chemistry; | 82% |
| With potassium tert-butylate In o-xylene at 140℃; for 36h; Inert atmosphere; | 55% |
-
-
96631-91-3
3-methoxy-trans-2-[β-(dimethylamino)vinyl]-nitrobenzene

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In tetrahydrofuran for 1h; | 70% |
| With hydrogen; palladium on activated charcoal In methanol; dichloromethane at 20℃; | 63% |

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tert-butyl alcohol for 5h; Heating; | 70% |

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With titanium(III) chloride; ammonium acetate buffer for 0.166667h; | 61% |
-
-
105896-37-5
N-(3-methoxyphenyl)ethenesulfinamide

-
A
-
3189-13-7
6-methoxylindole

-
B
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 1h; | A 25% B 5% |
-
-
22630-08-6
tris(piperidino)-methane

-
-
4837-88-1
6-methoxy-2-nitrotoluene

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With iron; silica gel 1) 120 deg C, 4 h, under vacuum 2) 1 h reflux in toluene-acetic acid; Yield given. Multistep reaction; |
-
-
85355-48-2
(2-Methoxy-6-nitro-phenyl)-acetaldehyde

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With sodium acetate; iron; acetic acid In ethanol; water for 2h; Heating; Yield given; |
-
-
117609-03-7
1-[(E)-2-(2-Methoxy-6-nitro-phenyl)-vinyl]-piperidine

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With titanium(III) chloride; ammonium acetate In acetone Yield given; |
-
-
85355-37-9
Acetic acid 1-chloro-2-(2-methoxy-6-nitro-phenyl)-ethyl ester

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With sodium acetate; iron; acetic acid In ethanol; water for 2h; Heating; |
-
-
75934-38-2
N-(2,2-Diethoxy-ethyl)-2,2,2-trifluoro-N-(3-methoxy-phenyl)-acetamide

-
A
-
3189-13-7
6-methoxylindole

-
B
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With potassium hydroxide; trifluoroacetic anhydride 1) CF3COOH, 56 deg C, 56 h; 2) methanol, room temperature; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
4837-88-1
6-methoxy-2-nitrotoluene

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With pyrrolidine; hydrazine hydrate; nickel 1.) DMF, 125 deg C, 3 h; 2.) methanol, 50-60 deg C, 3 h; Yield given. Multistep reaction; |
-
-
102-71-6
triethanolamine

-
-
536-90-3
m-Anisidine

-
A
-
3189-13-7
6-methoxylindole

-
B
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With tin(ll) chloride; ruthenium trichloride; triphenylphosphine In 1,4-dioxane at 180℃; for 20h; Yield given. Yields of byproduct given; |


-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| at 245 - 250℃; |
-
-
111258-23-2
methyl 4-methoxy-1H-indole-2-carboxylate

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 99 percent / aq. NaOH / 0.5 h / Heating 2: 94 percent / Cu; quinoline / 2 h / Heating View Scheme |
-
-
143702-35-6
(Z)-2-azido-3-(2-methoxyphenyl)acrylic acid methyl ester

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 88 percent / xylene / 1 h / Heating 2: 99 percent / aq. NaOH / 0.5 h / Heating 3: 94 percent / Cu; quinoline / 2 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 68 percent / methanol / 1.5 h / -10 °C 2: 88 percent / xylene / 1 h / Heating 3: 99 percent / aq. NaOH / 0.5 h / Heating 4: 94 percent / Cu; quinoline / 2 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 54 percent / CuI; DMF / acetonitrile / 10 h / 180 °C / 6000.6 - 7500.75 Torr / microwave irradiation 2: 63 percent / H2 / Pd/C / methanol; CH2Cl2 / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 83 percent / CuI; DMF / 3 h / 180 °C / 6000.6 - 7500.75 Torr / microwave irradiation 2: 63 percent / H2 / Pd/C / methanol; CH2Cl2 / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: pyrrolidine / dimethylformamide / 3 h / 125 °C 2: 61 percent / aq. TiCl3, ammonium acetate buffer / 0.17 h View Scheme | |
| Multi-step reaction with 2 steps 1: 4 h / 110 °C 2: TiCl3, 4M NH4OAc / acetone View Scheme | |
| Multi-step reaction with 2 steps 1: N,N-dimethyl-formamide / 3 h / Reflux 2: zinc; acetic acid; hydrogenchloride / water / 0.5 h / 20 °C View Scheme |
-
-
67853-37-6
2-bromo-1-methoxy-3-nitrobenzene

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 89 percent / PPh3 / Pd(dba)2 / toluene / 23.5 h / Heating 2: 89 percent / PPh3, CO / Pd(OAc)2 / acetonitrile / 20 h / 3040 Torr / Heating View Scheme | |
| Multi-step reaction with 5 steps 1: 88 percent / iron / ethanol; acetic acid / 3.5 h / Heating 2: 72 percent / pyridine / 6 h / Ambient temperature 3: tetrakis(triphenylphosphine)palladium(0) / toluene / 36 h / Heating 4: 68 percent / bis(acetonitrile)palladium chloride, p-benzoquinone, lithium chloride / dimethylformamide / 28 h / 100 - 110 °C 5: 96 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
194869-14-2
1-tert-butoxycarbonyl-2-iodo-3-methoxyaniline

-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 85 percent / CuI, Pd(PPh3)2Cl2 / triethylamine / 24 h / 80 °C 2: 70 percent / t-BuOK / 2-methyl-propan-2-ol / 5 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 41 percent / tBuOK / dimethylformamide / 0.5 h / -20 - -10 °C 2: 96 percent / H2, acetic acid / Pd/C (10percent Pd) / ethanol / 2 h / 2280 Torr / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: potassium tert-butylate / N,N-dimethyl-formamide / 2 h / -20 °C 2: acetic acid; palladium 10% on activated carbon; hydrogen / 12 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 1.) hydrobromic acid, sodium nitrite, 2.) coopper(I) bromide / 1.) water, 1,4-dioxane, 0 deg C, 15 min, 2.) a) 0 deg C, 15 min, b) 60 deg C, 15 min 2: sodium hydroxide, / aq. ethanol / 14 h / Ambient temperature 3: 88 percent / iron / ethanol; acetic acid / 3.5 h / Heating 4: 72 percent / pyridine / 6 h / Ambient temperature 5: tetrakis(triphenylphosphine)palladium(0) / toluene / 36 h / Heating 6: 68 percent / bis(acetonitrile)palladium chloride, p-benzoquinone, lithium chloride / dimethylformamide / 28 h / 100 - 110 °C 7: 96 percent / 20percent aq. NaOH / methanol / 12 h / Heating View Scheme |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
90734-97-7
4-methoxyindole-3-carboxaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethyl-formamide With N-chloro-succinimide; triphenylphosphine In tetrahydrofuran for 1h; Reflux; Stage #2: 4-methoxy-1H-indole In tetrahydrofuran for 1h; Reflux; | 100% |
| Stage #1: N,N-dimethyl-formamide With N-chloro-succinimide; triphenylphosphine In tetrahydrofuran at 20℃; Reflux; Stage #2: 4-methoxy-1H-indole In tetrahydrofuran for 1h; Reflux; | 100% |
| Stage #1: 4-methoxy-1H-indole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 45℃; for 1.08333h; Inert atmosphere; Stage #2: With sodium hydroxide In water Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sodium cyanoborohydride; acetic acid | 100% |
| With sodium cyanoborohydride; acetic acid for 0.5h; Ambient temperature; | 98% |
| With sodium cyanoborohydride; acetic acid at 20℃; for 0.833333h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-1H-indole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; Inert atmosphere; Stage #2: bromopentene In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-1H-indole With sodium hydride In acetonitrile at 0℃; for 0.5h; Stage #2: methyl iodide In acetonitrile at 0 - 20℃; for 16h; | 99% |
| With sodium hydride In tetrahydrofuran at 0 - 20℃; | 95% |
| With sodium hydride In tetrahydrofuran for 4h; | 82% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
33797-51-2
eschenmoser's salt

-
-
52335-75-8
(4-methoxy-1H-indol-3-ylmethyl)dimethylamine

| Conditions | Yield |
|---|---|
| In acetic acid; acetonitrile at 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 3h; Electrochemical reaction; Inert atmosphere; | 99% |
| With copper(II) choride dihydrate; oxygen In ethanol at 20℃; for 24h; Irradiation; | 98% |
| With iodine In methanol for 1h; | 75% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
1093759-59-1
4-methoxyindole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap In acetonitrile at 20℃; for 1h; | 99% |
| With dmap; triethylamine In 4-(dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran at 20℃; for 11h; | 97% |
| With dmap In dichloromethane at 20℃; for 8h; Inert atmosphere; | 95% |
-
-
5153-67-3
nitrostyrene

-
-
4837-90-5
4-methoxy-1H-indole

-
-
1187817-63-5, 1236277-14-7
4-methoxy-3-(2-nitro-1-phenylethyl)-1H-indole

| Conditions | Yield |
|---|---|
| With pyrrolidinium 1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoroethoxy)ethanesulfonate at 50℃; for 12h; Friedel Crafts alkylation; | 99% |
-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With (11aR)-3,7-di-9-anthracenyl-10,11,12,13-tetrahydro-5-hydroxy-5-oxide diindeno[7,1de:10,70-fg][1,3,2] dioxaphosphocin In toluene at 60℃; for 3h; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 0.0708333h; Flow reactor; | 99% |
| With tris(pentafluorophenyl)borate In water at 60℃; for 2h; | 52% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
1033591-96-6
1-(1,3-benzothiazol-2-yl)-2,2,2-trifluoroethane-1,1-diol

| Conditions | Yield |
|---|---|
| With (R)-6,6'-bis(2,4,6-triisopropylphenyl)-1,1'-spirobiindanyl-7,7'-diyl hydrogen phosphate In dichloromethane at 20℃; Friedel-Crafts Alkylation; Molecular sieve; enantioselective reaction; | 99% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
18162-48-6
tert-butyldimethylsilyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-1H-indole With n-butyllithium In tetrahydrofuran; hexane at -78 - -10℃; for 0.25h; Stage #2: tert-butyldimethylsilyl chloride In tetrahydrofuran; hexane at 0℃; for 3h; | 98% |

| Conditions | Yield |
|---|---|
| With 1,2,2-tris[(4S)-4-iPr-4,5-dihydro-1,3-oxazol-2-yl]propane; copper(II) bis(trifluoromethanesulfonate) In 2-methyl-propan-1-ol at -25℃; for 50h; Friedel-Crafts alkylation of indoles; | 98% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
5292-53-5
diethyl benzalmalonate

-
-
458563-96-7
(S)-ethyl-2-ethoxycarbonyl-3-[3-(4-methoxyindolyl)]-3-phenyl-propanoate

| Conditions | Yield |
|---|---|
| Stage #1: diethyl benzalmalonate With (3aR,8aS)-2-(3-(3,5-di-tert-butylphenyl)-2,2-bis((S)-4-isopropyl-4,5-dihydrooxazol-2-yl)propyl)-8,8a-dihydro-3aH-indeno[1,2-d]oxazole; copper(II) bis(trifluoromethanesulfonate) In iso-butanol at -25℃; for 0.25h; Friedel Crafts alkylation; Stage #2: 4-methoxy-1H-indole In iso-butanol at -25℃; Friedel Crafts alkylation; optical yield given as %ee; enantioselective reaction; | 98% |
| 1,1'-bis(4-(S)-isopropyl-1,3-oxazolin-2-yl)ethane*Cu(OTf)2 In water; iso-butanol at -15 - 0℃; | 90% |
-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With iodine In methanol at 20℃; for 24h; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; sodium iodide In acetonitrile at 60℃; for 1.5h; Schlenk technique; Inert atmosphere; | 98% |
| With phosphonic acid diethyl ester In acetonitrile at 90℃; for 10h; | 90% |
| With 1,2,2,3,4,4-hexamethylphosphetane 1-oxide; phenylsilane In 1,4-dioxane at 40℃; for 1h; Schlenk technique; Sealed tube; Inert atmosphere; | 89% |
-
-
4837-90-5
4-methoxy-1H-indole

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In acetonitrile at 20℃; for 16h; Inert atmosphere; Irradiation; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane; water at 20℃; | 97% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
75586-32-2
2-phenylethynylbenzaldehyde p-toluenesulphonylhydrazone

-
-
1198746-32-5
1-(4-methoxyindol-3-yl)-2-amino-3-phenylisoquinolinium triflate

| Conditions | Yield |
|---|---|
| With dysprosium(III) trifluoromethanesulfonate In 1,2-dichloro-ethane at 70℃; Inert atmosphere; | 97% |

-
-
4837-90-5
4-methoxy-1H-indole

-
-
13081-18-0
ethyl-3,3,3-trifluoropyruvate

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-1H-indole With C33H52N4O4; zinc trifluoromethanesulfonate In dichloromethane at 35℃; for 0.5h; Stage #2: ethyl-3,3,3-trifluoropyruvate In dichloromethane at 0℃; for 0.5h; Friedel Crafts alkylation; optical yield given as %ee; enantioselective reaction; | 97% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
776-76-1
methyldiphenylsilane

-
-
1343516-70-0
4-methoxy-1-(methyldiphenylsilyl)-1H-indole

| Conditions | Yield |
|---|---|
| With dodecacarbonyl-triangulo-triruthenium In toluene at 120℃; for 12h; Schlenk technique; Inert atmosphere; | 97% |
| With pyridine; zinc trifluoromethanesulfonate In propiononitrile at 80℃; for 15h; Inert atmosphere; | 94% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
34157-83-0
celastrol

| Conditions | Yield |
|---|---|
| With aluminium(III) chloride hexahydrate In dichloromethane at 20℃; for 3h; Friedel-Crafts Alkylation; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In acetonitrile at 50℃; for 24h; Schlenk technique; Inert atmosphere; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With copper diacetate; 4,4'-di-tert-butyl-2,2'-bipyridine at 90℃; for 24h; Schlenk technique; regioselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| With piperidine; 2-(((S)-1-hydroxy-1,1,3-triphenylpropan-2-ylimino)methyl)-6-chlorophenol; zinc trifluoromethanesulfonate In toluene at 20℃; for 24h; Friedel Crafts alkylation; optical yield given as %ee; enantioselective reaction; | 96% |
-
-
4837-90-5
4-methoxy-1H-indole

-
-
13081-18-0
ethyl-3,3,3-trifluoropyruvate

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-1H-indole With C33H52N4O4; zinc trifluoromethanesulfonate In dichloromethane at 35℃; for 0.5h; Stage #2: ethyl-3,3,3-trifluoropyruvate In dichloromethane at 0℃; for 0.5h; Friedel Crafts alkylation; optical yield given as %ee; enantioselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 20℃; for 0.0833333h; Green chemistry; | 96% |
4-Methoxyindole Specification
The 4-Methoxyindole, with the CAS registry number 4837-90-5, has the systematic name of 4-methoxy-1H-indole. And the molecular formula of this chemical is C9H9NO. It is a kind of light brown to grey crystalline powder which is sensitive to light, and it belongs to the folllowing product categories: Blocks; IndolesOxindoles; Indoles and derivatives; Pyrroles & Indoles; Indoline & Oxindole; Indole Series; Indoles; Simple Indoles; Pyrroles & Indoles; Building Blocks; Heterocyclic Building Blockss. What's more, it is used as medicine intermediate.
The physical properties of 4-Methoxyindole are as following: (1)ACD/LogP: 2.06; (2)# of Rule of 5 Violations: 0 ; (3)#H bond acceptors: 2; (4)#H bond donors: 1; (5)#Freely Rotating Bonds: 1; (6)Polar Surface Area: 14.16 Å2; (7)Index of Refraction: 1.637; (8)Molar Refractivity: 45.2 cm3; (9)Molar Volume: 125.8 cm3; (10)Polarizability: 17.92×10-24cm3; (11)Surface Tension: 45.7 dyne/cm; (12)Density: 1.169 g/cm3; (13)Flash Point: 109.2 °C; (14)Enthalpy of Vaporization: 52.56 kJ/mol; (15)Boiling Point: 306.9 °C at 760 mmHg; (16)Vapour Pressure: 0.00136 mmHg at 25°C.
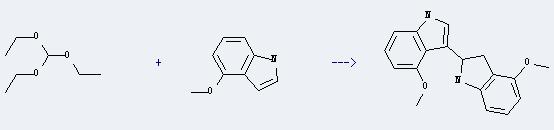
Uses of 4-Methoxyindole: It can react with triethoxymethane to produce 3-(4-Methoxyindolin-2-yl)-4-methoxyindole. This reaction will need reagent Cl3CCOOH, and the solvent CH2Cl2. The reaction time is 25 minutes with ambient temperature, and the yield is about 60%.
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Avoid contact with skin and eyes; Wear suitable protective clothing; In case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O(c1cccc2c1ccn2)C
(2)InChI: InChI=1/C9H9NO/c1-11-9-4-2-3-8-7(9)5-6-10-8/h2-6,10H,1H3
(3)InChIKey: LUNOXNMCFPFPMO-UHFFFAOYAZ
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View