Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:5153-67-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:5153-67-3
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:5153-67-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:5153-67-3
Min.Order:1 Kilogram
FOB Price: $70.0 / 90.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:5153-67-3
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:5153-67-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
(E)-2-Nitroethenylbenzene CAS:5153-67-3 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic
Cas:5153-67-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:5153-67-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:5153-67-3
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:5153-67-3
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:5153-67-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:5153-67-3
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Cas:5153-67-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:5153-67-3
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:5153-67-3
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:5153-67-3
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:5153-67-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:5153-67-3
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:5153-67-3
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryWin-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:5153-67-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:5153-67-3
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM is one of China's leading providers of integrated fine chemical services including offering, research and development, Custom manufacturing business, as well as other Value-added customer services, for diversified range products of chemicals
Cas:5153-67-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Chemsigma International Co.,Ltd.
bulk productiongoods in stockswe have the best competitive price in the marketmeantime,we are committed to service to our every customer Chemsigma International Co.,Ltd. is a chemical manufacturer, specialize in custom synthesis and organic chemical
Cas:5153-67-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryWuhan Circle Star Chem-medical Technology co.,Ltd.
1,we produce and sell good chemicals around the world.2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%.3,our staff consists of highly qualified in
Cas:5153-67-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With sulfonic acid amine MCM-41 mesoporous silica nanoparticles at 90℃; for 20h; Temperature; | 99% |
| With m-aminobenzoic acid-(L)-phenylglycinol ammonium at 20℃; for 40h; | 99% |
| With ammonium acetate In toluene at 100℃; for 22h; | 99% |
-
-
141377-54-0, 145920-96-3, 149495-00-1, 15990-45-1
2-nitro-1-phenylethan-1-ol

-
-
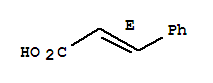
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| Stage #1: 2-nitro-1-phenylethan-1-ol With potassium carbonate In water for 0.25h; Green chemistry; Stage #2: With hydrogenchloride In water pH=6; Green chemistry; | 96% |
| With H-Y zeolite In toluene for 8h; Heating; | 93% |
| With H-Y zeolite In toluene for 8h; Product distribution; Heating; also with other zeolites; | 93% |
-
-
292638-84-7
styrene

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With silver(I) nitrite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In 1,2-dichloro-ethane at 70℃; for 12h; Kinetics; Reagent/catalyst; Solvent; Concentration; Molecular sieve; Inert atmosphere; stereoselective reaction; | 95% |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In 1,4-dioxane at 80℃; for 1h; Reagent/catalyst; Solvent; Green chemistry; stereoselective reaction; | 95% |
| Stage #1: styrene With chloro-trimethyl-silane; copper(ll) sulfate pentahydrate; guanidine nitrate In acetonitrile at 0 - 20℃; Inert atmosphere; Stage #2: With triethylamine In acetonitrile at 20℃; for 0.5h; Inert atmosphere; stereoselective reaction; | 92% |

| Conditions | Yield |
|---|---|
| With tetranitromethane In acetonitrile Irradiation; | 95% |
-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
A
-
5153-67-3
nitrostyrene

-
B
-
117538-84-8
(1,3-dinitropropan-2-yl)benzene

| Conditions | Yield |
|---|---|
| aminopropyl-functionalized silica at 90℃; for 2h; Product distribution; Further Variations:; Catalysts; | A 95% B 3% |
| MCM-41-NHMe at 90℃; for 1h; | A 81% B n/a |
| With azide amine MCM-41 mesoporous silica nanoparticles at 90℃; for 20h; | A 42% B 47% |


| Conditions | Yield |
|---|---|
| With copper(II) nitrate In acetonitrile at 110℃; for 8h; Reagent/catalyst; Solvent; Temperature; Sealed tube; Green chemistry; regioselective reaction; | 94% |
| With pyridine; Iron(III) nitrate nonahydrate In toluene at 100℃; for 12h; Reagent/catalyst; Solvent; | 90% |
| With chloro-trimethyl-silane; copper(II) nitrate trihydrate In acetonitrile at 100℃; for 2h; Reagent/catalyst; stereoselective reaction; | 89% |

| Conditions | Yield |
|---|---|
| With lepidocricite; water In acetonitrile at 105℃; for 48h; Henry Nitro Aldol Condensation; Inert atmosphere; | 93% |
| With phosphotungstic acid immobilized on amine-grafted mesoporous silica at 50℃; for 12h; Inert atmosphere; | 92% |
| With sulfonic acid and 3-aminopropyltriethoxysilane functionalized at K10-montmorillonite (SO3H-APTES at K10-MMT) at 90℃; for 2h; Reagent/catalyst; Time; Henry Nitro Aldol Condensation; | 99.2 %Chromat. |
| Stage #1: benzaldehyde dimethyl acetal With highly porous and stable acid-base bifunctional metal-organic framework chromium(III) terephthalate containing sulfonic acid and amine groups (MIL-101-NH2-SO3H) In o-xylene at 49.84℃; Stage #2: nitromethane In o-xylene at 49.84℃; Henry Nitro Aldol Condensation; |
-
-
75-52-5
nitromethane

-
-
1125-88-8
benzaldehyde dimethyl acetal

-
A
-
5153-67-3
nitrostyrene

-
B
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| With FeO2H; water In acetonitrile at 105℃; for 48h; Reagent/catalyst; Henry Nitro Aldol Condensation; Inert atmosphere; | A 93% B 6% |
| With lepidocricite; water In acetonitrile at 80℃; for 12h; Reagent/catalyst; Henry Nitro Aldol Condensation; Inert atmosphere; | A 41.6% B 57% |


-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With sodium iodide; tin(ll) chloride In ethanol for 0.116667h; Reflux; Green chemistry; | 92% |
-
-
31771-33-2
2-phenyl-4,4,6-trimethyltetrahydro-(2H)-1,3-oxazine

-
-
75-52-5
nitromethane

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With acetic acid In acetonitrile for 5h; Heating; | 90% |
-
-
3425-99-8, 37887-94-8
(1R*,2R*)-1,2-dibromo-2-phenyl-1-nitroethane

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With 1-methyl-3-pentyl-1H-imidazolium tetrafluoroborate at 130 - 135℃; for 0.05h; microwave irradiation; | 89% |
| With dimethyl sulfoxide at 25℃; for 18h; | 51% |
-
-
92550-70-4
4-methylphenylstyryl sulfide

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In water at 100℃; for 12h; | 87% |
-
-
292638-84-7
styrene

-
A
-
5153-67-3
nitrostyrene

-
B
-
100-52-7
benzaldehyde

-
C
-
100-47-0
benzonitrile

-
D
-
98-86-2
acetophenone

-
E
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With formic acid; sodium nitrite In acetonitrile at 70℃; for 4h; Mechanism; Reagent/catalyst; Solvent; Temperature; Schlenk technique; | A n/a B n/a C 84% D n/a E n/a |

-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
A
-
5153-67-3
nitrostyrene

-
B
-
141377-54-0, 145920-96-3, 149495-00-1, 15990-45-1
2-nitro-1-phenylethan-1-ol

| Conditions | Yield |
|---|---|
| With CaO modified benzyl bromide at 102℃; for 5h; Reagent/catalyst; Temperature; Henry Nitro Aldol Condensation; | A 15.6% B 82.9% |
| aminopropyltriethoxysilane on silica-alumina at 100℃; for 6h; Product distribution; Kinetics; Further Variations:; Catalysts; | A 99 % Chromat. B n/a |

-
-
3425-99-8, 37887-94-8
C8H7Br2NO2

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; sodium carbonate In dimethyl sulfoxide at 20℃; for 1.5h; Inert atmosphere; Irradiation; | 81% |

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With dmap; di-tert-butyl dicarbonate In toluene at 45 - 50℃; | 80% |

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In dimethyl sulfoxide at 32 - 35℃; for 0.5h; Green chemistry; | A 7% B 73% |
-
-
7422-28-8, 17421-57-7, 50849-52-0
(Z)-1-Phenyl-2-(trimethylstannyl)aethen

-
A
-
5153-67-3
nitrostyrene

-
B
-
15241-23-3
(Z)-nitrostyrene

| Conditions | Yield |
|---|---|
| With tetranitromethane In dimethyl sulfoxide for 3h; Ambient temperature; | A n/a B 65% |

-
A
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 110℃; for 48h; | A 15% B 61% |

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; 3-chloro-benzenecarboperoxoic acid at 0 - 60℃; for 10h; Henry Nitro Aldol Condensation; Inert atmosphere; | 61% |

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate; sodium nitrite for 0.133333h; Microwave irradiation; | 57% |
-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
-
590-86-3
isovaleraldehyde

-
A
-
5153-67-3
nitrostyrene

-
C
-
1000072-29-6
2-(prop-2-yl)-3-phenylbutanal

| Conditions | Yield |
|---|---|
| Stage #1: nitromethane; benzaldehyde With O-(tert-butyldiphenylsilyl)-L-tyrosine lithium salt; magnesium sulfate In dichloromethane at 25℃; for 48h; Stage #2: isovaleraldehyde In dichloromethane at 25℃; for 72h; Michael addition; optical yield given as %ee; enantioselective reaction; | A 50% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; silver(I) acetate In 1,2-dimethoxyethane; dimethyl sulfoxide at 100℃; for 24h; Glovebox; Schlenk technique; Inert atmosphere; regioselective reaction; | 50% |
-
-
292638-84-7
styrene

-
A
-
5153-67-3
nitrostyrene

-
B
-
7064-02-0
phenyl (5-phenyl-4,5-dihydroisoxazol-3-yl)methanone

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; manganese(III) triacetate dihydrate In acetonitrile at 20℃; for 5h; Schlenk technique; Inert atmosphere; | A 50% B 5% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; bismuth (III) nitrate pentahydrate In benzene at 70℃; for 12h; Inert atmosphere; | 48% |
-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
A
-
5153-67-3
nitrostyrene

-
B
-
149495-00-1
(S)-1-phenyl-2-nitroethanol

| Conditions | Yield |
|---|---|
| With bis(α-ethylphenylamine) cupric chloride In methanol at 20℃; for 72h; Henry reaction; optical yield given as %ee; enantioselective reaction; | A 46% B n/a |


| Conditions | Yield |
|---|---|
| With peroxynitrous acid In acetonitrile at 5 - 10℃; for 1h; | A 45% B n/a |
-
-
104-53-0
3-phenyl-propionaldehyde

-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
A
-
5153-67-3
nitrostyrene

-
B
-
1089665-81-5
2-benzyl-2-(2-nitro-1-phenylethyl)ethanal

-
C
-
1021394-80-8
(2S,3R)-4-nitro-2-(1-phenylmethyl)-3-phenylbutanal

-
D
-
1235612-22-2
2-benzyl-2-(2-nitro-1-phenylethyl)ethanal

| Conditions | Yield |
|---|---|
| Stage #1: nitromethane; benzaldehyde With O-(tert-butyldiphenylsilyl)-L-tyrosine lithium salt; magnesium sulfate In dichloromethane at 25℃; for 48h; Stage #2: 3-phenyl-propionaldehyde In dichloromethane at 25℃; for 72h; Michael addition; enantioselective reaction; | A 43% B n/a C n/a D n/a |

-
-
141377-54-0, 145920-96-3, 149495-00-1, 15990-45-1
2-nitro-1-phenylethan-1-ol

-
A
-
5153-67-3
nitrostyrene

-
B
-
149495-00-1
(S)-1-phenyl-2-nitroethanol

-
C
-
145920-96-3
(R)-2-nitro-1-phenylethanol

| Conditions | Yield |
|---|---|
| With vinyl acetate; Pseudomonas fluorescens amino lipase In hexane; toluene at 30℃; for 48h; Title compound not separated from byproducts; | A 40% B n/a C n/a |

-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
A
-
5153-67-3
nitrostyrene

-
B
-
149495-00-1
(S)-1-phenyl-2-nitroethanol

-
C
-
145920-96-3
(R)-2-nitro-1-phenylethanol

| Conditions | Yield |
|---|---|
| With bis(α-ethylphenylamine) copper acetate In methanol at 20℃; for 72h; Henry reaction; optical yield given as %ee; enantioselective reaction; | A 40% B n/a C n/a |
| With bis(α-ethylphenylamine) zinc acetate In methanol at 20℃; for 72h; Henry reaction; optical yield given as %ee; enantioselective reaction; | A 32% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With magnesium(II) perchlorate; model of NADH grafted on silica matrix In acetonitrile at 65℃; for 120h; | 100% |
| With sodium tetrahydroborate; silica gel In chloroform; isopropyl alcohol at 0 - 23℃; for 1.33333h; Inert atmosphere; | 99% |
| With hydrogen; tris(triphenylphosphine)rhodium(l) chloride In benzene at 50℃; under 2585.81 Torr; for 14h; | 98% |
-
-
5153-67-3
nitrostyrene

-
-
117390-40-6
5-carbamoyl thieno<2,3-b>pyridine

-
-
125056-17-9
bromo-8 octyl dimethylchlorosilane

-
-
6125-24-2
2-Phenylnitroethane

| Conditions | Yield |
|---|---|
| With magnesium(II) perchlorate; silice (Merck Kieselgel Art.:7734) Product distribution; multistep reaction; 1.) grafting on a silica matrix, reflux, in toluene, 12 h, 2.) reflux, toluene, 5 days. 3.) acetonitrile, 65 deg C, 5 days; reagent quqntity, reaction time varied; | 100% |

-
-
5153-67-3
nitrostyrene

-
-
60947-52-6
(1,4-dinitrobutane-2,3-diyl)dibenzene

| Conditions | Yield |
|---|---|
| With samarium diiodide; isopropylamine In water | 100% |
| With tetraethylammonium tosylate In water; N,N-dimethyl-formamide at 0℃; Electrolysis; | 71% |
| With titanium(III) chloride; ammonium acetate In tetrahydrofuran; water at 0℃; for 0.5h; | 6% |

| Conditions | Yield |
|---|---|
| With chiral (OCHPhCHMeN(i-Pr))PNMe2; copper(I) triflate In hexane; toluene at -30℃; for 3h; Addition; | 100% |
| copper(I) 2-(Me2NCH2)-3-(Me3Si)-naphthalene-1-thiolate In diethyl ether; hexane at -20℃; for 4h; Michael addition; | 98% |
| With magnesium bromide In diethyl ether for 1h; Ambient temperature; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: nitrostyrene With 2-[(S)-4-isopropyl-4,5-dihydrooxazol-2-yl]-N-[2-(-4-phenyl-4,5-dihydrothiazol-2-yl)phenyl]benzenamine; zinc trifluoromethanesulfonate In toluene at 20℃; for 0.166667h; Inert atmosphere; Stage #2: indole In toluene at -20℃; for 15h; Friedel Crafts alkylation; Inert atmosphere; | 100% |
| With Zn(2+)*2C6H6NO2S2(1-) In ethanol at 20℃; for 24h; Solvent; Reagent/catalyst; | 100% |
| Stage #1: nitrostyrene With (S)-10,10'-bis[(S)-4-isopropyl-4,5-dihydrooxazol-2-yl]-9,9'-biphenanthrene; samarium trifluoromethanesulfonate In chloroform at 20℃; for 0.25h; Inert atmosphere; Stage #2: indole In chloroform at 20℃; for 18h; Friedel-Crafts alkylation; Inert atmosphere; | 99% |
-
-
603-76-9
1-methylindole

-
-
5153-67-3
nitrostyrene

-
-
109811-96-3
1-methyl-3-(2-nitro-1-phenylethyl)-1H-indole

| Conditions | Yield |
|---|---|
| With Zn(2+)*2C6H6NO2S2(1-) In ethanol at 20℃; for 24h; | 100% |
| With 1H-pyrrolo[2,3-b]pyridinium substituted-borate salt In toluene at 0 - 20℃; for 24h; | 99% |
| Stage #1: nitrostyrene With (S)-10,10'-bis[(S)-4-isopropyl-4,5-dihydrooxazol-2-yl]-9,9'-biphenanthrene; zinc(II) trifluoroacetate In diethyl ether at 20℃; for 0.25h; Inert atmosphere; Stage #2: 1-methylindole In diethyl ether at 20℃; Friedel-Crafts alkylation; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With dibenzoyl peroxide In benzene for 6h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With dibenzoyl peroxide In benzene for 12h; Heating; | 100% |
| With di-tert-butyl peroxide; copper diacetate In 1,2-dichloro-ethane at 100℃; Schlenk technique; Inert atmosphere; | 86% |
| With di-tert-butyl peroxide; copper In 1,2-dichloro-ethane at 110℃; for 8h; Sealed tube; Inert atmosphere; Schlenk technique; | 85% |
-
-
5153-67-3
nitrostyrene

-
-
108-94-1
cyclohexanone

-
-
4591-64-4, 51262-17-0, 51262-18-1, 84025-84-3, 84025-87-6, 84025-83-2
(S)-2-[(R)-2-nitro-1-phenylethyl]cyclohexanone

| Conditions | Yield |
|---|---|
| With 1-[(S)-2'-methylpyrrolidine]-4-aza-1-azoniabicyclo[2.2.2]octane tetrafluoroborate at 20℃; for 22h; Michael addition; Ionic liquid; optical yield given as %ee; enantioselective reaction; | 100% |
| With (S)-6-phenyl-3-[(S)-pyrrolidin-2-ylmethyl]-2-thioxotetrahydropyrimidin-4(1H)-one; water; 4-nitro-benzoic acid In tetrahydrofuran for 18h; Michael reaction; optical yield given as %ee; enantioselective reaction; | 100% |
| With 4-nitro-benzoic acid; (S)-6-phenyl-3-[(S)-pyrrolidin-2-ylmethyl]-2-thioxotetrahydropyrimidin-4(1H)-one In tetrahydrofuran; water Product distribution / selectivity; Michael Reaction; | 100% |

| Conditions | Yield |
|---|---|
| With calcium p-methoxyphenolate; anti-5,4-diphenyl pyridinebisoxazoline In toluene at -20℃; for 432h; Michael condensation; Molecular sieve; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 100% |
| With C34H46N4*Ni(2+)*2Br(1-) In dichloromethane at 4℃; for 16h; Catalytic behavior; Solvent; Michael Addition; enantioselective reaction; | 99% |
| With (5,7-bis(trifluoromethyl)-1H-benzoimidazol-2-yl)-9-epiquinine amine In dichloromethane at -20℃; for 40h; asymmetric Michael addition; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 98% |
-
-
29943-42-8
Tetrahydro-4H-pyran-4-one

-
-
5153-67-3
nitrostyrene

| Conditions | Yield |
|---|---|
| With 1-[(S)-2'-methylpyrrolidine]-4-aza-1-azoniabicyclo[2.2.2]octane tetrafluoroborate at 20℃; for 17h; Michael addition; Ionic liquid; optical yield given as %ee; enantioselective reaction; | 100% |
| With bis(((S)-pyrrolidin-2-yl)methyl) phosphonate dihydrochloride; sodium hydrogencarbonate at 20℃; for 17h; Michael condensation; Ionic liquid; optical yield given as %ee; enantioselective reaction; | 98% |
| Stage #1: Tetrahydro-4H-pyran-4-one With N-((S)-1-phenylethyl)-5-((S)-pyrrolidin-2-yl)oxazole-4-carboxamide; acetic acid In neat (no solvent) at 20℃; for 0.0833333h; Michael Addition; Stage #2: nitrostyrene In neat (no solvent) at 20℃; for 30h; Reagent/catalyst; Michael Addition; stereoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: nitrostyrene; tris(allyl)aluminum In diethyl ether at 0℃; for 0.0333333h; Stage #2: With hydrogen bromide In diethyl ether at 0℃; Further stages.; | 100% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 4h; Michael-type addition; | 100% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 4h; Michael-type addition; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | 100% |

| Conditions | Yield |
|---|---|
| With (S)-di-tert-butyl 2-{3-[(1S,2S)-2-amino-1,2-diphenylethyl]thioureido}succinate; acetic acid In water; toluene at 20℃; for 48h; Michael reaction; optical yield given as %ee; enantioselective reaction; | 100% |
| Stage #1: acetone With ((S)-pyrrolidin-2-yl)methyl phenylcarbamate In toluene at 20℃; for 0.5h; Michael Addition; Stage #2: nitrostyrene In toluene at 20℃; for 24h; Michael Addition; stereoselective reaction; | 93% |
| With water; O,O-diethyl [(1S,2S)-2-amino-1,2-diphenylethyl]phosphoramidothioate; acetic acid In toluene at 70℃; for 24h; Reagent/catalyst; Solvent; Temperature; Michael Addition; enantioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With : H-D-Pro-Pro-Glu supported on polyethyleneglycol-polystyrene (TentaGel resin) In chloroform; isopropyl alcohol at -15℃; for 72h; optical yield given as %ee; enantioselective reaction; | 100% |
| With 4-methyl-morpholine; C16H26N4O5 In neat (no solvent) at 20℃; for 72h; stereoselective reaction; | 96% |
| With 4-methyl-morpholine; H-D-Pro-L-Pro-L-Glu-NH2*TFA In chloroform; isopropyl alcohol at 20℃; for 48h; optical yield given as %ee; stereoselective reaction; | 93% |
-
-
104-53-0
3-phenyl-propionaldehyde

-
-
5153-67-3
nitrostyrene

-
-
1021394-80-8
(2S,3R)-4-nitro-2-(1-phenylmethyl)-3-phenylbutanal

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; C16H26N4O5 In neat (no solvent) at 20℃; for 24h; stereoselective reaction; | 100% |
| With 4-methyl-morpholine; (R)-2-(((2S,3S,4R)-2-((2-carboxyethoxy)methyl)-4-methoxytetrahydro-2H-pyran-3-yl)carbamoyl)pyrrolidin-1-ium 2,2,2-trifluoroacetate In dichloromethane at 20℃; for 22h; Michael Addition; Inert atmosphere; stereoselective reaction; | 97% |
| With 4-methyl-morpholine; H-D-Pro-L-Pro-L-Glu-NH2*TFA In chloroform; isopropyl alcohol at 20℃; optical yield given as %ee; enantioselective reaction; | 94% |
-
-
5153-67-3
nitrostyrene

-
-
123-38-6
propionaldehyde

-
-
475294-87-2
(2S,3R)-2-methyl-4-nitro-3-phenylbutanal

| Conditions | Yield |
|---|---|
| With : H-D-Pro-Pro-Glu supported on polyethyleneglycol-polystyrene (TentaGel resin) In chloroform; isopropyl alcohol at -15℃; for 17h; optical yield given as %ee; enantioselective reaction; | 100% |
| With (S)-2-(triphenylsilyl)pyrrolidine In hexane at -20℃; for 48h; Michael reaction; optical yield given as %de; diastereoselective reaction; | 97% |
| With 4-methyl-morpholine; C16H26N4O5 In neat (no solvent) at 20℃; for 24h; stereoselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With : H-D-Pro-Pro-Glu supported on polyethyleneglycol-polystyrene (TentaGel resin) In chloroform; isopropyl alcohol at -15℃; for 20h; optical yield given as %ee; enantioselective reaction; | 100% |
| With 4-methyl-morpholine; (R)-2-(((2S,3S,4R)-2-((2-carboxyethoxy)methyl)-4-methoxytetrahydro-2H-pyran-3-yl)carbamoyl)pyrrolidin-1-ium 2,2,2-trifluoroacetate In dichloromethane at 20℃; for 22h; Michael Addition; Inert atmosphere; stereoselective reaction; | 97% |
| With 4-methyl-morpholine; H-D-Pro-L-Pro-L-Glu-NH2*TFA In chloroform; isopropyl alcohol at 20℃; optical yield given as %ee; enantioselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With : H-D-Pro-Pro-Glu supported on polyethyleneglycol-polystyrene (TentaGel resin) In chloroform; isopropyl alcohol at -15℃; for 19h; optical yield given as %ee; enantioselective reaction; | 100% |
| With 4-methyl-morpholine; C16H26N4O5 In neat (no solvent) at 20℃; for 24h; stereoselective reaction; | 100% |
| With 4-methyl-morpholine; H-D-Pro-L-Pro-L-Glu-NH2*TFA In chloroform; isopropyl alcohol at 20℃; optical yield given as %ee; enantioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With (S)-6-phenyl-3-[(S)-pyrrolidin-2-ylmethyl]-2-thioxotetrahydropyrimidin-4(1H)-one; water; 4-nitro-benzoic acid In tetrahydrofuran for 18h; Michael reaction; optical yield given as %ee; enantioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With 2Zn(2+)*2NO3(1-)*2C3H7NO*2Y(3+)*4C14H11NO3(2-) In ethanol; water at 20℃; for 0.25h; Reagent/catalyst; Michael Addition; | 100% |
| With water; diethylamine at 20℃; for 1h; Reagent/catalyst; Time; Michael Addition; Green chemistry; | 99% |
-
-
5153-67-3
nitrostyrene

-
-
104497-09-8
6-amino-1,3-dimethylbarbituric acid

-
-
95834-23-4
6-amino-1,3-dimethyl-5-(2-nitro-1-phenylethyl)pyrimidine-2,4(1H,3H)-dione

| Conditions | Yield |
|---|---|
| With 2Zn(2+)*2NO3(1-)*2C3H7NO*2Y(3+)*4C14H11NO3(2-) In ethanol; water at 20℃; for 0.25h; Michael Addition; | 100% |
-
-
104-53-0
3-phenyl-propionaldehyde

-
-
5153-67-3
nitrostyrene

-
-
1089665-81-5
2-benzyl-2-(2-nitro-1-phenylethyl)ethanal

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; (S)-2-(((2S,3R)-2-((2-carboxyethoxy)methyl)tetrahydro-2H-pyran-3-yl)carbamoyl)pyrrolidin-1-ium 2,2,2-trifluoroacetate In dichloromethane at 20℃; for 19h; Michael Addition; Inert atmosphere; stereoselective reaction; | 100% |
-
-
95-20-5
2-methyl-1H-indole

-
-
5153-67-3
nitrostyrene

-
-
109809-52-1
2-methyl-3-(2-nitro-1-phenylethyl)-1H-indole

| Conditions | Yield |
|---|---|
| With aminosulfonic acid at 60℃; for 0.5h; Michael addition; | 99% |
| Stage #1: nitrostyrene With (S)-10,10'-bis[(S)-4-isopropyl-4,5-dihydrooxazol-2-yl]-9,9'-biphenanthrene; zinc(II) trifluoroacetate In diethyl ether at 20℃; for 0.25h; Inert atmosphere; Stage #2: 2-methyl-1H-indole In diethyl ether at 20℃; Friedel-Crafts alkylation; Inert atmosphere; | 99% |
| With pyrrolidinium 1,1,2,2-tetrafluoro-2-(1,1,2,2-tetrafluoroethoxy)ethanesulfonate at 50℃; for 12h; Friedel Crafts alkylation; | 99% |
-
-
5153-67-3
nitrostyrene

-
-
141-97-9
ethyl acetoacetate

-
-
72709-62-7
ethyl 2-acetyl-4-nitro-3-phenylbutyrate

| Conditions | Yield |
|---|---|
| With lithium phenolate; (S)-3,3′-bis[hydroxy(diphenyl)methyl]-1,1′-binaphthalene-2,2′-diol In dichloromethane at 20℃; for 48h; Michael reaction; Inert atmosphere; | 99% |
| With triethylamine at 20℃; Michael Addition; Green chemistry; | 99% |
| bis(acetylacetonate)nickel(II) In 1,4-dioxane at 90℃; for 17h; | 98% |
-
-
5153-67-3
nitrostyrene

-
-
123-54-6
acetylacetone

-
-
72709-61-6
(+/-)-3-(2-nitro-1-phenylethyl)pentane-2,4-dione

| Conditions | Yield |
|---|---|
| With triethylamine at 20℃; Michael Addition; Green chemistry; | 99% |
| With N21,N23-bis(4-bromobenzyl)-2-(((2-(pyridin-3-yl)ethyl)amino)methyl)-5,10,15,20-tetrakis(3,5-ditert-butyl-4-oxo-cyclohexa-2,5-dienylidene)porphyrinogen In ethanol at 20℃; for 16h; Catalytic behavior; Kinetics; Reagent/catalyst; Solvent; Michael Addition; Inert atmosphere; | 99% |
| With 1-methyl-3-(2-(piperidin-1-yl)ethyl)-1H-imidazol-3-ium-chloride In neat (no solvent) at 20℃; for 0.5h; Reagent/catalyst; Time; Michael Addition; Sonication; | 98% |

| Conditions | Yield |
|---|---|
| With lithium phenolate; (S)-3,3′-bis[hydroxy(diphenyl)methyl]-1,1′-binaphthalene-2,2′-diol In dichloromethane at 20℃; for 48h; Michael reaction; Inert atmosphere; | 99% |
| With triethylamine at 20℃; Michael Addition; Green chemistry; | 99% |
| With C16H25N5S In tetrahydrofuran at 25℃; for 2h; Michael Addition; | 96% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
















 Xi
Xi