-
Name
4-Phenyltoluene
- EINECS 211-409-0
- CAS No. 644-08-6
- Article Data1152
- CAS DataBase
- Density 0.984 g/cm3
- Solubility Insoluble in water. Soluble in alcohol and ether, very soluble in organic solvents.
- Melting Point 44-47 °C(lit.)
- Formula C13H12
- Boiling Point 267.799 °C at 760 mmHg
- Molecular Weight 168.238
- Flash Point 117.819 °C
- Transport Information
- Appearance White crystalline powder
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Biphenyl,4-methyl- (8CI);(4-Methylphenyl)benzene;1-Methyl-4-phenylbenzene;4-Methyl-1,1'-biphenyl;4-Methylbiphenyl;4-Methyldiphenyl;4-Phenyltoluene;NSC 407669;p-Methylbiphenyl;p-Methyldiphenyl;p-Phenyltoluene;
- PSA 0.00000
- LogP 3.66200
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium phosphate; (η5-C5H5)Re(NO)(P(C6H5)3)(C(CH3)3)2; palladium diacetate In toluene at 100℃; for 1h; Suzuki cross-couplings reaction; | 100% |
| With N,N-dicyclohexyl-2-(dicyclohexylphosphanyl)benzenesulfonamide; tris(dibenzylideneacetone)dipalladium (0); caesium carbonate In 1,4-dioxane at 80℃; for 3h; Suzuki-Miyaura cross coupling; | 100% |
| With potassium phosphate; 3-(2,6-diisopropylphenyl)-1-(2-diphenylphosphanylbenzyl)-3H-imidazol-1-ium chloride; bis(η3-allyl-μ-chloropalladium(II)) In 1,4-dioxane at 80℃; for 12h; Product distribution; Further Variations:; Catalysts; Reagents; Solvents; Suzuki cross-coupling; | 100% |

| Conditions | Yield |
|---|---|
| With Cs2O3; PCy3 adduct of cyclopalladated ferrocenylimine In 1,4-dioxane at 100℃; for 15h; Suzuki cross-coupling reaction; | 100% |
| With [PdI(2-(2-thiophenyl)-4,4-dimethyloxazoline)(1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene)]; potassium carbonate In tetrahydrofuran; water at 90℃; for 18h; Catalytic behavior; Reagent/catalyst; Suzuki-Miyaura Coupling; | 100% |
| With caesium carbonate; palladium diacetate; 1,3-di-([N-(2,4,6-Me3Ph)imidazolium-3-yl]Me)-2,4,6-Me3Ph*2Cl- In 1,4-dioxane at 80℃; for 1.5h; Suzuki reaction; | 99% |

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,6-diisopropylphenyl)-imidazolium][Ni(PPh3)Cl3] In tetrahydrofuran at 30℃; for 1h; Kumada Cross-Coupling; Schlenk technique; Inert atmosphere; | 100% |
| With C70H40Cl2F68N2O6Pd2 In tetrahydrofuran at 100℃; for 0.0166667h; Kumada coupling reaction; Microwave irradiation; | 96% |
| With C17H36ClN6NiP2(1+)*Cl(1-) In tetrahydrofuran for 6h; Reagent/catalyst; Kumada Cross-Coupling; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; palladium dichloride In pyridine for 3h; Suzuki cross-coupling; Heating; | 100% |
| With potassium carbonate; carbapalladacycle complex*periodic mesoporous organosilica for 24h; Suzuki coupling; Heating; | 100% |
| With tetra-butylammonium acetate; Pd EnCat-30TM In ethanol at 120℃; for 0.166667h; Suzuki cross-coupling; microwave irradiation; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In water at 80℃; Catalytic behavior; Suzuki-Miyaura Coupling; Green chemistry; | 100% |
| With aluminum oxide; potassium fluoride; palladium at 100℃; for 4h; Suzuki reaction; | 99% |
| With sodium hydroxide; tetrabutylammomium bromide; palladium on activated charcoal at 100℃; for 2h; Suzuki-Miyaura cross-coupling; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; copper-palladium In N,N-dimethyl-formamide at 110℃; for 2h; Suzuki cross-coupling; | 100% |
| With sodium hydroxide; Pd-dodecanethiolate nanoparticles In tetrahydrofuran at 20℃; for 24h; Suzuki-Miyaura cross-coupling; | 100% |
| With Br4Pd(2-)*C25H30N4O2(2+); potassium carbonate In N,N-dimethyl-formamide at 120℃; for 12h; Suzuki-Miyaura reaction; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 3-butyl-1-methyl-5-[(3-methyl-1H-imidazol-3-ium-1-yl)methyl]-3H-1,2,3-triazol-1-ium ditetrafluoroborate; caesium carbonate In 1,4-dioxane Suzuki-Miyaura reaction; Inert atmosphere; Heating; | 100% |
| With potassium fluoride; palladium diacetate; tricyclohexylphosphine In tetrahydrofuran at 20℃; for 18h; Suzuki cross-coupling; | 95% |
| With 2H(1+)*Cl4Pd(2-)*2H3N; poly[N-isopropylacrylamide-co-diphenyl(4'-styryl)phosphine]; sodium carbonate In water at 100℃; for 9h; Suzuki-Miyaura reaction; | 93% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium hydroxide; poly(ethylene glycol)-400; palladium diacetate In water at 80℃; for 15h; Stille coupling; | 100% |
| With 1,1-Dimethylurea; ammonium chloride; LACTOSE; tris(dibenzylideneacetone)dipalladium(0) chloroform complex; triphenyl-arsane at 90℃; for 6h; Stille coupling; | 100% |
| With tris(dibenzylideneacetone)dipalladium(0) chloroform complex; cesium fluoride; 3-tert-butyl-5-methyl-1-(2-(diphenylphosphino)phenyl)-1H-pyrazole In toluene at 60℃; for 10h; Stille coupling; Inert atmosphere; | 96% |
-
-
106-43-4
para-chlorotoluene

-
-
596-38-3
9-phenyl-9H-xanthen-9-ol

-
A
-
90-47-1
xanth-9-one

-
B
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; tricyclohexylphosphine In o-xylene for 4h; Heating; | A n/a B 100% |

-
-
106-38-7
para-bromotoluene

-
-
596-38-3
9-phenyl-9H-xanthen-9-ol

-
A
-
90-47-1
xanth-9-one

-
B
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; tricyclohexylphosphine In o-xylene for 4h; Heating; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); tetrabutylammonium triphenyldifluorosilicate; silver(l) oxide In tetrahydrofuran at 70℃; for 0.5h; Hiyama-type coupling; | 100% |
| With palladium; tetrabutyl ammonium fluoride In tetrahydrofuran at 80℃; for 48h; | 76% |

| Conditions | Yield |
|---|---|
| With cesium fluoride In dimethyl sulfoxide at 120℃; for 5h; Catalytic behavior; Stille Cross Coupling; Inert atmosphere; | 99% |
| With copper(l) iodide; triphenyl-arsane; 1-butyl-3-methylimidazolium Tetrafluoroborate; bis(benzonitrile)palladium(II) dichloride at 80℃; for 18h; Stille coupling; | 95% |
| With sodium acetate; nanocristalline MgO-stabilized nanopalladium(0) In 1-methyl-pyrrolidin-2-one at 100℃; for 2.5h; Stille cross-coupling; | 92% |

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium (0); N,N′-bis(2,6-diisopropylphenyl)imidazol-2-ylidene hydrochloride In tetrahydrofuran; 1,4-dioxane at 80℃; for 3h; Phenylation; | 99% |
| With [1,3-bis(2,4,6-trimethylphenyl)imidazol]-2-ylidene; Ni(1,5-bis-cyclooctadiene)2 In various solvent(s) Kumada-Corriu cross-coupling reaction; | 88% |
| With N-heterocyclic carbene-based nickel(II) complex In tetrahydrofuran at 20℃; for 12h; Kumada reaction; | 83% |
| With 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether at 20℃; for 0.333333h; | 97 % Chromat. |

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium (0); N,N′-bis(2,6-diisopropylphenyl)imidazol-2-ylidene hydrochloride In tetrahydrofuran; 1,4-dioxane at 80℃; for 1h; Phenylation; | 99% |
| With 1-[2-(diphenylphosphino)phenyl]ethanol; bis(acetylacetonate)nickel(II) In diethyl ether at 20℃; for 3h; | 91 % Chromat. |

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; palladium(II) acetylacetonate; 3,9-bis(2,4-tBu-PhO)tetraoxa-3,9-diphosphaspiro[5.5]undecane In xylene at 80℃; for 4h; Hiyama coupling; | 99% |
| With tetrabutyl ammonium fluoride; tris-(o-tolyl)phosphine; dichloro bis(acetonitrile) palladium(II) at 80℃; for 0.666667h; Hiyama cross-coupling reaction; | 99% |
| With tetrabutyl ammonium fluoride In para-xylene at 120℃; Hiyama coupling; Continuous flow system; | 99% |

| Conditions | Yield |
|---|---|
| With 4-di-tert-butylphosphanyl-1,5-diphenyl-1H-[1,2,3]triazole; potassium phosphate; bis(dibenzylideneacetone)-palladium(0) In toluene at 100℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With 4-di-tert-butylphosphanyl-1,5-diphenyl-1H-[1,2,3]triazole; potassium phosphate; bis(dibenzylideneacetone)-palladium(0) In toluene at 100℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With potassium phosphate; POPd1 In tetrahydrofuran for 2h; Product distribution; Further Variations:; Reagents; Catalysts; Solvents; reaction times; Suzuki coupling; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With C34H52Cl2N2Pd; potassium carbonate; Trimethylacetic acid In N,N-dimethyl acetamide at 120℃; for 16h; Catalytic behavior; Kinetics; Reagent/catalyst; Concentration; | 99% |
| With potassium tert-butylate; vasicine at 20 - 110℃; for 48h; Schlenk technique; Inert atmosphere; | 94% |
| With 1,10-Phenanthroline; potassium tert-butylate at 100℃; for 18h; Inert atmosphere; Sealed tube; | 89% |

| Conditions | Yield |
|---|---|
| With C35H31BrN4NiP In tetrahydrofuran at 25℃; for 24h; Schlenk technique; Inert atmosphere; | 99% |
| With supramolecular ensemble of HP-T with Au-Fe3O4 nanoparticles; air In tetrahydrofuran; water at 20℃; for 3h; Kumada Cross-Coupling; Irradiation; | 60% |
| 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In tetrahydrofuran at 20℃; for 24h; | 92 % Chromat. |
-
-
1015-49-2
N,N-diethyl phenyl O-sulfamate

-
-
4294-57-9
para-methylphenylmagnesium bromide

-
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| NiClCpIMes In diethyl ether at 40℃; for 24h; Grignard cross-coupling reaction; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 100℃; for 4h; Suzuki Coupling; High pressure; Green chemistry; | 99% |
| With potassium phosphate; nanocristalline MgO-stabilized nanopalladium(0) In water at 20℃; Suzuki cross-coupling; | 97% |
-
-
143-66-8
sodium tetraphenyl borate

-
-
6293-68-1
bis(4-methylphenyl)iodonium bromide

-
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| palladium dichloride In water for 0.00555556h; Suzuki coupling; microwave irradiation; | 99% |
| for 0.05h; Suzuki coupling; microwave irradiation; | 95% |
| In water at 100℃; for 0.05h; Suzuki coupling reaction; microwave irradiation; | 93% |
| In water at 100℃; for 0.05h; Suzuki coupling; microwave irradiation; | 93% |

| Conditions | Yield |
|---|---|
| amido pincer complex of nickel chloride catalyst In toluene at 20℃; for 12h; Kumada cross-coupling reaction; | 99% |
| [Ni-(o-MeC6H4){2-{OC(Ph)2CH2}-6-(3,5-Me2C3HN2)C5H3N}] In tetrahydrofuran; toluene for 12h; Kumada reaction; Heating; | 99% |
| With supramolecular ensemble of HP-T with Au-Fe3O4 nanoparticles; air In tetrahydrofuran; water at 20℃; for 0.333333h; Kumada Cross-Coupling; Irradiation; | 87% |

| Conditions | Yield |
|---|---|
| amido pincer complex of nickel chloride catalyst In tetrahydrofuran at 20℃; for 24h; Kumada cross-coupling reaction; | 99% |
| With ((C6H4)(OPPh2)(NCHPhPPh2))NiCl In tetrahydrofuran at 25℃; for 24h; Kumada cross-coupling; Inert atmosphere; | 99% |
| With Fe2(OtBu)6; sodium t-butanolate; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In tetrahydrofuran at 80℃; for 16h; Catalytic behavior; Reagent/catalyst; Time; Kumada Cross-Coupling; Glovebox; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| [Pd(Cl){2-{OC(Ph)=CH}-6-(3,5-Me2C3HN2)C5H3N}] In tetrahydrofuran; toluene for 6h; Kumada reaction; Heating; | 99% |
| With C16H16Cl2CoN8(1+)*F6P(1-) In tetrahydrofuran at 20℃; for 2h; Kumada-Corriu cross-coupling; | 96% |
| With [1,3-bis(2,6-diisopropylphenyl)-imidazolium][Ni(PPh3)Cl3] In tetrahydrofuran at 30℃; for 1h; Kumada Cross-Coupling; Schlenk technique; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: para-chlorotoluene With Ni(PPh3)(1,3-di-tert-butylimidazol-2-ylidene)Br2 In tetrahydrofuran at 0℃; for 0.0333333h; Inert atmosphere; Schlenk technique; Stage #2: phenylmagnesium bromide In tetrahydrofuran at 0 - 25℃; for 3h; Reagent/catalyst; Inert atmosphere; Schlenk technique; | 99% |
| With C46H55ClFeN3Pd; lithium chloride In tetrahydrofuran at 60℃; for 12h; Kumada coupling reaction; Inert atmosphere; | 93% |
| With Pd/Al(OH)3 In toluene at 140℃; for 36h; Kumada Cross-Coupling; Inert atmosphere; | 93% |
-
-
108-86-1
bromobenzene

-
-
4294-57-9
para-methylphenylmagnesium bromide

-
A
-
613-33-2
(4,4'-dimethyl-1,1'-biphenyl)

-
B
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| With CoF2*4H2O; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In tetrahydrofuran at 60℃; for 3h; Inert atmosphere; | A 7% B 99% |
| phenylnickel N-Bu-6-[1-(2,6-(Me)2PhN)ethyl]picolinamide In tetrahydrofuran; toluene at 20℃; for 24h; | |
| With (N-(dimethylaminoethyl)-2-diphenylphosphinoaniline(-1H))NiCl In tetrahydrofuran at 25℃; for 12h; Kumada coupling reaction; Inert atmosphere; | A 32 %Chromat. B 63 %Chromat. |

-
-
108-90-7
chlorobenzene

-
-
4294-57-9
para-methylphenylmagnesium bromide

-
A
-
613-33-2
(4,4'-dimethyl-1,1'-biphenyl)

-
B
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| With C68H72Cl2N6NiP2 In diethyl ether at 25℃; for 12h; Kumada coupling reaction; Inert atmosphere; | A n/a B 99% |
| With FeF3*H2O; tricyclohexylphosphine In tetrahydrofuran at 20 - 60℃; for 24h; Inert atmosphere; | A 4% B 5% |
| (1-naphthyl)Ni N-Bu-6-[1-(2,6-(i-Pr)2PhN)ethyl]picolinamide In tetrahydrofuran; toluene at 20℃; for 24h; |


| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In water; toluene at 100℃; for 20h; Solvent; Reagent/catalyst; Hiyama Coupling; | 99% |
| With tetrabutyl ammonium fluoride In para-xylene at 50℃; for 20h; Hiyama coupling; | 98% |
| With palladium; tetrabutyl ammonium fluoride at 80℃; for 6h; | 98% |

| Conditions | Yield |
|---|---|
| With Ni(Cl){2-(Ph2P)C6H4NCH(Ph)P(O)Ph2} In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 25℃; for 24h; Negishi coupling reaction; Inert atmosphere; | 99% |
| With C21H18N8Ni2O(2+)*2F6P(1-) In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 80℃; for 2h; Negishi coupling reaction; | 97% |
-
-
644-08-6
4-Methylbiphenyl

| Conditions | Yield |
|---|---|
| With hydrogen; water-d2; palladium on activated charcoal at 110℃; under 760.051 Torr; for 24h; | 99% |
4-Phenyltoluene Chemical Properties
4-methyldiphenyl(644-08-6) is also named as 4-Phenyltoluene;1’-Biphenyl,4-methyl-1;4-methyl-1’-biphenyl;4-methyl-bipheny;Biphenyl, 4-methyl-;p-Methylbiphenyl;p-Methyldiphenyl;p-Phenyltoluene,and so on.4-methyldiphenyl(644-08-6) is usually white crystalline powder.
CAS: 644-08-6
Molecular Formula: C13H12
Molecular Weight: 168.23
Molecular structure: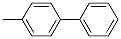
EINECS: 211-409-0
Melting point: 44-47 °C(lit.)
Boiling point: 267-268 °C(lit.)
Density: 1,015 g/cm3
FEMA: 3186
Flash point: >230 °F
BRN: 1904468
Index of Refraction: 1.564
Molar Refractivity: 55.66 cm3
Molar Volume: 170.9 cm3
Polarizability: 22.06 10-24cm3
Surface Tension: 35.8 dyne/cm
Enthalpy of Vaporization: 48.55 kJ/mol
Vapour Pressure: 0.0132 mmHg at 25°C
4-Phenyltoluene Uses
4-Phenyltoluene Toxicity Data With Reference
| 1. | orl-rat LD50:2570 mg/kg | FCTXAV Food and Cosmetics Toxicology. 13 (1975),487. |
4-Phenyltoluene Consensus Reports
4-Phenyltoluene Safety Profile
Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating vapors.
Safety Statements: 24/25
WGK Germany: 2
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View