-
Name
5-ACETYLVALERIC ACID
- EINECS 221-512-2
- CAS No. 3128-07-2
- Article Data103
- CAS DataBase
- Density 1.059 g/mL at 25 °C(lit.)
- Solubility Soluble in water
- Melting Point 35-37 °C(lit.)
- Formula C7H12O3
- Boiling Point 299.3 °C at 760 mmHg
- Molecular Weight 144.17
- Flash Point 149.1 °C
- Transport Information UN 3261 8/PG 2
- Appearance
- Safety 26-36/37/39-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms 5-Acetylvalericacid;6-Ketoheptanoic acid;6-Oxoenanthic acid;e-Ketoheptanoic acid;Acetovaleric acid;NSC 167591;
- PSA 54.37000
- LogP 1.22040
Synthetic route

| Conditions | Yield |
|---|---|
| With tropylium tetrafluoroborate; water In neat (no solvent) at 100℃; for 16h; Solvent; Temperature; Reagent/catalyst; Claisen Condensation; Inert atmosphere; | 99% |
| With iron(III) chloride; water at 80℃; for 16h; Neat (no solvent); | 95% |
| With indium(III) triflate; 2,6-di-tert-butyl-pyridine; water at 80℃; for 24h; Neat (no solvent); Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With oxygen; acetic acid In water at 60℃; under 750.075 Torr; for 8h; | 98% |
| With oxygen; H7*10H2O In water at 60℃; under 750.06 Torr; for 8h; | 97% |
| With oxygen; H6[PMo9V3O40]*11H2O In methanol at 60℃; under 750.06 Torr; for 24h; Product distribution; Kinetics; Further Variations:; Catalysts; Reagents; Solvents; Temperatures; reaction times; | 97% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; 12-tungstophosphoric acid/Al/C In tert-butyl alcohol at 60℃; for 24h; | 96% |
| With tris(cetylpyridinium) 12-tungstophosphate; dihydrogen peroxide In tert-butyl alcohol for 24h; Heating; | 90% |
| With tert.-butylhydroperoxide; indium(III) chloride In water at 90℃; for 8.2h; | 84% |

| Conditions | Yield |
|---|---|
| With tris(cetylpyridinium) 12-tungstophosphate; dihydrogen peroxide In tert-butyl alcohol for 24h; Heating; | 95% |
| With potassium permanganate; acetone at 0℃; | |
| With dihydrogen peroxide In water; tert-butyl alcohol at 80℃; for 24h; | 91 %Chromat. |
-
-
6296-84-0, 19534-08-8, 52718-65-7, 96455-86-6, 105928-96-9, 105929-88-2, 108392-44-5, 119007-94-2, 133964-50-8
trans-1-methylcyclohexane-1,2-diol

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium tungstate; phosphoric acid; dihydrogen peroxide at 90℃; for 5h; | 93% |
| With hydrogenchloride; sodium tungstate; phosphoric acid; dihydrogen peroxide at 90℃; for 5h; other 1,2-diols, other catalysts; | 93% |
| With potassium carbonate at 20℃; for 3.3h; electrolysis: nickel(III) oxide hydroxide electrode, 0.3 A; | 70% |
-
-
30964-00-2
hept-6-ynoic acid

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With [RhCl2(p-cymene)]2; water at 20℃; for 12h; | 88% |
| With platinum catalyst In water at 37℃; for 96h; | 58% |

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With iron(III) chloride In dichloromethane at 20℃; for 0.0833333h; Inert atmosphere; Green chemistry; | 75% |
-
-
19980-35-9
1-trimethylsilyloxy-2-methyl-1-cyclohexene

-
A
-
3128-07-2
6-oxoheptanoic acid

-
B
-
96304-02-8, 96304-40-4, 144178-58-5, 3476-78-6
2-hydroxy-2-methylcyclohexanone

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; titanium silicate for 24h; Heating; | A 73% B 6% |

| Conditions | Yield |
|---|---|
| With ozone In neat (no solvent) at 20℃; for 5h; UV-irradiation; | A 70% B 10% |

| Conditions | Yield |
|---|---|
| With dodecyltrimethylammonium phosphotungstate; water; dihydrogen peroxide In toluene at 80℃; for 12h; chemoselective reaction; | 69% |
| With dihydrogen peroxide In water; tert-butyl alcohol at 80℃; for 24h; |
-
-
6296-84-0, 19534-08-8, 52718-65-7, 96455-86-6, 105928-96-9, 108392-44-5, 119007-94-2, 133964-50-8, 105929-88-2
(1R,2R)-1-methylcyclohexane-1,2-diol

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; bis(acetylacetonato)dioxidomolybdenum(VI) In chlorobenzene at 60℃; for 24h; | 62% |
-
-
65700-06-3
percarbonate de O,O-tert-butyle et O-isopropyle

-
-
107-92-6
butyric acid

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| at 130℃; for 2.5h; | 54% |
-
-
583-59-5
2-Methylcyclohexanol

-
A
-
3128-07-2
6-oxoheptanoic acid

-
B
-
626-70-0
2-methylhexane-1,6-dioic acid

| Conditions | Yield |
|---|---|
| With oxygen; sodium nitrite In trifluoroacetic acid at 0 - 20℃; for 5h; | A 52% B 14% |

| Conditions | Yield |
|---|---|
| With sulfuric acid | 24% |
| With chromium(VI) oxide; sulfuric acid at 30℃; |
-
-
1196-73-2
1-acetoxy-2-methylcyclohexene

-
A
-
3128-07-2
6-oxoheptanoic acid

-
B
-
80594-87-2
2-methyl-2-nitrocyclohexan-1-one

| Conditions | Yield |
|---|---|
| With nitric acid; acetic anhydride at 15 - 22℃; for 2h; | A 17.3% B 21% |
-
-
931-88-4, 931-87-3
(Z)-Cyclooctene

-
A
-
124-04-9
Adipic acid

-
B
-
111-16-0
heptanedioic acid

-
C
-
505-48-6
octane-1,8-dioic acid

-
D
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioctylammonium tetrakis(oxodiperoxotungsto)phos In water at 85℃; for 7h; Further byproducts given; | A 7% B 2.6% C 24 % Chromat. D 4% |

-
-
591-49-1
1-methylcyclohex-1-ene

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
124-04-9
Adipic acid

-
C
-
3128-07-2
6-oxoheptanoic acid

-
D
-
3128-06-1
5-ketohexanoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioctylammonium tetrakis(oxodiperoxotungsto)phos In water at 85℃; for 6h; Yield given; Further byproducts given; | A 0.4% B 0.3% C n/a D 0.5% |

-
-
1071-71-2
ethyl 6-chloro-6-oxohexanoate

-
-
996-82-7
sodium diethylmalonate

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With toluene Kochen des Reaktionsprodukts mit 20prozentig. HCl; |
-
-
32591-08-5
1-ethynyl-2-methyl-cyclohexene

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| Ozonolyse; |
-
-
925-90-6
ethylmagnesium bromide

-
-
611-10-9
2-ethoxycarbonyl-1-cyclopentanone

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With diethyl ether Erwaermen des Reaktionsgemisches mit Acetylchlorid und Erhitzen des Reaktionsprodukts mit wss. Natriumcarbonat-Loesung; |

| Conditions | Yield |
|---|---|
| Verseifung; |

| Conditions | Yield |
|---|---|
| (i) H2, Pd-C, (ii) KOH, MeOH, (iii) H2SO4, CrO3; Multistep reaction; |
-
-
90124-78-0
diethyl 2-acetylhexanedioate

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
124-38-9
carbon dioxide

-
-
57558-50-6
2-(4-chlorobutyl)-2-methyl[1,3]dioxolane

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| (i) Mg, THF, (ii) /BRN= 1900390/, Et2O, (iii) aq. HCl; Multistep reaction; |
-
-
73185-97-4
(E)-2-Diethylamino-7-oxo-oct-2-enenitrile

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
66030-48-6
1-Methyl-2-methylselanyl-cyclohexene

-
-
3128-07-2
6-oxoheptanoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In tetrahydrofuran |
-
-
64-18-6
formic acid

-
-
109-49-9
1-hexen-5-one

-
A
-
3128-07-2
6-oxoheptanoic acid

-
B
-
54248-02-1
α-methyl-δ-oxocaproic acid

| Conditions | Yield |
|---|---|
| With carbon monoxide; palladium on activated charcoal; 1,4-di(diphenylphosphino)-butane In 1,2-dimethoxyethane at 150℃; under 5168 Torr; for 24h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
591-49-1
1-methylcyclohex-1-ene

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
3128-07-2
6-oxoheptanoic acid

-
C
-
19480-04-7
6-oxoheptanal

-
D
-
591-78-6
n-hexan-2-one

-
E
-
3128-06-1
5-ketohexanoic acid

-
F
-
111-30-8
Glutaraldehyde

| Conditions | Yield |
|---|---|
| With ozone at 30℃; Mechanism; Product distribution; multistep reaction; |

-
-
23381-92-2
2-methyl-2-decene

-
A
-
3128-07-2
6-oxoheptanoic acid

-
B
-
96304-02-8, 96304-40-4, 144178-58-5, 3476-78-6
2-hydroxy-2-methylcyclohexanone

| Conditions | Yield |
|---|---|
| With cobalt(II); oxygen; 2-Methylcyclohexanone |

| Conditions | Yield |
|---|---|
| iron(III) chloride | 100% |
| With sulfuric acid In 1,2-dichloro-ethane Fisher esterification; | 100% |
| With sulfuric acid In 1,2-dichloro-ethane for 12h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 6-oxoheptanoic acid With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetone at 23℃; for 0.25h; Stage #2: methyl iodide In acetone for 1h; | 95% |

| Conditions | Yield |
|---|---|
| With phenylsilane; C9H8FeN2O8S2 In neat (no solvent) at 100℃; for 20h; Schlenk technique; Inert atmosphere; Glovebox; Irradiation; | 92% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 96h; Ambient temperature; | 90% |
-
-
3128-07-2
6-oxoheptanoic acid


| Conditions | Yield |
|---|---|
| With 4-pyrrolidin-1-ylpyridine; dicyclohexyl-carbodiimide In dichloromethane | 90% |

| Conditions | Yield |
|---|---|
| With phenylsilane; C9H8FeN2O8S2 In neat (no solvent) at 100℃; for 20h; Schlenk technique; Inert atmosphere; Glovebox; Irradiation; | 90% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
538-75-0
dicyclohexyl-carbodiimide

-
-
107399-01-9, 212783-03-4, 107711-42-2
4-(tri-n-butylstannyl)-but-3-en-1-ol

-
-
134311-32-3
4-(tri-n-butylstannyl)-3(E)-butenyl 6-oxoheptanoate

| Conditions | Yield |
|---|---|
| In dichloromethane catalyst: (dimethylamino)pyridine, stirred at room temp. for 2 days; filtrated, concd., chromy. (silica gel, hexane/EtOAc), elem. anal.; | 89% |


| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; isobutyl chloroformate In tetrahydrofuran for 8h; Ambient temperature; | 87% |
| With dicyclohexyl-carbodiimide In tetrahydrofuran | 70% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
455-14-1
4-trifluoromethylphenylamine

-
-
84417-40-3
6-oxo-N-<4-(trifluoromethyl)phenyl>heptanamide

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; isobutyl chloroformate In tetrahydrofuran | 86% |
| With 4-methyl-morpholine; isobutyl chloroformate 1.) THF, -15 deg C, 15 min, 2.) THF, RT, overnight; Yield given. Multistep reaction; |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
13175-27-4
heptane-1,6-diol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; | 85% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 81% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 25℃; Inert atmosphere; | 70% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; Schlenk technique; | 35% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
764658-26-6
4-butoxy-benzoic acid N-(4-methoxy-phenyl)-hydrazide

| Conditions | Yield |
|---|---|
| With acetic acid at 80℃; for 2h; | 84% |
| With acetic acid |

| Conditions | Yield |
|---|---|
| With O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 0.166667h; | 84% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
2450-71-7
Propargylamine

-
-
280577-92-6
6-oxo-N-(prop-2-yn-1-yl)heptanamide

| Conditions | Yield |
|---|---|
| Stage #1: 6-oxoheptanoic acid With 4-methyl-morpholine; isobutyl chloroformate In tetrahydrofuran at 0℃; for 0.25h; Substitution; Stage #2: Propargylamine In tetrahydrofuran at 20℃; for 0.5h; Substitution; | 80% |
| With 4-methyl-morpholine In tetrahydrofuran at 0 - 20℃; for 0.5h; | 80% |
| Stage #1: 6-oxoheptanoic acid With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane for 0.0333333h; Stage #2: With benzotriazol-1-ol In dichloromethane for 0.0333333h; Stage #3: Propargylamine In dichloromethane at 20℃; for 16h; | 52% |

| Conditions | Yield |
|---|---|
| Stage #1: 6-oxoheptanoic acid With thionyl chloride In N,N-dimethyl-formamide at 30℃; for 1h; Stage #2: 2-Phenoxyethanol In pentane at 20℃; for 1h; | 80% |

| Conditions | Yield |
|---|---|
| In benzene at 50℃; for 1h; | 79.7% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
25080-63-1
5-(3-methyl-3H-diazirin-3-yl)pentanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 6-oxoheptanoic acid With ammonia; hydroxylamine-O-sulfonic acid at -75 - 20℃; for 12h; Sealed tube; Stage #2: With potassium tert-butylate at -75 - 20℃; for 2h; Sealed tube; | 79% |
| Multi-step reaction with 2 steps 1: hydroxylamine-O-sulfonic acid / ammonia / 12 h / -78 - 20 °C / Sealed tube; liquid NH3 2: potassium hydroxide / ammonia / 2 h / -78 - 20 °C / Sealed tube; liquid NH3 View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In hexanes; dimethyl sulfoxide at 0 - 20℃; for 1h; Wittig Reaction; Stage #2: 6-oxoheptanoic acid With n-butyllithium In tetrahydrofuran; hexanes; dimethyl sulfoxide at 20℃; for 48h; Stage #3: With hydrogenchloride; water more than 3 stages; | 78% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
5212-67-9
6-methyl-hept-6-enoic acid

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In hexane; dimethyl sulfoxide at 20℃; for 1h; Stage #2: 6-oxoheptanoic acid With n-butyllithium In tetrahydrofuran; hexane; dimethyl sulfoxide at 20℃; for 48h; Wittig olefination; Further stages.; | 78% |
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In hexanes; dimethyl sulfoxide at 0 - 20℃; for 1h; Stage #2: 6-oxoheptanoic acid In tetrahydrofuran; hexanes; dimethyl sulfoxide at 40℃; for 48h; Stage #3: With hydrogenchloride; water In tetrahydrofuran; hexanes; dimethyl sulfoxide | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: sodium cyanide; 6-oxoheptanoic acid With sodium carbonate; hydrazinium sulfate In water for 44h; Stage #2: With hydrogenchloride In water Stage #3: With bromine In water for 1h; | 78% |
-
-
3128-07-2
6-oxoheptanoic acid

-
-
64-17-5
ethanol

-
-
50709-33-6
2-bromophenylhydrazine hydrochloride

-
-
1232860-64-8
ethyl 4-(7-bromo-2-methyl-1H-indol-3-yl)butanoate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 100℃; for 1.5h; Fischer Indole Synthesis; | 76% |
| Stage #1: 6-oxoheptanoic acid; 2-bromophenylhydrazine hydrochloride In ethanol at 50℃; for 0.666667h; Stage #2: ethanol With sulfuric acid for 16h; Reflux; | 15 g |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; tetrabutylammomium bromide In acetone at 120℃; under 33753.4 Torr; for 0.5h; Inert atmosphere; Microwave irradiation; | 75% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; for 2h; | 74% |
6-Oxoheptanoic acid Specification
The Heptanoic acid, 6-oxo- with CAS registry number of 3128-07-2 is also known as 5-Acetylvalericacid. The IUPAC name is 6-Oxoheptanoic acid. It belongs to product categories of C7; Carbonyl Compounds; Carboxylic Acids. Its EINECS registry number is 221-512-2. In addition, the formula is C7H12O3 and the molecular weight is 144.17. This chemical may destroy living tissue on contact.
Physical properties about Heptanoic acid, 6-oxo- are: (1)ACD/LogP: 0.17; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.7; (4)ACD/LogD (pH 7.4): -2.5; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 3.96; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 3; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 5; (12)Index of Refraction: 1.443; (13)Molar Refractivity: 36.07 cm3; (14)Molar Volume: 135.8 cm3; (15)Surface Tension: 38.1 dyne/cm; (16)Density: 1.061 g/cm3; (17)Flash Point: 149.1 °C; (18)Enthalpy of Vaporization: 59.33 kJ/mol; (19)Boiling Point: 299.3 °C at 760 mmHg; (20)Vapour Pressure: 0.000284 mmHg at 25 °C.
Preparation of Heptanoic acid, 6-oxo-: it is prepared by reaction of 2-methyl-cyclohexanone. The reaction needs reagent O2, catalyst H7*10H2O and solvent H2O at the temperature of 60 °C for 8 hours. The yield is about 97%.

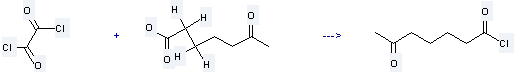
Uses of Heptanoic acid, 6-oxo-: it is used to produce 6-oxoheptanoyl chloride by reaction with oxalyl dichloride. The reaction occurs with reagent benzene at the temperature of 50 °C for 1 hour. The yield is about 79.7%.
When you are using this chemical, please be cautious about it. As a chemical, it causes burns. During using it, wear suitable protective clothing, gloves and eye/face protection. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: CC(=O)CCCCC(=O)O
2. InChI: InChI=1S/C7H12O3/c1-6(8)4-2-3-5-7(9)10/h2-5H2,1H3,(H,9,10)
3. InChIKey: IZOQMUVIDMLRDC-UHFFFAOYSA-N
Related Products
- 6-Oxoheptanoic acid
- 31281-88-6
- 31282-04-9
- 31282-95-8
- 31284-04-5
- 312-84-5
- 31284-89-6
- 31284-96-5
- 3128-52-7
- 31286-88-1
- 3128-83-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View