-
Name
Androstenedione
- EINECS 200-554-5
- CAS No. 63-05-8
- Article Data201
- CAS DataBase
- Density 1.11 g/cm3
- Solubility 57.28mg/L(25 oC)
- Melting Point 170-171 °C(lit.)
- Formula C19H26O2
- Boiling Point 431.4 °C at 760 mmHg
- Molecular Weight 286.414
- Flash Point 161.1 °C
- Transport Information
- Appearance white to off-white solid
- Safety 22-24/25-36
- Risk Codes 40
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 17-Ketotestosterone;3,17-Dioxoandrost-4-ene;4-Androstene-3,17-dione;Androstenedione;Fecundin;NSC 9563;SKF 2170;D4-Androstene-3,17-dione;Androstenedione;4-Androstenedione;
- PSA 34.14000
- LogP 4.08740
Synthetic route

| Conditions | Yield |
|---|---|
| With Jones reagent In acetone at 0℃; Jones Oxidation; | 100% |
| With oxygen; nitrosonium tetrafluoroborate In dichloromethane at 20℃; for 4h; | 99% |
| With N-chloro-succinimide; dodecyl methyl sulfide; triethylamine In toluene at -40℃; for 16h; Corey-Kim oxidation; | 97% |
-
-
25375-38-6
3-hydroxy-5-androsten-17-one

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; ferric nitrate In ethyl acetate at 0℃; for 65h; Reagent/catalyst; | 100% |

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 20℃; Electrochemical reaction; | 99% |
| With tetrapropylammonium perruthennate; 4 A molecular sieve; 4-methylmorpholine N-oxide In dichloromethane; acetonitrile for 0.75h; | 95% |
| With chromium(VI) oxide; acetic acid |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 40℃; for 2h; pH=3; Temperature; Green chemistry; | 98.6% |
| With sulfuric acid; water; silica gel In toluene for 2h; | 92% |
| Multi-step reaction with 4 steps 1: hydrogenchloride / water; acetone / pH 1-6 2: potassium tert-butylate / tetrahydrofuran / Inert atmosphere; Reflux 3: tetrahydrofuran / Acidic conditions 4: sodium hydrogencarbonate; ruthenium trichloride; Oxone / ethyl acetate; acetonitrile; water / -20 - 30 °C View Scheme |

| Conditions | Yield |
|---|---|
| With calcium hypochlorite; acetic acid In ethyl acetate at 30℃; for 1h; Temperature; Green chemistry; | 98.5% |
-
-
81176-75-2
(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[α]phenanthrene-3,17-diol

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With tetrapropylammonium perruthennate; 4 A molecular sieve; 4-methylmorpholine N-oxide In dichloromethane for 0.5h; | 98% |
| With 9-fluorenone; oxygen In dimethyl sulfoxide at 20℃; Irradiation; | 72% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol; water Ambient temperature; | 97% |
| With sodium hydroxide In methanol; water at 25℃; | |
| With sodium hydroxide; 3-oxo-Δ5-steroid isomerase In methanol; water Rate constant; pH 5.0 to 5.1; |
-
-
162657-73-0
3-cycloethylenedithio-17-cycloethylenedioxy-androst-4-ene

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) sulfate In benzene at 80℃; for 5h; | 90% |

| Conditions | Yield |
|---|---|
| aluminum oxide; copper In toluene at 60℃; | A 12% B 88% |

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 20℃; Electrochemical reaction; | 86% |
| With ammonium hydroxide; iodine In water; acetonitrile at 50℃; for 24h; | 78% |
| With manganese(IV) oxide In chloroform for 4h; Heating; | 67.6% |
-
-
152-58-9, 601-05-8, 3614-16-2, 122405-55-4
17,21-dihydroxy-pregn-3,20-dione

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With sodium methylate In 1,4-dioxane for 3h; Heating; | 86% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; iron pentacarbonyl In methanol; water at 50 - 60℃; for 16h; | 85% |
| With thallium(III) nitrate trihydrate In diethylene glycol dimethyl ether for 24h; Ambient temperature; | 29% |
| With hydrogen; silica gel; copper In toluene at 60℃; under 760 Torr; for 5h; |

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| IrH5(P-(i-Pr)3)2 In various solvent(s) at 100℃; for 28h; | 83% |

| Conditions | Yield |
|---|---|
| With <(C4Ph4COHOCC4Ph4)(μ-H)><(CO)4Ru2>; acetone at 56℃; for 24h; | 81% |
| Behandeln mit verschiedenen Dehydrierungsmitteln; | |
| Einwirkung von Flavobacterium-Arten; |

| Conditions | Yield |
|---|---|
| at 25℃; for 25h; electrolysis: nickel net anode, cylindrical stainless steel cathode; electrolyte: 0.01M KOH/t-butanol - water (1:1); | 80% |
-
-
103708-01-6
3-<2,3,5,6-tetrafluoro-4-(trifluoromethyl)phenoxy>androsta-3,4-diene-17-one

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrahydrofuran at 60℃; for 12h; | 80% |
| With sulfuric acid In tetrahydrofuran at 60℃; for 12h; | 80% |
-
-
76183-49-8
3-(m-chlorobenzoyloxy)androsta-3,5-dien-17-one

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 1h; Heating; | 74% |
-
-
57-88-5
cholesterol

-
A
-
63-05-8
Androstenedione

-
B
-
897-06-3
Androsta-1,4-diene-3,17-dione

-
C
-
40736-33-2
(20S)-20-hydroxymethylpregn-4-en-3-one

-
D
-
80981-37-9
24-norchol-4-ene-3,22-dione

-
E
-
80981-38-0
24-norchola-1,4-diene-3,22-dione

-
F
-
35525-27-0
(20S)-20-(hydroxymethyl)pregna-1,4-dien-3-one

| Conditions | Yield |
|---|---|
| Product distribution; degradation by the Mycobacterium mutant CCM 3528; | A n/a B 0.20 g C n/a D 0.36 g E 1.21 g F 71% |

-
-
51154-62-2
(8S,9S,10R,13S,14S,Z)-17-ethylidene-10,13-dimethyl-1,2,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-3H-cyclopenta[a]phenanthren-3-one

-
A
-
63-05-8
Androstenedione

-
B
-
68-96-2
17-hydroxyprogesterone

-
C
-
1662-06-2
17alpha,20beta-Dihydroxypregn-4-en-3-one

| Conditions | Yield |
|---|---|
| With ruthenium trichloride; Oxone; sodium hydrogencarbonate In water; ethyl acetate; acetonitrile at -20 - 30℃; | A 10% B 70% C 20% |


| Conditions | Yield |
|---|---|
| With ruthenium trichloride; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane; water at 20℃; | A 10% B 70% C 20% |

-
-
110801-53-1
(8R,9S,10R,13S,14S)-10,13-Dimethyl-17-[1-tripropylsilanyloxy-eth-(Z)-ylidene]-1,2,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-cyclopenta[a]phenanthren-3-one

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate; 3-chloro-benzenecarboperoxoic acid | 68% |

-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) sulfate In toluene for 30h; Heating; | 62% |

| Conditions | Yield |
|---|---|
| With penicillium citreo-viride A.C.C.C. 0402 In acetone at 27.5℃; for 120h; Microbiological reaction; | A 8.4% B 60.9% |

| Conditions | Yield |
|---|---|
| With tetrapropylammonium perruthennate; 4 A molecular sieve; 4-methylmorpholine N-oxide In dichloromethane; acetonitrile for 0.75h; | A 60% B 20% |
-
-
315-37-7
testosterone heptanoate

-
A
-
58-22-0
testosterone

-
B
-
63-05-8
Androstenedione

-
C
-
62-99-7, 2944-87-8, 34096-63-4, 34442-08-5, 145772-79-8
6β-hydroxytestosterone

| Conditions | Yield |
|---|---|
| With Fusarium fujikuroi PTCC 5144 In ethanol; water at 26℃; for 312h; Reagent/catalyst; Microbiological reaction; | A 20% B 17% C 59% |


| Conditions | Yield |
|---|---|
| With trifluoroacetic acid; 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In fluorobenzene; dimethyl sulfoxide at 60℃; for 40h; Temperature; regioselective reaction; | 54% |
| With sodium In methanol at 20℃; for 1h; | |
| Multi-step reaction with 2 steps 1: 72percent HClO4 / ethyl acetate / 0.5 h / Ambient temperature 2: CrO3*2Py (Collins oxidant) / CH2Cl2 / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: HBr; acetic acid; bromine 2: pyridine View Scheme |
-
-
501433-34-7
5α/β-androst-3-en-17-one

-
A
-
63-05-8
Androstenedione

-
B
-
1229-12-5
androstane-3,17-dione

-
C
-
566-48-3
4-hydroxyandrost-4-ene-3,17-dione

| Conditions | Yield |
|---|---|
| Multistep reaction; | A n/a B n/a C 54% |

-
-
63-05-8
Androstenedione

-
-
122-51-0
orthoformic acid triethyl ester

-
-
972-46-3
3-ethoxyandrosta-3,5-dien-17-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In tetrahydrofuran; ethanol at 45℃; for 2h; Inert atmosphere; | 100% |
| With toluene-4-sulfonic acid In ethanol at 20 - 25℃; | 99.2% |
| Stage #1: Androstenedione; orthoformic acid triethyl ester With toluene-4-sulfonic acid In ethanol at 40℃; for 6.5h; Stage #2: With triethylamine In ethanol at 5℃; for 0.5h; Temperature; | 97% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol at 20℃; Temperature; Solvent; Reagent/catalyst; | 100% |
| With potassium borohydride In methanol |
-
-
63-05-8
Androstenedione

-
-
81176-75-2
(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[α]phenanthrene-3,17-diol

| Conditions | Yield |
|---|---|
| With lithium borohydride In diethyl ether for 2h; Product distribution; Heating; other solvents and reducing agents also investigated; | 100% |
| With lithium borohydride In diethyl ether for 2h; Heating; | 100% |
-
-
63-05-8
Androstenedione

-
-
149-73-5
trimethyl orthoformate

-
-
57144-06-6
3-methoxyandrosta-3,5-dien-17-one

| Conditions | Yield |
|---|---|
| With p-toluenesulfonic acid hydrochloride In methanol at 35 - 45℃; for 5.25h; Inert atmosphere; | 100% |
| With toluene-4-sulfonic acid In tetrahydrofuran at 30℃; for 4h; Temperature; Inert atmosphere; | 100% |
| With toluene-4-sulfonic acid In tetrahydrofuran at 5℃; for 5h; Reagent/catalyst; Solvent; Inert atmosphere; | 97.3% |
-
-
63-05-8
Androstenedione

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
77881-13-1
17β-cyano-17α-hydroxy-4-androsten-3-one

| Conditions | Yield |
|---|---|
| With sodium carbonate In methanol; water at 17 - 40℃; for 29h; pH=8 - 9; | 100% |
| With water; sodium carbonate In methanol at 38 - 42℃; for 4h; pH=8 - 9; | 100% |
| With methanol; potassium hydroxide at 50℃; for 6h; Autoclave; Large scale; | 95.9% |

| Conditions | Yield |
|---|---|
| With acetic acid In methanol at 20℃; for 2h; Inert atmosphere; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In tetrahydrofuran at 20℃; for 24h; | 99.26% |
| With palladium 10% on activated carbon; hydrogen at 20℃; under 760.051 Torr; for 23h; | 74% |
| With hydrogen; palladium on activated charcoal In ethyl acetate Ambient temperature; | |
| Multi-step reaction with 2 steps 1: 467 mg / Zn; glacial acetic acid / 0.17 h View Scheme |
-
-
63-05-8
Androstenedione

-
-
108-24-7
acetic anhydride

-
-
4968-05-2
androstane-3,5-dien-17-one-3β-ol acetate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 35 - 45℃; for 12h; | 99% |
| With toluene-4-sulfonic acid for 0.05h; microwave irradiation; | 97% |
| With toluene-4-sulfonic acid at 65℃; for 1.5h; | 76% |

| Conditions | Yield |
|---|---|
| With Gelasinospora retispora GRK002 In methanol; water at 34℃; for 144h; pH=5; Microbiological reaction; | 98% |
| With Beauveria bassiana CCTCC AF206001 In water for 24h; pH=6.0; Microbiological reaction; | 90.9% |
| mit Hilfe von Rhizopus arrhizus; |

| Conditions | Yield |
|---|---|
| With foie de boeuf; levure de boulanger; tampon phosphate In water at 37℃; for 144h; Clostridium paraputrificum ATCC No. 25780; Yields of byproduct given; | A 98% B n/a |
| With foie de boeuf; levure de boulanger; tampon phosphate In water at 37℃; for 144h; Clostridium paraputrificum ATCC No. 25780; Yields of byproduct given; | A n/a B 85% |
| With foie de boeuf; levure de boulanger; tampon phosphate In water at 37℃; for 144h; Product distribution; Clostridium paraputrificum ATCC No. 25780; composition of the culture medium, different species and influence of the steroid concentration examined; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; ethanol | 95% |
| Stage #1: acetylene With potassium hydroxide In tetrahydrofuran; ethanol at 3℃; for 2h; Inert atmosphere; Stage #2: Androstenedione In tetrahydrofuran; ethanol for 2h; Temperature; Solvent; Reagent/catalyst; | 109.3 g |
-
-
63-05-8
Androstenedione

-
-
122-51-0
orthoformic acid triethyl ester

-
-
57144-06-6
3-methoxyandrosta-3,5-dien-17-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In tetrahydrofuran; chloroform at 38℃; for 2h; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; | 94.27% |
-
-
1445-45-0
Trimethyl orthoacetate

-
-
63-05-8
Androstenedione

-
-
57144-06-6
3-methoxyandrosta-3,5-dien-17-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In tetrahydrofuran; methanol at 30℃; for 4h; Inert atmosphere; Industrial scale; | 94% |

| Conditions | Yield |
|---|---|
| With zygowilliopsis sp. WY7905 In aq. phosphate buffer at 30℃; for 24h; pH=8; Enzymatic reaction; stereoselective reaction; | 93% |
| With potassium borohydride In tetrahydrofuran; dichloromethane; water at -10 - -5℃; Temperature; | 93% |
| With Trichoderma virens In ethanol at 24℃; for 96h; Microbiological reaction; | 60% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 48h; Heating; | 93% |
-
-
63-05-8
Androstenedione

-
-
260352-51-0
C19H25IO2

| Conditions | Yield |
|---|---|
| Stage #1: Androstenedione With trimethylsilylazide In dichloromethane at 0℃; for 2h; Stage #2: With pyridine; iodine In dichloromethane at 0 - 20℃; | 93% |

| Conditions | Yield |
|---|---|
| With Beauveria bassiana CCTCC AF206001 In water for 48h; pH=7.0; Microbiological reaction; | 91.3% |
| (microbiological transformation); |
-
-
63-05-8
Androstenedione

| Conditions | Yield |
|---|---|
| With hydrogenchloride; deuteromethanol In water at 56℃; Temperature; Inert atmosphere; | 91% |
-
-
63-05-8
Androstenedione

-
-
86420-28-2
dehydroisoandrosterone

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; 2,2'-azobis(isobutyronitrile); diphenylstibane In toluene at 90℃; for 16h; | 90% |

| Conditions | Yield |
|---|---|
| With sodium dithionite; phase transfer catalyst; sodium hydrogencarbonate In toluene for 4h; Heating; | 88% |
| With hydrogen; (η4-1,5-cyclooctadiene)(pyridine)(tricyclohexylphosphine)iridium(I) hexafluorophosphate In dichloromethane for 4h; | 70% |
| biochem. proc. with 5α-reductase of Penicillin decumbens; | 70% |
-
-
63-05-8
Androstenedione

-
-
7191-30-2
2,2-dibutyl-1,3,2-dithiastannolane

-
A
-
63910-90-7
4-Androstene-3,17-dione 3-ethylene dithioacetal

-
B
-
10455-49-9
4-Androstene-3,17-dione 3,17-bis(ethylene dithioacetal)

| Conditions | Yield |
|---|---|
| di-n-butylbis(trifluoromethanesulfonyloxy)stannane In 1,2-dichloro-ethane at 20℃; for 4h; | A 87% B 7% |

Androst-4-ene-3,17-dione History
Andro was legal and able to be purchased over the counter and consequently it was common use in Major League Baseball throughout the 1990s by record-breaking sluggers like Mark McGwire. The supplement is banned by the World Anti-Doping Agency, and hence from the Olympic Games.
On March 12, 2004, the Anabolic Steroid Control Act of 2004 was introduced into the United States Senate. The law took effect on January 20, 2005. Surprisingly, andro was legally defined as an anabolic steroid, even though there is scant evidence that androstenedione itself is anabolic in nature.
On April 11, 2004, the United States Food and Drug Administration banned the sale of Andro.
Androstenedione is currently banned by the US military.
Androst-4-ene-3,17-dione Specification
The Androstenedione is an organic compound with the formula C19H26O2. The IUPAC name of this chemical is (8R,9S,10R,13S,14S)-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthrene-3,17-dione. With the CAS registry number 63-05-8, it is also named as 4-Androsten-3,17-dione. The product's categories are Pharmaceutical Intermediates; Steroids; Metabolites & Impurities. Besides, it is a white to off-white solid, which should be stored in a closed place. It is used for biochemical studies.
Physical properties about Androstenedione are: (1)ACD/LogP: 2.90; (2)ACD/LogD (pH 5.5): 2.9; (3)ACD/LogD (pH 7.4): 2.9; (4)ACD/BCF (pH 5.5): 93.64; (5)ACD/BCF (pH 7.4): 93.64; (6)ACD/KOC (pH 5.5): 896.96; (7)ACD/KOC (pH 7.4): 896.96; (8)#H bond acceptors: 2; (9)Polar Surface Area: 34.14 Å2; (10)Index of Refraction: 1.551; (11)Molar Refractivity: 81.71 cm3; (12)Molar Volume: 255.8 cm3; (13)Polarizability: 32.39×10-24cm3; (14)Surface Tension: 42.6 dyne/cm; (15)Density: 1.11 g/cm3; (16)Flash Point: 161.1 °C; (17)Enthalpy of Vaporization: 68.71 kJ/mol; (18)Boiling Point: 431.4 °C at 760 mmHg; (19)Vapour Pressure: 1.2E-07 mmHg at 25°C.
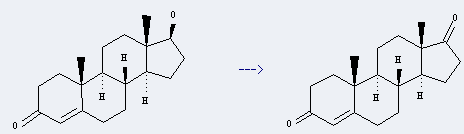
Preparation: this chemical can be prepared by 17-hydroxy-androst-4-en-3-one. This reaction will need reagent CrO3 and acetic acid.
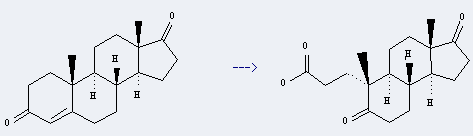
Uses of Androstenedione: it can be used to produce 5,17-dioxo-3,5-seco-A-nor-androstan-3-oic acid. It will need reagent KMnO4 and acetone.
When you are using this chemical, please be cautious about it as the following:
It has limited evidence of a carcinogenic effect. When you are using it, wear suitable protective clothing, do not breathe dust and avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C4/C=C3/CC[C@@H]2[C@H](CC[C@@]1(C(=O)CC[C@H]12)C)[C@@]3(C)CC4
(2)InChI: InChI=1/C19H26O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h11,14-16H,3-10H2,1-2H3/t14-,15-,16-,18-,19-/m0/s1
(3)InChIKey: AEMFNILZOJDQLW-QAGGRKNEBJ
(4)Std. InChI: InChI=1S/C19H26O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h11,14-16H,3-10H2,1-2H3/t14-,15-,16-,18-,19-/m0/s1
(5)Std. InChIKey: AEMFNILZOJDQLW-QAGGRKNESA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View