-
Name
Arbidol hydrochloride
- EINECS 680-680-9
- CAS No. 131707-23-8
- Article Data2
- CAS DataBase
- Density
- Solubility
- Melting Point 133-137?C
- Formula C22H26BrClN2O3S
- Boiling Point 591.8 ºC at 760 mmHg
- Molecular Weight 513.883
- Flash Point 311.7 ºC
- Transport Information
- Appearance White or off-white crystalline powder
- Safety 26
- Risk Codes 36/38
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms 1H-Indole-3-carboxylicacid, 6-bromo-4-[(dimethylamino)methyl]-5-hydroxy-1-methyl-2-[(phenylthio)methyl]-, ethyl ester, monohydrochloride(9CI);Arbidol;Arbidolhydrochloride;
- PSA 80.00000
- LogP 5.97900
Synthetic route
-
-
50-00-0
formaldehyd

-
-
124-40-3
dimethyl amine

-
-
131707-24-9
6-bromo-5-hydroxy-1-methyl-2-phenylthiomethylindole-3-carboxylic acid ethyl ester

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; dimethyl amine; 6-bromo-5-hydroxy-1-methyl-2-phenylthiomethylindole-3-carboxylic acid ethyl ester With aminosulfonic acid In water at 30 - 40℃; for 3h; Cooling with ice; Stage #2: With hydrogenchloride In water; acetone at 60℃; pH=1; | 85.6% |

-
-
131707-25-0
arbidol

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In acetone for 0.5h; Reflux; | 75% |

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: potassium carbonate; palladium diacetate; copper diacetate / N,N-dimethyl-formamide / 3 h / 80 °C 2.1: potassium carbonate / N,N-dimethyl-formamide / 6 h / 100 °C 3.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 4.1: sodium hydroxide / methanol / 2 h 4.2: 3 h 5.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 5.2: 60 °C / pH 1 View Scheme |
-
-
20862-91-3
ethyl 5-acetyloxy-2-methyl-1H-indole-3-carboxylate

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 6 h / 100 °C 2.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 3.1: sodium hydroxide / methanol / 2 h 3.2: 3 h 4.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 4.2: 60 °C / pH 1 View Scheme |
-
-
40945-79-7
1,2-dimethyl-5-acetoxy-1H-indole-3-carboxylic acid ethyl ester

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 2.1: sodium hydroxide / methanol / 2 h 2.2: 3 h 3.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 3.2: 60 °C / pH 1 View Scheme | |
| Multi-step reaction with 4 steps 1.1: [RhCl2(p-cymene)]2; N-Bromosuccinimide / water; N,N-dimethyl acetamide / 24 h / 90 °C / Inert atmosphere 2.1: potassium hydroxide; methanol / 0.25 h / 20 °C 2.2: 4 h / 20 °C 3.1: acetic acid / water / 0.25 h / 0 °C 3.2: 4 h / 80 °C 4.1: hydrogenchloride / acetone / 0.5 h / Reflux View Scheme |
-
-
100-02-7
4-nitro-phenol

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: pyridine / acetone / 4 h / 20 °C / Reflux 2.1: iron; ammonium chloride / ethanol; water / 2 h / Reflux 3.1: indium(III) bromide / dichloromethane / 3 h / Reflux 4.1: potassium carbonate; palladium diacetate; copper diacetate / N,N-dimethyl-formamide / 3 h / 80 °C 5.1: potassium carbonate / N,N-dimethyl-formamide / 6 h / 100 °C 6.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 7.1: sodium hydroxide / methanol / 2 h 7.2: 3 h 8.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 8.2: 60 °C / pH 1 View Scheme |
-
-
830-03-5
4-nitrophenol acetate

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: iron; ammonium chloride / ethanol; water / 2 h / Reflux 2.1: indium(III) bromide / dichloromethane / 3 h / Reflux 3.1: potassium carbonate; palladium diacetate; copper diacetate / N,N-dimethyl-formamide / 3 h / 80 °C 4.1: potassium carbonate / N,N-dimethyl-formamide / 6 h / 100 °C 5.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 6.1: sodium hydroxide / methanol / 2 h 6.2: 3 h 7.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 7.2: 60 °C / pH 1 View Scheme |
-
-
13871-68-6
p-aminophenyl acetate

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: indium(III) bromide / dichloromethane / 3 h / Reflux 2.1: potassium carbonate; palladium diacetate; copper diacetate / N,N-dimethyl-formamide / 3 h / 80 °C 3.1: potassium carbonate / N,N-dimethyl-formamide / 6 h / 100 °C 4.1: dibenzoyl peroxide; bromine / chloroform / 3 h / Reflux 5.1: sodium hydroxide / methanol / 2 h 5.2: 3 h 6.1: aminosulfonic acid / water / 3 h / 30 - 40 °C / Cooling with ice 6.2: 60 °C / pH 1 View Scheme |
-
-
15574-49-9
mecarbinate

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 4 h / Reflux 2.1: [RhCl2(p-cymene)]2; N-Bromosuccinimide / water; N,N-dimethyl acetamide / 24 h / 90 °C / Inert atmosphere 3.1: potassium hydroxide; methanol / 0.25 h / 20 °C 3.2: 4 h / 20 °C 4.1: acetic acid / water / 0.25 h / 0 °C 4.2: 4 h / 80 °C 5.1: hydrogenchloride / acetone / 0.5 h / Reflux View Scheme |
-
-
110543-98-1
5-acetoxy-6-bromo-2-bromomethyl-1-methylindole-3-carboxylic acid ethyl ester

-
-
131707-23-8
Arbidol hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: potassium hydroxide; methanol / 0.25 h / 20 °C 1.2: 4 h / 20 °C 2.1: acetic acid / water / 0.25 h / 0 °C 2.2: 4 h / 80 °C 3.1: hydrogenchloride / acetone / 0.5 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: Arbidol hydrochloride With sodium hydroxide In water Stage #2: maleic acid In ethyl acetate |

| Conditions | Yield |
|---|---|
| Stage #1: Arbidol hydrochloride With sodium hydroxide In water Stage #2: salicylic acid In ethyl acetate |
-
-
490-79-9
2,5-dihydroxybenzoic acid.

-
-
131707-23-8
Arbidol hydrochloride

-
-
1515677-35-6
umifenovir gentisate

| Conditions | Yield |
|---|---|
| Stage #1: Arbidol hydrochloride With sodium hydroxide In water Stage #2: 2,5-dihydroxybenzoic acid. In ethyl acetate |
-
-
110-94-1
1,5-pentanedioic acid

-
-
131707-23-8
Arbidol hydrochloride

-
-
1515677-33-4
umifenovir hydrogen glutarate

| Conditions | Yield |
|---|---|
| Stage #1: Arbidol hydrochloride With sodium hydroxide In water Stage #2: 1,5-pentanedioic acid In ethyl acetate |
Arbidol hydrochloride Chemical Properties
The Molecular Structure of Arbidol hydrochloride (CAS NO.131707-23-8):

Empirical Formula: C22H26BrClN2O3S
Molecular Weight: 513.8754
Flash Point: 311.7 °C
Enthalpy of Vaporization: 91.52 kJ/mol
Boiling Point: 591.8 °C at 760 mmHg
Vapour Pressure: 1.34E-14 mmHg at 25°C
Synonyms: 6-Bromo-4-[(dimethylamino)methyl]-5-hydroxy-1-methyl-2-[(phenylthio)methyl]-1H-indole-3-carboxylic acid ethyl ester monohydrochloride
Arbidol hydrochloride Uses
Arbidol hydrochloride (CAS NO.131707-23-8) is a medicinal agent for treating viral infections.
Arbidol hydrochloride Production
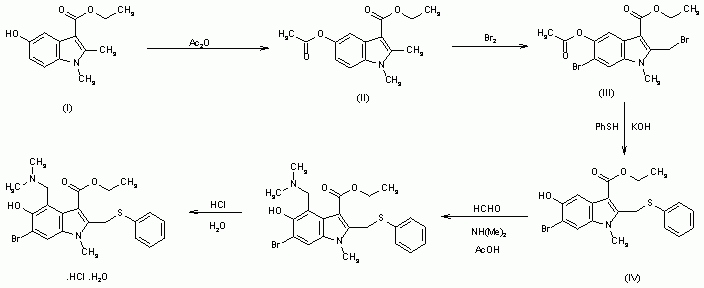
1,2-Dimethyl-5-hydroxyindole-3-acetic acid ethyl ester (I) is acetylated with acetic anhydride affording the O-acyl derivative (II) , which is brominated to the corresponding dibromide compound (III) . The reaction of (III) with thiophenol in KOH yields (IV) , which is then submitted to a conventional Mannich condensation with formaldehyde and dimethylamine in acetic acid, giving the free base of arbidol (V), which is treated with aqueous hydrochloric acid .
Arbidol hydrochloride Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | > 3gm/kg (3000mg/kg) | Drugs of the Future. Vol. 17, Pg. 1079, 1992. | |
| mouse | LD50 | oral | 340mg/kg (340mg/kg) | Drugs of the Future. Vol. 17, Pg. 1079, 1992. | |
| rat | LD50 | oral | > 3gm/kg (3000mg/kg) | Drugs of the Future. Vol. 17, Pg. 1079, 1992. |
Related Products
- Arbidol
- Arbidol hydrochloride
- 131707-24-9
- 131707-25-0
- 13171-00-1
- 13171-18-1
- 13171-21-6
- 13171-25-0
- 13171-64-7
- 13171-93-2
- 131724-43-1
- 131724-45-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View