-
Name
Benzyltrimethylammonium chloride
- EINECS 200-300-3
- CAS No. 56-93-9
- Article Data14
- CAS DataBase
- Density 1.08 g/mL at 25 °C
- Solubility 800 g/L in water
- Melting Point 236 °C (Decomposes)
- Formula C10H16ClN
- Boiling Point >135oC (Some decomp)
- Molecular Weight 185.697
- Flash Point
- Transport Information UN 2811 6.1/PG 3
- Appearance white to light yellow crystal powder
- Safety 26-37/39
- Risk Codes 22-36/38-36
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms benzyltrim-ethyl-ammonium chloride;Ammonium,benzyltrimethyl-, chloride (8CI);Benzenemethanaminium, N,N,N-trimethyl-,chloride (9CI);Benzenemethanaminium,N,N,N-trimethyl-, chloride (1:1);BTM;BTMAC 100;Hipochem Migrator J;TMBAC;Trimethylbenzylammonium chloride;Variquat B 200;
- PSA 0.00000
- LogP -1.10320
Synthetic route

| Conditions | Yield |
|---|---|
| In ethanol; acetone Alkylation; | 95% |

| Conditions | Yield |
|---|---|
| In diethyl ether; water Ambient temperature; | 94% |
| With methanol | |
| With ethanol |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol at 0℃; for 0.5h; | 73% |

| Conditions | Yield |
|---|---|
| With 1-ethyl-3-methylimidazolium bromide at 169.84℃; for 8h; | 71.3% |

-
A
-
56-93-9
benzyltrimethylammonium chloride

-
B
-
232279-44-6
Trp-Aib-Gly-Leu-NHPh

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.84℃; Equilibrium constant; complex formation; |

-
A
-
56-93-9
benzyltrimethylammonium chloride

-
B
-
271770-12-8
Trp-Aib-Gly-Leu-NH-C6H3(3,5-Me2)

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.84℃; Equilibrium constant; complex formation; |

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In chloroform Equilibrium constant; |
-
-
100-46-9
benzylamine

-
-
74-88-4
methyl iodide

-
A
-
56-93-9
benzyltrimethylammonium chloride

-
B
-
103-67-3
benzyl-methyl-amine

| Conditions | Yield |
|---|---|
| Stage #1: benzylamine; methyl iodide With 1,1,1,3',3',3'-hexafluoro-propanol In d(4)-methanol at 20℃; for 1h; Stage #2: With hydrogenchloride In water for 1h; Reagent/catalyst; Time; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetonitrile / 12 h / 50 °C 2: Dowex 22 chloride form / acetonitrile View Scheme |

| Conditions | Yield |
|---|---|
| With Dowex 22 chloride form In acetonitrile | 108 mg |
-
-
998-40-3
tributylphosphine

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
1224-56-2
tributylbenzylphosphonium chloride

| Conditions | Yield |
|---|---|
| at 100℃; for 0.166667h; Irradiation; | 100% |
-
-
14104-20-2
silver tetrafluoroborate

-
-
169697-22-7
iron(II) octaethyltetraazaporphyrin

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
141045-99-0
chloroiron(III) 2,3,7,8,12,13,17,18-octaethyl-5,10,15,20-tetraazaporphyrinate

| Conditions | Yield |
|---|---|
| In dichloromethane; toluene byproducts: Ag; inert atmosphere; addn. of AgBF4 to the Fe-complex in toluene; stirred for 30 min; filtration off of Ag (Celite), evapn. (vac.), dissoln. in CH2Cl2, addn. of PhCH2N(CH3)3Cl, stirring (15 min);; filtration (Celite), partial evapn. (vac.), crystn. (hexane/C6H6, slow evapn.);; | 100% |


-
-
56-93-9
benzyltrimethylammonium chloride

-
-
189502-86-1
C6H5CH2N(CH3)3(1+)*(CO)2(C3H5)MoC2B9H9(C6H5)2(1-)=[C6H5CH2N(CH3)3][(CO)2(C3H5)MoC2B9H9(C6H5)2]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran refluxing equimolar amts. of carborane and No-complex, solvent removal (vac.), dissoln. in CH2Cl2, addn. of BzNMe3Cl; trituration with pentane; | 99% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
84864-61-9
rac-Potassium mandelate

| Conditions | Yield |
|---|---|
| In water at 60℃; for 6h; | 99% |
-
-
6161-87-1
2-(2,4,6-trichloro-phenoxy)-ethanol

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
13001-29-1
2-(2,4,6-trichlorophenoxy)-ethyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In tetrachloromethane; water | 98% |
-
-
10213-10-2
sodium tungstate (VI) dihydrate

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide In toluene | 97.6% |


| Conditions | Yield |
|---|---|
| With sodium formate | 97% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
114971-52-7
N,N,N-trimethylbenzenemethanaminium dichloroiodate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dihydrogen peroxide; iodine In water; acetonitrile at 20℃; Cooling with ice; | 96% |
| With Iodine monochloride In dichloromethane; water for 0.5h; Ambient temperature; | 86% |
| With Iodine monochloride In dichloromethane; water for 0.5h; Ambient temperature; | 86% |
| With Iodine monochloride In dichloromethane; water at 20℃; for 0.666667h; |
-
-
56-93-9
benzyltrimethylammonium chloride

-
-
121309-88-4
benzyltrimethylazanium tetrachloro-λ3-iodanuide

| Conditions | Yield |
|---|---|
| With iodine trichloride In dichloromethane for 0.5h; Ambient temperature; | 96% |
-
-
13463-40-6
iron pentacarbonyl

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With KOH In methanol; water N2-atmosphere; addn. of Te-salt in water to Fe(CO)5 in 50 % KOH (aq.) and MeOH; stirring (2 h, room temp.); filtration; dropwise addn. of ammonium salt in water (pptn.); filtration; washing (water); drying (vac.); recrystn. (CH2Cl2); elem. anal.; | 96% |

-
-
52427-61-9
potassium 1,2-cyclobutane-3,4-dithiolate

-
-
10025-82-8
indium(III) chloride

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In water 3-3 equiv. of K-salt and N-compd. were dissolved in H2O, 1 equiv. of InCl3 was slowly added in H2O; ppt. was filtered off, dried in an exsiccator, elem. anal.; | 96% |


| Conditions | Yield |
|---|---|
| In dichloromethane Inert atmosphere; Schlenk technique; | 96% |
-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide; potassium bromide; molybdic acid In water at 0 - 20℃; for 0.916667h; | 95.25% |
| With sodium bromate; hydrogen bromide In dichloromethane; water for 0.25h; | 85% |
| With sodium bromate; hydrogen bromide In water Ambient temperature; | 78% |
-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide; potassium bromide; molybdic acid In water at 0 - 20℃; for 0.916667h; Product distribution / selectivity; | 95.25% |
-
-
56-93-9
benzyltrimethylammonium chloride

-
-
603-35-0
triphenylphosphine

-
-
1100-88-5
benzyltriphenylphosphonium chloride

| Conditions | Yield |
|---|---|
| at 250℃; for 0.166667h; Irradiation; | 95% |
-
-
56-93-9
benzyltrimethylammonium chloride

-
-
70857-63-5
bis<3-<3-(3-hydroxypropyl)-1,1,3,3-tetramethyldisiloxanyl>propyl> sulfosuccinate sodium salt

-
-
82803-88-1
bis<3-<3-(3-hydroxypropyl)-1,1,3,3-tetramethyldisiloxanyl>propyl> sulfosuccinate trimethylbenzylammonium salt

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 6h; | 94.8% |

-
-
13463-40-6
iron pentacarbonyl

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With KOH In methanol N2-atmosphere; addn. of 1 equiv. of solid K2SeO3 to equimolar amt. Fe(CO)5 in MeOH (contg. excess KOH), prolonged stirring (observation of intermediates), filtration, dropwise addn. of excess aq. PhCH2NMe3Cl to filtrate (pptn.); collection (filtration), washing (water), drying (vac.), recrystn. (THF/ether); elem. anal.; | 94% |
| With KOH In methanol; water N2-atmosphere; addn. of 1 equiv. aq. K2SeO3 to equimolar amt. Fe(CO)5 in MeOH (contg. excess KOH), stirring for 5 h (room temp.), filtration, dropwise addn. of excess aq. PhCH2NMe3Cl to filtrate (pptn.); collection (filtration), washing (water), drying (vac.), recrystn. (THF/ether); elem. anal.; | 94% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
62635-69-2
sodium di(1,1,3-trihydrotetrafluoropropyl) sulfosuccinate

| Conditions | Yield |
|---|---|
| In ethanol at 75 - 80℃; for 2h; | 92.8% |
-
-
110-65-6
1,4-dihydroxybut-2-yne

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
107-05-1
3-chloroprop-1-ene

-
-
90925-51-2
1,4-bis(allyloxy)-2-butyne

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 92% |

-
-
107-31-3
Methyl formate

-
-
50987-90-1
1,4-bis(chloromethyl)-2,5-dichlorobenzene

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
2800-99-9
2,5-dichlorobenzene-1,4-dimethanol

| Conditions | Yield |
|---|---|
| With sodium formate In methanol; sodium methylate | 92% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
33252-64-1
3-nitro-5-trifluoromethylpyridin-2-ol

-
-
72587-15-6
2-chloro-3-nitro-5-(trifluoromethyl)pyridine

| Conditions | Yield |
|---|---|
| With trichlorophosphate In acetonitrile for 3h; Heating; | 92% |
-
-
56-93-9
benzyltrimethylammonium chloride

-
-
1413396-80-1
benzyltrimethylammonium dibromoiodate(I)

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dihydrogen peroxide; iodine In water; acetonitrile at 20℃; Cooling with ice; | 92% |
-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
834-14-0
p-benzylanizole

| Conditions | Yield |
|---|---|
| With potassium phosphate; (NC5H3(N2C7H4(CH2)3CH3)2)NiBr(1+)*Br(1-)=Ni(NC5H3(N2C7H4(CH2)3CH3)2)Br2; triphenylphosphine In 1,4-dioxane at 80℃; for 24h; Suzuki-Miyaura Coupling; Schlenk technique; | 92% |
| With potassium phosphate; palladium diacetate; XPhos In tetrahydrofuran at 50℃; for 14h; Sealed tube; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 72h; Inert atmosphere; Schlenk technique; Reflux; | 91% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
62607-32-3
sodium bis(2,2,3,3,4,4,5,5-octafluoro-1-pentyl) sulfosuccinate

-
-
80556-00-9
1,2-Bis-(2,2,3,3,4,4,5,5-octafluoro-pentyloxycarbonyl)-ethanesulfonatebenzyl-trimethyl-ammonium;

| Conditions | Yield |
|---|---|
| In ethanol at 75 - 80℃; | 90% |
| In ethanol at 78℃; for 2h; |
-
-
421-85-2
Trifluoromethanesulfonamide

-
-
711-82-0
2-(1-naphthyloxy)-1-ethanol

-
-
56-93-9
benzyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| Stage #1: Trifluoromethanesulfonamide With pyridine; sulfuryl dichloride In acetonitrile at 20℃; for 1.5h; Cooling with ice; Stage #2: 2-(1-naphthyloxy)-1-ethanol With dmap In acetonitrile at 50 - 80℃; for 24h; Cooling with ice; Stage #3: benzyltrimethylammonium chloride In methanol; water; acetonitrile for 0.5h; | 90% |

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
4559-70-0
Diphenylphosphine oxide

-
-
2959-74-2
benzyldiphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium tert-butylate at 60℃; for 3h; Schlenk technique; Inert atmosphere; | 90% |
| With [1,1'-bis(diphenylphosphino)ferrocene]nickel(II) chloride; caesium carbonate In acetonitrile at 100℃; for 18h; Schlenk technique; Inert atmosphere; | 67% |

-
-
10147-36-1
n-propanesulfonyl chloride

-
-
56-93-9
benzyltrimethylammonium chloride

-
-
189206-85-7
6-(1-Ethyl-5-n-propanesulfonyloxypyrazol-4-yl)carbonyl-3,3,5,8-tetramethylthiochroman-4-one-1,1-dioxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane; water | 89% |

Benzyltrimethylammonium chloride Chemical Properties
Molecule structure of Benzyltrimethylammonium chloride (CAS NO.56-93-9):
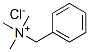
IUPAC Name: Benzyl(trimethyl)azanium chloride
Molecular Formula: C10H16ClN
Molecular Weight: 185.70 g/mol
EINECS: 200-300-3
Density: 1.08 g/mL at 25 °C
Melting Point: 236 °C (Decomposes)
Water Solubility: 800 g/L
Sensitive: hygroscopic
H-Bond Acceptor: 1
Rotatable Bond Count: 2
Exact Mass: 185.097127
MonoIsotopic Mass: 185.097127
Canonical SMILES: C[N+](C)(C)CC1=CC=CC=C1.[Cl-]
InChI: InChI=1S/C10H16N.ClH/c1-11(2,3)9-10-7-5-4-6-8-10;/h4-8H,9H2,1-3H3;1H/q+1;/p-1
InChIKey: KXHPPCXNWTUNSB-UHFFFAOYSA-M
EINECS: 200-300-3
Product Categories: Pharmaceutical Intermediates;Organics;Phenyls & Phenyl-Het;Ammonium Chlorides (Quaternary);Quaternary Ammonium Compounds;Asymmetric Synthesis;Synthetic Organic Chemistry;Phenyls & Phenyl-Het;Ammonium Salts;Greener Alternatives: Catalysis;Phase Transfer Catalysts
Benzyltrimethylammonium chloride Uses
Benzyltrimethylammonium chloride (CAS NO.56-93-9) can be used as pharmaceutical intermediate and phase transfer catalyst. It is also used for the determination of platinum, palladium and mercury and gold and used as emulsifier and inhibitor.
Benzyltrimethylammonium chloride Toxicity Data With Reference
| 1. | orl-rat LDLo:250 mg/kg | NCNSA6 National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. 5 (1953),39. | ||
| 2. | orl-mus LDLo:1600 mg/kg | JPMSAE Journal of Pharmaceutical Sciences. 69 (1980),327. |
Benzyltrimethylammonium chloride Consensus Reports
Reported in EPA TSCA Inventory.
Benzyltrimethylammonium chloride Safety Profile
Hazard Codes:  Xn
Xn
Risk Statements: 22-36/38-36
R22:Harmful if swallowed.
R36/38:Irritating to eyes and skin.
R36:Irritating to eyes.
Safety Statements: 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
RIDADR: UN 2811 6.1/PG 3
WGK Germany: 3
RTECS: BO8400000
F: 3
HS Code: 29239000
Poison by ingestion. Combustible. When heated to decomposition it emits very toxic fumes of NH3, NOx, and Cl−.
Benzyltrimethylammonium chloride Specification
Benzyltrimethylammonium chloride (CAS NO.56-93-9) is also named as Ammonium, benzyltrimethyl-, chloride ; BTM ; Benzenemethanaminium, N,N,N-trimethyl-, chloride ; Benzyl trimethylammonium chloride ; CCRIS 4587 ; HSDB 4196 ; N,N,N-Trimethylbenzenemethanaminium chloride ; Tmbac . Benzyltrimethylammonium chloride (CAS NO.56-93-9) is white to light yellow crystal powder. It is soluble in water, ethanol and butanol and insoluble in ether . Benzyltrimethylammonium chloride can react with oxidizing materials. Toxic oxides of nitrogen and hydrochloric acid fumes may form in fire. Ingestion causes gastrointestinal disturbances. Contact with liquid irritates eyes and may irritate skin.
Related Products
- Benzyltrimethylammonium bromide
- Benzyltrimethylammonium chloride
- Benzyltrimethylammonium dichloroiodate
- Benzyltrimethylammonium fluoride
- Benzyltrimethylammonium hydroxide
- Benzyltrimethylammonium iodide
- Benzyltrimethylammonium methoxide
- Benzyltrimethylammonium tetrachloroiodate
- Benzyltrimethylammonium tribromide
- 56-94-0
- 569-41-5
- 569-42-6
- 56946-83-9
- 5694-72-4
- 56949-40-7
- 5694-95-1
- 56-95-1
- 569-51-7
- 5695-52-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View