-
Name
Butyldiglycol
- EINECS 203-961-6
- CAS No. 112-34-5
- Article Data25
- CAS DataBase
- Density 0.949 g/cm3
- Solubility soluble
- Melting Point -68 °C(lit.)
- Formula C8H18O3
- Boiling Point 230.4 °C at 760 mmHg
- Molecular Weight 162.229
- Flash Point 100 °C
- Transport Information
- Appearance colourless liquid
- Safety 24-26
- Risk Codes 36
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-(2-Butoxyethoxy)ethanol;2-(2-n-Butoxyethoxy)ethanol;3,6-Dioxa-1-decanol;BDG;BDG-NS;BDGE 20;BikanolB 2;Butadigol;Butoxyethoxyethanol;Butycenol 20P;Butyl Carbitol;ButylDiglysolv;Butyl Oxitol glycol ether;Butyl digol;Diethylene glycol butylether;Diethylene glycol mono-n-butyl ether;Diethylene glycol monobutyl ether;Ethanol, 2,2'-oxybis-, monobutyl ether;
- PSA 38.69000
- LogP 0.81200
Synthetic route
-
-
75-21-8
oxirane

-
-
71-36-3
butan-1-ol

-
A
-
111-76-2
2-Butoxyethanol

-
B
-
112-34-5
Diethylene glycol monobutyl ether

| Conditions | Yield |
|---|---|
| With potassium aluminum sulfate at 170℃; for 24h; Reagent/catalyst; Autoclave; | A 93.1% B 6.63% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; water at 100℃; for 24h; | 70% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate at 50℃; |

| Conditions | Yield |
|---|---|
| sulfuric acid In toluene Heating; |

| Conditions | Yield |
|---|---|
| With diethylene glycol |
-
-
75-21-8
oxirane

-
-
71-36-3
butan-1-ol

-
A
-
111-76-2
2-Butoxyethanol

-
B
-
112-34-5
Diethylene glycol monobutyl ether

-
C
-
143-22-6
triethyleneglycol monobutyl ether

| Conditions | Yield |
|---|---|
| 2,4,6-trimethyl-pyridine at 160℃; under 2311.54 - 3345.86 Torr; for 2.5h; Product distribution / selectivity; Inert atmosphere; | |
| sodium butanolate at 140℃; under 7500.75 Torr; for 5h; Product distribution / selectivity; | A 20.16 %Chromat. B 9.07 %Chromat. C 6.10 %Chromat. |
| catalyst of invention (Sb2O3, copper acetate, hydrogen peroxide; calcined) at 140℃; under 30003 Torr; for 24 - 120h; Product distribution / selectivity; | A 76 - 77 %Chromat. B 13 - 14 %Chromat. C 9 %Chromat. |

-
-
129-00-0
pyrene

-
A
-
85-44-9
phthalic anhydride

-
B
-
90-47-1
xanth-9-one

-
C
-
629-62-9
pentadecane

-
D
-
629-97-0
n-docosane

-
E
-
630-01-3
n-hexacosane

-
F
-
629-59-4
tetradecane

-
G
-
544-76-3
Hexadecane

-
H
-
112-34-5
Diethylene glycol monobutyl ether

-
I
-
97-87-0
n-butyl isobutyrate

-
J
-
629-94-7
heneicosane

-
K
-
638-67-5
n-tricosane

-
L
-
646-31-1
tetracosane

-
M
-
629-99-2
n-pentacosane

-
N
-
203-63-4
cyclopenta[def]phenanthrene

-
P
-
128-37-0
2,6-di-tert-butyl-4-methyl-phenol

-
Q
-
84-66-2
Diethyl phthalate

-
R
-
84-74-2
Phthalic acid dibutyl ester

-
S
-
203-64-5
4H-Cyclopenta[def]phenanthrene

-
T
-
85-68-7
benzyl n-butyl phthalate

-
U
-
4371-26-0
1,1'-Biphenyl-2,2',6,6'-tetracarboxaldehyde

-
V
-
16162-34-8
4,5-phenanthrene-8,9-dicarbaldehyde

-
W
-
57-10-3
1-hexadecylcarboxylic acid

| Conditions | Yield |
|---|---|
| With oxygen; ozone In water for 0.25 - 2h; |

-
-
112-60-7
Tetraethylene glycol

-
-
107-21-1
ethylene glycol

-
-
123-72-8
butyraldehyde

-
-
111-46-6
diethylene glycol

-
-
112-27-6
2,2'-[1,2-ethanediylbis(oxy)]bisethanol

-
A
-
3390-13-4
2-propyl-1,3-dioxolane

-
B
-
111-76-2
2-Butoxyethanol

-
C
-
112-34-5
Diethylene glycol monobutyl ether

-
D
-
143-22-6
triethyleneglycol monobutyl ether

-
E
-
1559-34-8
tetraethylene glycol monobutyl ether

-
F
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; palladium 10% on activated carbon at 180℃; under 51716.2 Torr; for 2h; |


| Conditions | Yield |
|---|---|
| With hydrogen; palladium 10% on activated carbon at 200℃; under 51716.2 Torr; for 2h; Inert atmosphere; | 94.5 %Chromat. |
| With palladium 10% on activated carbon; hydrogen at 200℃; under 51755.2 Torr; for 2h; | 94.5 %Chromat. |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
50964-16-4
diethylene glycol monobutyl ether tosylate

| Conditions | Yield |
|---|---|
| Stage #1: Diethylene glycol monobutyl ether; p-toluenesulfonyl chloride With dmap In dichloromethane at 20℃; Stage #2: With triethylamine In dichloromethane at 20℃; | 100% |
| With sodium hydroxide In tetrahydrofuran; water at 0℃; for 2h; | 85% |
| With pyridine In dichloromethane at 20℃; for 3h; | 57% |
| With pyridine |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
25354-97-6
2-hexyldecanoic acid

| Conditions | Yield |
|---|---|
| With p-toluenesulfonic acid monohydrate | 99% |
| With p-toluenesulfonic acid monohydrate | 99% |

| Conditions | Yield |
|---|---|
| With sodium borohydrid In 5,5-dimethyl-1,3-cyclohexadiene | 98% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
598-21-0
2-Bromoacetyl bromide

-
-
56521-78-9
2-(2-butoxyethoxy)ethyl 2-bromoacetate

| Conditions | Yield |
|---|---|
| With sodium carbonate In dichloromethane at -5 - 20℃; for 12h; Green chemistry; | 98% |
| With triethylamine In dichloromethane at -78℃; Inert atmosphere; | 72% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
107-13-1
acrylonitrile

-
-
35633-48-8
3-[2-(2-butoxyethoxy)ethoxy]propanenitrile

| Conditions | Yield |
|---|---|
| With sodium methylate for 1.16667h; | 97% |
-
-
124-04-9
Adipic acid

-
-
111-77-3
2-(2-methoxyethoxy)ethyl alcohol

-
-
112-34-5
Diethylene glycol monobutyl ether

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In toluene at 118 - 145℃; for 6.5h; Heating / reflux; | 96.9% |

-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
123-63-7
paracetaldehyde

-
-
3895-17-8
2-butoxyethyl 2-ethoxyethyl ether

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal at 160℃; under 760 Torr; for 18h; Etherification; | 96% |
| With hydrogen; palladium on activated charcoal at 160℃; for 10h; atmospheric pressure; | 96% |

| Conditions | Yield |
|---|---|
| With pyridine; thionyl chloride In chloroform for 10h; Reflux; | 95% |
| With pyridine; thionyl chloride |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
124-13-0
Octanal

-
-
101433-27-6
2-butoxyethyl 2-(octoxy)ethyl ether

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal at 160℃; under 760 Torr; for 18h; Etherification; | 95% |
| With hydrogen; palladium on activated charcoal at 160℃; for 10h; atmospheric pressure; | 95% |

| Conditions | Yield |
|---|---|
| With pyridine 1.) 0 deg C, 5 h, 2.) rt, 12 h; | 90% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
1121-22-8, 1436-59-5, 20439-47-8, 21436-03-3, 41013-43-8, 694-83-7
trans-1,2-Diaminocyclohexane

| Conditions | Yield |
|---|---|
| Stage #1: Diethylene glycol monobutyl ether; Hexamethylene diisocyanate; dibutyltin dilaurate In hexane at 20 - 45℃; Stage #2: trans-1,2-Diaminocyclohexane In hexane at 15 - 20℃; for 0.5h; | 88.8% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
626-00-6
1,3-Diiodobenzene

-
-
71784-21-9
1,3-Bis-[2-(2-butoxy-ethoxy)-ethoxy]-benzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In 5,5-dimethyl-1,3-cyclohexadiene for 20h; Reflux; Inert atmosphere; | 86% |
-
-
50-00-0
formaldehyd

-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
118600-20-7, 134731-86-5
rccc-2,8,14,20-tetrakis-(iso-butyl)-resorcin[4]arene

| Conditions | Yield |
|---|---|
| With iminodiacetic acid In acetonitrile | 81% |


| Conditions | Yield |
|---|---|
| In pentane byproducts: HCl; N2, equimol., V compd. added dropwise to a soln. of alcohol, stirred for2 h, heated on water bath; decanted, concd., stored at 0°C, ppt. collected, dried (vac.); elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In 5,5-dimethyl-1,3-cyclohexadiene at 145℃; for 26h; Inert atmosphere; | 79% |
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In 5,5-dimethyl-1,3-cyclohexadiene at 145℃; for 26h; Inert atmosphere; | 79% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
34713-70-7
2-Phenylpropanal

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; methanesulfonic acid sodium salt In toluene for 15h; Inert atmosphere; Reflux; Dean-Stark; | 79% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
31250-16-5
[2-(2-chloro-2-oxoethoxy)phenoxy]acetyl chloride

| Conditions | Yield |
|---|---|
| In tetrachloromethane Ambient temperature; | 76% |
-
-
1938-32-5
5-(chloromethyl)-6-propyl-1,3-benzodioxole

-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
51-03-6
Piperonyl butoxide

| Conditions | Yield |
|---|---|
| Stage #1: Diethylene glycol monobutyl ether With sodium hydroxide In water Reflux; Stage #2: 5-(chloromethyl)-6-propyl-1,3-benzodioxole In water for 5h; Reflux; | 75% |
| With sodium hydroxide at 30℃; for 5h; | 230 g |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
615-42-9
1,2-Diiodobenzene

-
-
71784-20-8
1,2-bis(2-(2-(butyloxy)ethoxy)ethoxy)benzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In 5,5-dimethyl-1,3-cyclohexadiene for 40h; Reflux; Inert atmosphere; | 75% |
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In 5,5-dimethyl-1,3-cyclohexadiene at 140℃; for 24h; Inert atmosphere; | 4 g |

| Conditions | Yield |
|---|---|
| With NAH In tetrahydrofuran at 80℃; Inert atmosphere; | 71% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
40926-73-6
2-(2-methoxyphenoxy)acetyl chloride

| Conditions | Yield |
|---|---|
| In chloroform for 12h; Heating; | 69.01% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
103781-33-5
3-{[(4-bromobutyl)oxy]methyl}-3-methyloxetane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In hexane for 5h; Heating; | 68% |
-
-
38020-81-4
2-iodoacetyl chloride

-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
56521-91-6
2-(2-butoxyethoxy)ethyl-2-iodoacetate

| Conditions | Yield |
|---|---|
| With sodium carbonate In dichloromethane at -5 - 20℃; for 12h; Green chemistry; | 68% |
-
-
112-34-5
Diethylene glycol monobutyl ether

-
-
20142-87-4
2-(2-nitrophenoxy)acetyl chloride

| Conditions | Yield |
|---|---|
| In chloroform for 12h; Heating; | 66.89% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
112-34-5
Diethylene glycol monobutyl ether

-
A
-
1245625-50-6
C24H51Cl3N3O9P3

-
B
-
1245625-47-1
C24H51Cl3N3O9P3

-
C
-
1245625-36-8
C24H51Cl3N3O9P3

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at -60 - 20℃; for 3.5h; Inert atmosphere; | A 20% B 16% C 64% |

-
-
112-34-5
Diethylene glycol monobutyl ether

-
A
-
1245625-50-6
C24H51Cl3N3O9P3

-
B
-
1245625-47-1
C24H51Cl3N3O9P3

-
C
-
1245625-36-8
C24H51Cl3N3O9P3

| Conditions | Yield |
|---|---|
| With 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydride In tetrahydrofuran at -60 - 20℃; for 3.5h; Inert atmosphere; | A 20% B 16% C 64% |
| With 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydride In tetrahydrofuran at -60 - 20℃; for 3.5h; Inert atmosphere; | A 20% B 16% C 64% |

-
-
112-34-5
Diethylene glycol monobutyl ether

| Conditions | Yield |
|---|---|
| With pyridine In acetonitrile for 24h; Reflux; | 60% |
| With pyridine In acetonitrile for 24h; Reflux; |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 18h; Dean-Stark; Reflux; | 60% |
Butyldiglycol Consensus Reports
Butyldiglycol Standards and Recommendations
Butyldiglycol Specification
The Diethylene glycol monobutyl ether with CAS registry number of 112-34-5 is also called Ethanol,2-(2-butoxyethoxy)-. The IUPAC name is 2-(2-butoxyethoxy)ethanol. Its EINECS registry number is 203-961-6. In addition, the molecular formula is C8H18O3 and the molecular weight is 162.23. It is a kind of colourless liquid with a mild odour and belongs to the classes of Ethylene Glycols Monofunctional Ethylene Glycols and Monofunctional Ethylene Glycols. And it is stable and incompatible with strong oxidizing agents and strong bases.
Physical properties about this chemical are: (1)ACD/LogP: 0.44; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.44; (4)ACD/LogD (pH 7.4): 0.44; (5)ACD/BCF (pH 5.5): 1.27; (6)ACD/BCF (pH 7.4): 1.27; (7)ACD/KOC (pH 5.5): 41.26; (8)ACD/KOC (pH 7.4): 41.26; (9)#H bond acceptors: 3; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 9; (12)Polar Surface Area: 38.69 Å2; (13)Index of Refraction: 1.43; (14)Molar Refractivity: 44.13 cm3; (15)Molar Volume: 170.8 cm3; (16)Polarizability: 17.49 ×10-24cm3; (17)Surface Tension: 32 dyne/cm; (18)Density: 0.949 g/cm3; (19)Flash Point: 100 °C; (20)Enthalpy of Vaporization: 54.29 kJ/mol; (21)Boiling Point: 230.4 °C at 760 mmHg; (22)Vapour Pressure: 0.0126 mmHg at 25°C.
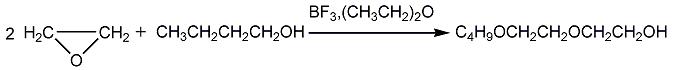
Preparation of Diethylene glycol monobutyl ether: it can be prepared by butanol and ethyl ether boron trifluoride. This reaction will need reagent epoxyethane. The reaction time is 2-3 hours at reaction temperature of 75-80 °C. After the reaction, use butanol sodium to make pH into 8, then put it into the distillation tower for distillation.
Uses of Diethylene glycol monobutyl ether: it can be used as solvent of nitrocellulose, varnish, printing ink, the oil ans the resin. It also can be used for adhesive thinner and emulsioni paint stabilizer. In addition, it can react with (2-methoxy-phenoxy)-acetyl chloride to get Butoxyethoxyethyl o-methoxyphenoxyacetate. This reaction will need reagent CHCl3. The reaction time is 21 hours by heating. The yield is about 69.01%.
.jpg)
When you are using this chemical, please be cautious about it as the following:
It is irritating to the eyes. during using it, avoid contact with skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES:CCCCOCCOCCO
(2)InChI:InChI=1/C8H18O3/c1-2-3-5-10-7-8-11-6-4-9/h9H,2-8H2,1H3
(3)InChIKey:OAYXUHPQHDHDDZ-UHFFFAOYAO
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 2gm/kg (2000mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC GASTROINTESTINAL: OTHER CHANGES KIDNEY, URETER, AND BLADDER: OTHER CHANGES | Journal of Industrial Hygiene and Toxicology. Vol. 23, Pg. 259, 1941. |
| mouse | LD50 | intraperitoneal | 850mg/kg (850mg/kg) | LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" BLOOD: CHANGES IN SPLEEN | Federation Proceedings, Federation of American Societies for Experimental Biology. Vol. 6, Pg. 342, 1947. |
| mouse | LD50 | oral | 2400mg/kg (2400mg/kg) | Journal of the American College of Toxicology. Vol. 12, Pg. 139, 1993. | |
| mouse | LD50 | unreported | 6050mg/kg (6050mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 46(2), Pg. 14, 1981. | |
| rabbit | LD50 | oral | 2200mg/kg (2200mg/kg) | "Patty's Industrial Hygiene and Toxicology," 3rd rev. ed., Clayton, G.D., and F.E. Clayton, eds., New York, John Wiley & Sons, Inc., 1978-82. Vol. 3 originally pub. in 1979; pub. as 2n rev. ed. in 1985.Vol. 2C, Pg. 3964, 1982. | |
| rabbit | LD50 | skin | 2700mg/kg (2700mg/kg) | Journal of the American College of Toxicology. Vol. 12, Pg. 139, 1993. | |
| rat | LD50 | oral | 5660mg/kg (5660mg/kg) | Dow Chemical Company Reports. Vol. MSD-41, | |
| rat | LD50 | unreported | 4500mg/kg (4500mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 46(2), Pg. 14, 1981. |
Related Products
- Butyldiglycol
- 1123-49-5
- 1123515-36-5
- 1123515-90-1
- 1123-54-2
- 1123-55-3
- 112-35-6
- 1123-56-4
- 1123-61-1
- 1123620-89-2
- 112362-50-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View