Qingdao KMS Biochemical Technology Co.,Ltd.
Appearance:White crystal powder Storage:Be airtight in a cool and dry environment Package:25 kg cardboard barrels Application:Due to the fluidity of the phosphorus-chlorine bond, chlorine can be easily replaced, and a series of phosphoronitrile deriv
Cas:940-71-6
Min.Order:25 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:940-71-6
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryJinan Finer Chemical Co., Ltd
Product Name Phosphonitrilic chloride trimer CAS No. 940-71-6 Appearance White crystal powder Assay ≥99% Capacity 500mt/year MOQ 100G Application Polyphosphazene h
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $10.0 / 15.0
Type:Lab/Research institutions
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience 2. Free samples will be provided,ensure specifications and quality are right for customer 3. Customers will receive the most professional techn
Cas:940-71-6
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:940-71-6
Min.Order:1
Negotiable
Type:Other
inquiryChemwill Asia Co., Ltd.
Phosphonitrilic chloride trimer Manufacturer Factory CAS 940-71-6 Phosphonitrilic chloride trimer Manufacturer High quality Best price In stock factory CAS 940-71-6 Phosphonitrilic chloride trimer COA TDS price MSDS highly quality and immeda
Cas:940-71-6
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:Phosphonitrilic chloride trimer CAS NO: 940-71-6 Grade:Medical scientific research and export Molecular formula:Cl6N3P3 Molecular weight: 347.6594 Product Quality 12 years of chemical raw materials Mature operation of the industry Sys
Cas:940-71-6
Min.Order:25 Kilogram
FOB Price: $2.0 / 3.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Phosphonitrilic chloride trimer Basic information Product Name: Phosphonitrilic chloride trimer Synonyms: PPZ;PHOSPHORUS NITRILE DICHLORIDE TRIMER;PHOSPHONITRILIC CHLORIDE TRIMER;PHOSPHO
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:940-71-6
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $298.0 / 478.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Name: Phosphonitrilic chloride trimer Synonyms: 1,3,5-Triaza-2,4,6-triphosphorin-2,2,4,4,6,6-hexachloride CAS: 940-71-6 MF: Cl6N3P3 Appearance: white powder Storage:Store in cool and dry place, away from sun light. Package: 25kgs/drum Applic
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Other
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:white or off-white powder Storage:Store in sealed containers
Cas:940-71-6
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:940-71-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Product Name Triethanolamine Other names Triethanolamine supplier,Triethanolamine factory,Triethanolamin eprice,Triethanolamine manufactuer,Triethanolamine china CAS NO 102-71-6 Molecular Formula
Cas:940-71-6
Min.Order:1 Gram
FOB Price: $1.0 / 5.0
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:940-71-6
Min.Order:1 Kilogram
FOB Price: $76.0 / 85.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Phosphonitrilic chloride trimer CAS:940-71-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality or
Cas:940-71-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:940-71-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:940-71-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Fandachem Co.,Ltd
Hangzhou Fanda Chemical Co.,Ltd (FandaChem) , a China-based chemical company, specialize in exporting Phosphonitrilic chloride trimer;Ppz CAS:940-71-6 , Please contact us by email freely. We are leading exporter in China. If you reall
Cas:940-71-6
Min.Order:1 Metric Ton
Negotiable
Type:Other
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Afine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Cas:940-71-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:940-71-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySinoway Industrial Co., Ltd.
Why is SINOWAY: 1) Specialized in pharmaceutical and healthcare industrial for 34 years. 2) ISO 9001:2015 & SGS audited supplier . 3) Accept various payment terms : T.T 30-60 days. 4) We have warehouse in USA with quickly shipment . 5) We c
Cas:940-71-6
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryWuhan Xinhao Biotechnology Co., Ltd.
1. We can provide customers with "one-stop"packaging service,from research,development,production,export and so on 2. Powerful R&D strength let our technology in a leading level,forever,in turn,to provide customers with better service .
Cas:940-71-6
Min.Order:100 Gram
FOB Price: $10.0 / 100.0
Type:Trading Company
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:940-71-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and als
Cas:940-71-6
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white crystalline powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used in
Cas:940-71-6
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquirySynthetic route
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
12125-02-9
ammonium chloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| With pyridine; magnesium chloride In chlorobenzene at 80 - 132℃; Inert atmosphere; | 71% |
| Stage #1: ammonium chloride With pyridine; magnesium chloride In 1,1,2,2-tetrachloroethane at 145℃; for 12h; Stage #2: phosphorus pentachloride In 1,1,2,2-tetrachloroethane at 145℃; Solvent; Temperature; | 61.78% |
| In chlorobenzene at 120 - 130℃; Inert atmosphere; Reflux; | 60.6% |
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
12125-02-9
ammonium chloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
14514-98-8
(NPCl2)8

-
C
-
2950-45-0
octachlorocyclotetraphosphazene

-
E
-
13596-41-3
pentakisphosphorus nitride dichloride

-
F
-
2851-52-7
hexakisphosphorus nitride dichloride

-
G
-
13827-30-0
heptakisphosphorus nitride dichloride

| Conditions | Yield |
|---|---|
| In chlorobenzene Schlenk technique; Reflux; | A 64% B 6% C 5% D 2% E 16% F 16% G 13% |

-
-
1586-73-8
tris(trimethylsilyl)amine

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In dichloromethane inert atmosphere; dropwise addn. of 1 equiv. PCl5 to amine at 25°C, stirring for 12 h; not isolated, detd. by (31)P-NMR spectroscopy; | 49% |
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In chlorobenzene react. of PCl5 with NH4Cl (ratio 1:1.5) in C6H5Cl at 124-127 °C (20-50 h);; | 40% |
| In chlorobenzene react. of PCl5 with NH4Cl (ratio 1:1.2) in C6H5Cl at 132-134 °C (20 h);; | 20-22 |
| In further solvent(s) in 1,1,2,2-tetrachloroethane; recrystn. from petroleum ether; |
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
7664-41-7
ammonia

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In further solvent(s) passing gaseous NH3 in a soln. of PCl5 in (CHCl2)2;; | 16% |
-
-
1586-73-8
tris(trimethylsilyl)amine

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
40678-60-2
trichloro(trimethylsilyl)phosphoranimine

| Conditions | Yield |
|---|---|
| In dichloromethane inert atmosphere; dropwise addn. of 0.5-1 equiv. PCl5 to amine at -78 or25°C, stirring for 30 min to 12 h; yields depending on educt rat io, reaction temp., time and rate of addn.; product mixt. not sepd., detd. by (31)P-NMR spectroscopy; | A 5% B 5% |

-
-
7719-09-7
thionyl chloride

-
-
28950-34-7
tetrasulfur tetranitride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In benzene addn. of PCl3 to a soln. of S4N4 in C6H6 containing SOCl2; formation of a brown main product and crystals of (PNCl2)3;; | |
| In benzene addn. of PCl3 to a soln. of S4N4 in C6H6 containing SOCl2; formation of a brown main product and crystals of (PNCl2)3;; |

-
-
10124-48-8
mercury amidochloride

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating with HgNH2Cl; formation of a mixture of P3N3Cl6, (PNCl2)(n), HgCl2 and NH4Cl;; | |
| In neat (no solvent) heating with HgNH2Cl; formation of a mixture of P3N3Cl6, (PNCl2)(n), HgCl2 and NH4Cl;; |

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
7664-41-7
ammonia

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In tetrachloromethane pptn. of NH4Cl from a soln. of 3 g PCl5 in 1000 ml CCl4 half-saturated with gaseous NH3 at -3 °C; soln. contains (PNCl2)3 and (PNCl2)4;; evapn. of the mother liquor, fractional crystn. from toluene;; | |
| In tetrachloromethane pptn. of NH4Cl from a soln. of 3 g PCl5 in 1000 ml CCl4 half-saturated with gaseous NH3 at -3 °C; soln. contains (PNCl2)3 and (PNCl2)4;; evapn. of the mother liquor, fractional crystn. from toluene;; |


-
-
7782-50-5
chlorine

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In further solvent(s) dissolving PCl3 distd. under N2 in benzyl chloride distd. under N2; addn. of NH4Cl dryed in vac. and passing in the corresponding amount of Cl2; heating in a stream of N2 at 130 °C; removal of formed HCl by a stream of N2;; filtration, distg. off the solvent at 40 °C and 0.5 Torr; recrystn. from petroleum ether; product contains small amounts of higher polymers;; |


-
-
7782-50-5
chlorine

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
7647-01-0
hydrogenchloride

-
B
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
D
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In further solvent(s) dissolving PCl3 distd. under N2 in (CHCl2)2 distd. under N2; addn. of NH4Cl dryed in vac. and passing in the corresponding amount of Cl2; heating in a stream of N2 at 130 °C; removal of formed HCl by a stream of N2;; filtration, distg. off the solvent at 40 °C and 0.5 Torr; recrystn. from petroleum ether; products (NPCl2)3, (NPCl2)4 and a small amount of oily polymers;; | |
| In chlorobenzene dissolving PCl3 distd. under N2 in C6H5Cl distd. under N2; addn. of NH4Cl dryed in vac. and passing in the corresponding amount of Cl2; heating in a stream of N2 at 130 °C; removal of formed HCl by a stream of N2;; filtration, distg. off the solvent at 40 °C and 0.5 Torr; recrystn. from petroleum ether; product contains small amounts of higher polymers;; | |
| In further solvent(s) dissolving PCl3 distd. under N2 in (CHCl2)2 distd. under N2; addn. of NH4Cl dryed in vac. and passing in the corresponding amount of Cl2; heating in a stream of N2 at 130 °C; removal of formed HCl by a stream of N2;; filtration, distg. off the solvent at 40 °C and 0.5 Torr; recrystn. from petroleum ether; yield 35-40 %, product contains small amounts of higher polymers;; | A n/a B 35-40 C n/a D n/a |

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In not given react. of 400 g PCl5 with 120 g NH4Cl in an inert solvent;; sucking off the formed mass, washing with ice-cold C6H6; fractional distn.; yield 50 % (NPCl2)3, 25 % (NPCl2)4 and 25 % higher polymers;; |

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In not given react. of 400 g PCl5 with 120 g NH4Cl in an inert solvent;; sucking off the formed mass, washing with ice-cold C6H6; fractional distn.; yield 75 g (NPCl2)3, 25 g (NPCl2)4;; | |
| In not given react. of PCl5 with NH4Cl;; fractionation at 185-190 °C and 13 Torr; fraction contains 10 % (NPCl2)3;; | |
| In not given react. of PCl5 with NH4Cl;; fractionation at 185-190 °C and 13 Torr; fraction contains 10 % (NPCl2)3;; | |
| In further solvent(s) react. of finely divided NH4Cl and PCl5 in refluxing sym-tetrachloroethane; fractional crystn. from glacial acetic acid, cold benzene and conc. H2SO4; |


-
-
7782-50-5
chlorine

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
D
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent) formation of a mixt. of phosphorus nitride dichlorides with a large amount of (NPCl2)3 and a small amount of PCl5 by react. of P3N5 with Cl2 at 700 °C;; |


-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In nitrobenzene small amount of azide; detected by NMR; |
-
-
19597-69-4
lithium azide

-
-
2007-97-8
trichloro(o-phenylenedioxy)phosphorane

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
22295-99-4
hexaazido cyclotriphosphazene

-
C
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In nitrobenzene not isolated, detected by NMR; |

-
-
64373-45-1
2,4-dichloro-1,3-bis(dichlorophosphoryl)-2,4-dioxocyclodiphosph(V)azane

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
13966-08-0
dichlorophosphinylphosphorimidic trichloride

-
C
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In trichlorophosphate byproducts: Cl2(O)P(NPCl2)5Cl; under reflux; treatment of the crude product with n-heptane, products identified in the sol. and in the insol. phase with NMR spectroscopy; |


-
-
7782-50-5
chlorine

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
D
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent) formation of a mixt. of phosphorus nitride dichlorides with a large amount of (NPCl2)3 and a small amount of PCl5 by react. of P4N6 with Cl2 at 700 °C;; |

-
-
7719-09-7
thionyl chloride

-
-
28950-34-7
tetrasulfur tetranitride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In benzene formation of phosphorus nitride dichlorides with a large amount of (NPCl2)3 as by-products by addn. of PCl3 to a soln. of S4N4 in C6H6 containing SOCl2;; | |
| In benzene |


-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In further solvent(s) passing dry gaseous HCl into a suspension of phospham in 1.1.2.2-tetrachlorethane at 148°C for several h;; |

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) formation of phosphorus nitride dichlorides with a large amount of (NPCl2)3 on heating PCl5*8NH3 at 175-200 °C and 50 Torr;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) sublimation of (PNCl2)3 at 175-200 °C and 50 Torr;; | |
| In neat (no solvent) sublimation of (PNCl2)3 at 175-200 °C and 50 Torr;; |

-
-
7782-50-5
chlorine

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
7727-37-9
nitrogen

-
D
-
7786-30-3
magnesium chloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) formation of phosphorus nitride dichlorides with a large amount of (NPCl2)3 on heating PN3Mg2 in a stream of Cl2 at 700 °C;; |

-
-
21246-65-1
{Cl(Cl2PN)3PCl3}(1+)*{BCl4}(1-)={Cl(Cl2PN)3PCl3}{BCl4}

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
18460-54-3
{PCl4}(1+)*{BCl4}(1-)={PCl4}{BCl4}

| Conditions | Yield |
|---|---|
| 130°C; |
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
13827-30-0
heptakisphosphorus nitride dichloride

| Conditions | Yield |
|---|---|
| With quinoline In further solvent(s) heating a soln. of PCl5 in (CHCl2)2 with quinoline at 145 °C on refluxing;; distn. off solvent; formation of a mixt. of 40 % (NPCl2)3, 55-60 % (PNCl2)7 and 0-5 % impurities;; | |
| With quinoline |
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
25758-26-3, 13597-72-3
phosphoramide

-
A
-
7647-01-0
hydrogenchloride

-
B
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
C
-
13966-08-0
dichlorophosphinylphosphorimidic trichloride

-
D
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

| Conditions | Yield |
|---|---|
| In further solvent(s) react. of a soln. in tetrachlorethane with PCl5; immediate removal of HCl on passing thruogh a stream of air;; | |
| In further solvent(s) react. of a soln. in tetrachlorethane with PCl5; immediate removal of HCl on passing thruogh a stream of air;; |

-
-
1586-73-8
tris(trimethylsilyl)amine

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
2950-45-0
octachlorocyclotetraphosphazene

-
C
-
13596-41-3
pentakisphosphorus nitride dichloride

-
D
-
2851-52-7
hexakisphosphorus nitride dichloride

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: high oligomers; inert atmosphere; dropwise addn. of 1 equiv. PCl5 to amine at reflux, refluxing for 8 h; distn.; product mixt. not sepd., detd. by (31)P-NMR spectroscopy; |

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
7664-41-7
ammonia

-
A
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
B
-
2950-45-0
octachlorocyclotetraphosphazene

| Conditions | Yield |
|---|---|
| In tetrachloromethane incomplete satn. of a soln. of PCl5 in CCl4 with gaseous NH3;; | |
| In tetrachloromethane |

-
-
19597-69-4
lithium azide

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In nitrobenzene small amount of azide; detected by NMR; |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
7664-39-3
hydrogen fluoride

-
A
-
7647-01-0
hydrogenchloride

| Conditions | Yield |
|---|---|
| In not given react. with HF;; | A n/a B 100% |

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
15599-91-4
hexafluorophosphazene

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium Tetrafluoroborate In N,N-dimethyl-formamide at 50℃; for 5h; Reagent/catalyst; Solvent; Temperature; | 99.8% |
| With H2O or HF In nitrobenzene dissolving (NPCl2)3 in freshly distd. C6H5NO2; addn. of an excess of NaF or a small amount of H2O or HF under exclusion of H2O (drying tube with P2O5); heating on refluxing;; fractionation;; | |
| In gas byproducts: NaCl; (NPCl2)3 gas was passed over heated NaF at 550°C; monitoring by IR spectroscopy; |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
333-20-0
potassium thioacyanate

-
A
-
1858-42-0
hexaisothiocyanatocyclotriphosphazene

| Conditions | Yield |
|---|---|
| In acetone | A n/a B 99% |
| In acetone | A n/a B 99% |


-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
15599-91-4
hexafluorophosphazene

| Conditions | Yield |
|---|---|
| With 4-n-butyl-4-methylmorpholinium hydroxide In acetonitrile at 30℃; for 2h; Temperature; Solvent; Reagent/catalyst; Ionic liquid; | 98.7% |
| Reflux; | 90% |
| With catalyst: 18-crown-6 In tetrahydrofuran refluxing KF with catalytic amt. of crown for 20 min, dropwise addn. of 0.1 equiv. of N3P3Cl6, refluxing (1 h); fractional distn.; |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
106-41-2
4-bromo-phenol

-
-
4376-72-1
hexakis(4-bromophenoxy)cyclotriphosphazene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 18h; Inert atmosphere; Reflux; | 98% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
15599-91-4
hexafluorophosphazene

| Conditions | Yield |
|---|---|
| With potassium fluoride; Triethylene glycol dimethyl ether; ethanol In hexane at 0 - 30℃; for 2h; Solvent; Reagent/catalyst; Temperature; | 97.2% |
| With sodium fluoride In diethylene glycol dimethyl ether at 100℃; for 2h; Temperature; Solvent; Inert atmosphere; | 86% |
| In not given (N2); as in: (Schmutzler, R. Inorg Synth. 1967, 9, 75); fractional distn.; | |
| With sodium fluoride In ethyl acetate at 10℃; for 3h; Solvent; Temperature; Reflux; | 130 g |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
78-81-9
isobutylamine

-
-
101201-65-4
hexakis(iso-butylamino) cyclotriphosphazene

| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; | 97% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
1806-29-7
2,2'-dihydroxybiphenyl

-
-
128175-80-4
2,2-dichloro-4,4,6,6-bis[spiro(2',2''-dioxy-1'',1''-biphenyl)]cyclotriphosphazene

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran at 65℃; for 12h; Inert atmosphere; | 95.4% |
| With potassium carbonate In acetone at 0 - 20℃; for 24h; Inert atmosphere; | 92% |
| With potassium carbonate In acetone at 0 - 20℃; for 24h; Inert atmosphere; | 92% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
108-95-2
phenol

-
-
1184-10-7
2,2,4,4,6,6-hexaphenoxycyclotriphosphazene

| Conditions | Yield |
|---|---|
| Stage #1: phenol With tetrabutylammomium bromide; chlorobenzene; potassium hydroxide at 50℃; for 0.5h; Stage #2: 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine at 90℃; for 9h; Reagent/catalyst; Temperature; | 95.04% |
| Stage #1: phenol With sodium hydride In tetrahydrofuran Inert atmosphere; Stage #2: 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine In tetrahydrofuran at 70℃; Inert atmosphere; | 85% |
| With potassium carbonate In tetrahydrofuran at 50℃; for 24h; | 85.6% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
7664-41-7
ammonia

-
-
10535-05-4
2,2-diamino-4,4,6,6-tetrachlorocyclotriphosphazene

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; for 1.5h; | 95% |
| In diethyl ether; ammonia NH3 (liquid); molar ratio P3N3Cl2:NH3 1:4; | |
| In diethyl ether Schlenk technique; |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
77958-57-7
hexakis(4-formylphenoxy)cyclotriphosphazene

| Conditions | Yield |
|---|---|
| With caesium carbonate In tetrahydrofuran at 20℃; for 48.5h; Reagent/catalyst; Cooling with ice; | 94% |
| Stage #1: 4-hydroxy-benzaldehyde With triethylamine In acetone at 0℃; for 0.5h; Stage #2: 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine In acetone at 0 - 20℃; | 94.19% |
| With potassium carbonate In tetrahydrofuran for 24h; Reflux; | 92.8% |
| In tetrahydrofuran for 48h; Reflux; | 82% |
| With sodium hydride In tetrahydrofuran at 65℃; for 48h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 80℃; | 93.6% |
| With caesium carbonate In tetrahydrofuran for 16h; Reflux; Schlenk technique; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; Glovebox; Darkness; | 92% |

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| With ammonia at 20℃; for 1.5h; | 92% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
107-11-9
1-amino-2-propene

-
-
986-11-8
hexakis(allylamino)cyclotriphosphazene

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 4h; Inert atmosphere; Reflux; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran at 20 - 60℃; for 23h; | 91% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
7664-41-7
ammonia

-
A
-
10535-05-4
2,2-diamino-4,4,6,6-tetrachlorocyclotriphosphazene

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 1.5h; Solvent; | A 1% B 91% |
| In diethyl ether at 20℃; for 1.5h; Solvent; | A 80% B 3% |


| Conditions | Yield |
|---|---|
| With caesium carbonate In tetrahydrofuran at 20℃; for 16h; Schlenk technique; Inert atmosphere; | 91% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
2752-17-2
2,2'-diaminodiethyl ether

-
-
129577-49-7
1,9,11,11,13,21,23,23-Octachloro-5,17-dioxa-2,8,10,12,14,20,22,24,25,26-decaaza-1λ5,9λ5,11λ5,13λ5,21λ5,23λ5-hexaphospha-tricyclo[19.3.1.19,13]hexacosa-1(25),9,11,13(26),21,23-hexaene

| Conditions | Yield |
|---|---|
| In diethyl ether; water Ambient temperature; | 90% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
929-59-9
3,6-dioxa-1,8-diaminooctane

-
-
129577-50-0
1,12,14,14,16,27,29,29-Octachloro-5,8,20,23-tetraoxa-2,11,13,15,17,26,28,30,31,32-decaaza-1λ5,12λ5,14λ5,16λ5,27λ5,29λ5-hexaphospha-tricyclo[25.3.1.112,16]dotriaconta-1(31),12,14,16(32),27,29-hexaene

| Conditions | Yield |
|---|---|
| In diethyl ether; water Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| In dichloromethane (under vac.); AlCl3 reacted with (PCl2N)3 in CH2Cl2; | 90% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
23783-42-8
tetraethylene glycol monomethyl ether

-
-
1289636-24-3
2,2,4,4,6,6-hexakis-(2-(2-(2-(2-methoxyethoxy)ethoxy)ethoxy)ethoxy)cyclotriphosphazene

| Conditions | Yield |
|---|---|
| Stage #1: tetraethylene glycol monomethyl ether With sodium hydride In 1,4-dioxane at 50℃; for 24h; Inert atmosphere; Stage #2: 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine In 1,4-dioxane at 60℃; for 48h; Inert atmosphere; | 90% |
-
-
7647-01-0
hydrogenchloride

-
-
7446-70-0, 7784-13-6
aluminum (III) chloride

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| In chloroform at 21.84℃; Schlenk technique; Glovebox; | 90% |

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
134177-52-9
2-[tricarbonyl(η(6)-phenoxy)chromium]ethanol

-
-
7646-69-7
sodium hydride

-
-
134208-38-1
N3P3{(η6-OCH2CH2OC6H5)Cr(CO)3}6

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (N2), added at 0. degree.C, warmed to 25°C and stirred for 5 h, added dropwise N3P3Cl6, stirred at 40-45°C for 12 h; removed solvent in vac., column chromy.; elem. anal., IR, mass. spectra,NMR; | 89% |

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
64275-73-6
(Z)-oct-5-en-1-ol

-
-
1289636-29-8
2,2,4,4,6,6-hexakis-(cis-5-octenyl-1-oxy)cyclotriphosphazene

| Conditions | Yield |
|---|---|
| Stage #1: (Z)-oct-5-en-1-ol With sodium hydride In 1,4-dioxane at 50℃; for 24h; Inert atmosphere; Stage #2: 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine In 1,4-dioxane at 60℃; for 48h; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran at 20 - 60℃; for 13.5h; | 89% |

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| With Cs2CO3 In tetrahydrofuran hydroxyphenyl and Cs2CO3 was added to hexachlorotriphosphazene in THF at0°C, stirred for 6 h at 50°C; concd. in vac., column chromy. (silica, CH2Cl2), evapd. in vac.; | 88% |
-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

-
-
1806-29-7
2,2'-dihydroxybiphenyl

-
-
128175-79-1
4',4',6',6'-tetrachloro-4'λ5,6λ5,6'λ5-spiro[dibenzo[d,f][1–3]-dioxaphosphepine-6,2'-[1–6]triazatriphosphinine]

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 0.25h; | 87% |
| With caesium carbonate In tetrahydrofuran at 20℃; for 10h; | |
| With potassium carbonate |
-
-
918636-17-6
C16H20NPO4

-
-
940-71-6
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 20℃; for 24h; | 87% |

| Conditions | Yield |
|---|---|
| In dichloromethane (under vac.); AlBr3 reacted with (PCl2N)3 in CH2Cl2; | 87% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

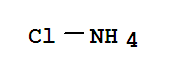














 C
C