-
Name
-
EINECS
231-155-4
-
CAS No.
7440-46-2
- Article Data128
- CAS DataBase
-
Density
1.873 g/mL at 25 °C(lit.)
-
Solubility
reacts
Stability
Flammable solid; highly flammable in powder form. Moisture-sensitive. Incompatible withchlorine, phosphorus, water.
Toxicology
Harmful if swallowed or inhaled and in contact with skin.
-
Melting Point
28.5 °C(lit.)
-
Formula
Cs
-
Boiling Point
705 °C(lit.)
-
Molecular Weight
132.905
-
Flash Point
-
Transport Information
-
Appearance
silvery, light ductile metal
-
Safety
Moderately toxic by intraperitoneal route. Cesium is quite similar to potassium in its elemental state. It has been shown, however, to have pronounced physiological action in experimentation with animals. Hyper-irritability, including marked spasms, has been shown to follow the administration of cesium in amounts equal to the potassium content of the diet. It has been found that replacing the potassium in the diet of rats with cesium caused death after 10–17 days. Ignites spontaneously in air. Violent reaction with water, moisture, or steam releases hydrogen gas which explodes. Violent reaction with acids, halogens, and other oxidizing materials. Incandescent reaction with nonmetals (e.g., sulfur, phosphorus). See also SODIUM.
-
Risk Codes
R14/15;R34;
-
Molecular Structure

-
Hazard Symbols
Dangerous fire and explosion risk, ignites spontaneously in moist air, may explode in contact with sulfur or phosphorus, reacts violently with oxidizing materials, causes burns in contact with skin.
-
Synonyms
Cesium-133
-
PSA
0.00000
-
LogP
0.11250
Synthetic route
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) byproducts: caesium nitride, caesium silicate; CsN3 was decomposed at 390 °C forming Cs and a yellowish gray residue (contains ca. 70% Cs-nitride beside Cs-silicate and not-reacting Cs-azide);; | 90% |
| In neat (no solvent) byproducts: caesium nitride, caesium silicate; CsN3 was decomposed at 390 °C forming Cs and a yellowish gray residue (contains ca. 70% Cs-nitride beside Cs-silicate and not-reacting Cs-azide);; | 90% |
| In neat (no solvent) byproducts: N2; decomposition of CsN3 in high vacuum starting at 290 °C (when Jena glass is used as vessel) forming finely distributed metall;; | |
-
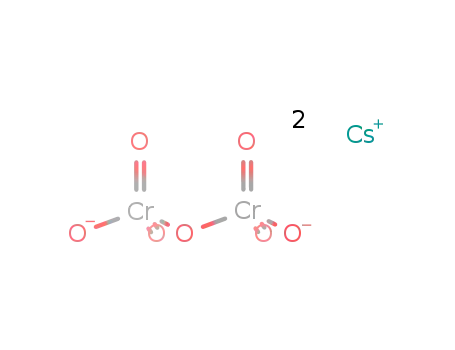
-
cesium dichromate
-
A

-
cesium oxide
Conditions
| Conditions | Yield |
|---|
| With zirconium In neat (no solvent) a mixture of Cs2Cr2O7 and Zr powder (weigt ratio = 1:10) was pressed to rods and heated in high vacuum at 380 °C forming Cs and Cs-oxide;; | A 10%
B 70% |
| With Zr In neat (no solvent) a mixture of Cs2Cr2O7 and Zr powder (weigt ratio = 1:10) was pressed to rods and heated in high vacuum at 380 °C forming Cs and Cs-oxide;; | A 10%
B 70% |
| With zirconium In neat (no solvent) a mixture of Cs2Cr2O7 and Zr powder (weight ratio = 1:20 and 1:40) was pressed to rods and heated in high vacuum at 500 °C forming Cs only;; | A 0%
B n/a |
| With Zr In neat (no solvent) a mixture of Cs2Cr2O7 and Zr powder (weight ratio = 1:20 and 1:40) was pressed to rods and heated in high vacuum at 500 °C forming Cs only;; | A 0%
B n/a |
-
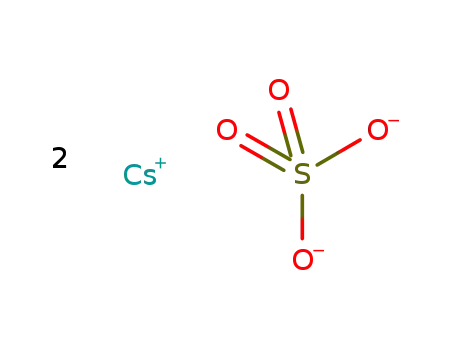
-
cesium sulfate
-
A

-
cesium oxide
-
B

-
cesium sulfide
Conditions
| Conditions | Yield |
|---|
| With zirconium In neat (no solvent) a mixture of Cs2SO4 and Zr powder (weight ratio = 1:4) was pressed to rods and heated in high vacuum at 550 °C forming Cs, oxide and sulfide, reaction explosive;; | A 15-25
B 10%
C 50% |
| With Zr In neat (no solvent) a mixture of Cs2SO4 and Zr powder (weight ratio = 1:4) was pressed to rods and heated in high vacuum at 550 °C forming Cs, oxide and sulfide, reaction explosive;; | A 15-25
B 10%
C 50% |
| With zirconium In neat (no solvent) a mixture of Cs2SO4 and Zr powder (weight ratio = 1:10) was pressed to rods and heated in high vacuum at 500-600 °C forming Cs only; quiet reaction at slow heating, at rapid heating explosion;; | A 0%
B 0%
C n/a |
| With Zr In neat (no solvent) a mixture of Cs2SO4 and Zr powder (weight ratio = 1:10) was pressed to rods and heated in high vacuum at 500-600 °C forming Cs only; quiet reaction at slow heating, at rapid heating explosion;; | A 0%
B 0%
C n/a |
Conditions
| Conditions | Yield |
|---|
| With iron In neat (no solvent) Cs2CO3 was heated with excess of Fe at 1000 °C for 1 h forming metallic Cs; the evolution of gas started at 650 °C and increased up to 1000°C;; the obtained metal is partially oxidized;; | 50% |
| With iron In neat (no solvent) byproducts: Fe2O3, CO, CO2; Cs2CO3 was heated with an excess of Fe in vacuum at 650-1000 °C forming metallic Cs, after 1 h heating almost 50% of the metal was reduced;; | 50% |
| With Fe In neat (no solvent) Cs2CO3 was heated with excess of Fe at 1000 °C for 1 h forming metallic Cs; the evolution of gas started at 650 °C and increased up to 1000°C;; the obtained metal is partially oxidized;; | 50% |
-
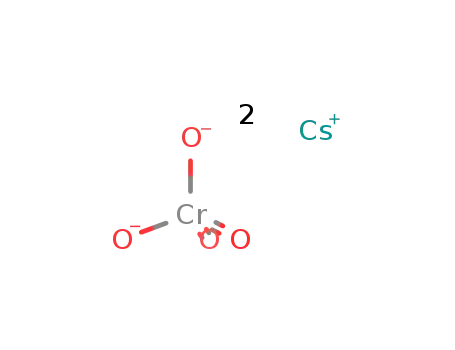
-
cesium chromate
-
A

-
cesium oxide
Conditions
| Conditions | Yield |
|---|
| With zirconium In neat (no solvent) a mixture of Cs2CrO4 and Zr powder (weight ratio = 1:2) was pressed to rods and heated in high vacuum at 725 °C forming Cs and Cs-oxide, inflammation;; | A 10%
B 50% |
| With Zr In neat (no solvent) a mixture of Cs2CrO4 and Zr powder (weight ratio = 1:2) was pressed to rods and heated in high vacuum at 725 °C forming Cs and Cs-oxide, inflammation;; | A 10%
B 50% |
| With zirconium In neat (no solvent) a mixture of Cs2CrO4 and Zr powder (weight ratio = 1:4) was pressed to rods and heated in high vacuum at 725-1000 °C forming Cs;; | A 0%
B 90-96 |
| With Zr In neat (no solvent) a mixture of Cs2CrO4 and Zr powder (weight ratio = 1:4) was pressed to rods and heated in high vacuum at 725-1000 °C forming Cs;; | A 0%
B 90-96 |
Conditions
| Conditions | Yield |
|---|
| iron In neat (no solvent) byproducts: Cs3N; thermal decompn. (380°C); extensive description of apparatus, handling and materials given; detailed discussion of conditions and materials; safe and simple high purity synthesis;; removal of N2 (vac. below 1E-4 Torr); distillation in closed system; identification/purity by atomic absorption spectroscopy;; | |
| copper In neat (no solvent) byproducts: Cs3N; thermal decompn. (380°C); extensive description of apparatus, handling and materials given; detailed discussion of conditions and materials; safe and simple high purity synthesis;; removal of N2 (vac. below 1E-4 Torr); distillation in closed system; identification/purity by atomic absorption spectroscopy;; | |
| In neat (no solvent) byproducts: Cs3N; thermal decompn. (580°C); extensive description of apparatus, handling and materials given; detailed discussion of conditions and materials; safe and simple high purity synthesis;; removal of N2 (vac. below 1E-4 Torr); distillation in closed system; identification/purity by atomic absorption spectroscopy;; | |
-
-
oxygen
-
A
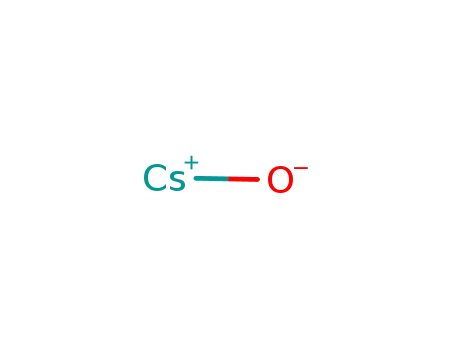
-
cesium monoxide radical
Conditions
| Conditions | Yield |
|---|
| In gas molecular Cs2 beam generated by expansion of alkali vapor through a nozzle crossed with a beam of ground state oxygen atoms; monitoring by chemiluminescence or induced fluorescence; | |
-

-
caesium bromide
Conditions
| Conditions | Yield |
|---|
| Kinetics; Ar-carrier gas, at 2800 K and 4000 K; time-resolved absorption; | |
-

-
caesium bromide
Conditions
| Conditions | Yield |
|---|
| With calcium In neat (no solvent) reduction of CsBr with Ca;; | |
| With Ca In neat (no solvent) reduction of CsBr with Ca;; | |
-

-
cesium chloride
Conditions
| Conditions | Yield |
|---|
| With barium azide In not given CsCl was dissolved in a 16% BaN3-soln.; Cs formation started at 250 °C, the reaction was completed at 350 °C;; | |
| With calcium In neat (no solvent) reduction of CsCl with Ca in a large excess, the temp. must be increased very slowly to 700 °C after 3-4 h, reaction (evolution of blue vapor) starts at 500 °C;; distillation at 300 °C; | |
| With Na or coal In neat (no solvent) CsCl was heated with Na or coal in a N2 or H2 atmosphere forming Cs-vapor;; | |
-

-
cesium chloride
-
B
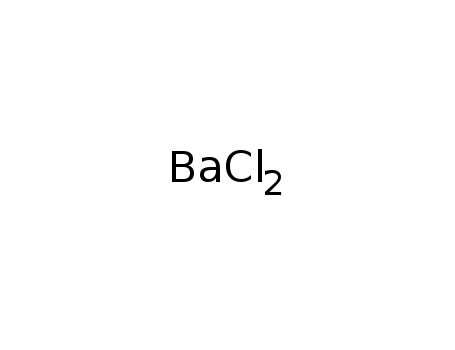
-
barium(II) chloride
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) | |
-

-
cesium chloride
Conditions
| Conditions | Yield |
|---|
| at 600-850°C in vacuo; | |
| In not given vac.; | |
| at 600-850°C in vacuo; | |
-
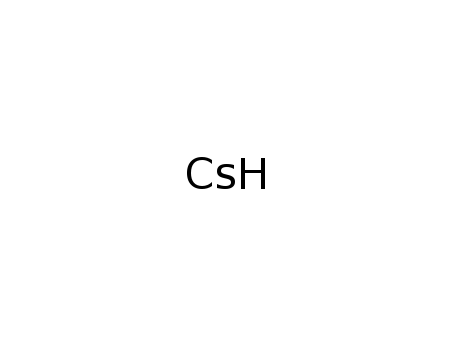
-
caesium hydride
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) byproducts: H2; dissociation of CsH on heating in vacuum at temp. below 300 °C forming liquid Cs;; | |
| In neat (no solvent) byproducts: H2; complete decomposition on heating in vacuum at temp. below 300 °C forming liquid Cs metal;,; | >99 |
| In neat (no solvent) byproducts: H2; dissociation pressures of CsH in dependencc on temp. between 190 and 330 °C;; | |
-

-
cesium iodide
-
A

-
iodine
Conditions
| Conditions | Yield |
|---|
| Kinetics; Ar-carrier gas, at 4000 K and <3000 K; time-resolved absorption; | |
| In neat (no solvent) Irradiation (UV/VIS); at 3800Å;; | |
-

-
cesium dichromate
Conditions
| Conditions | Yield |
|---|
| With Zr or Hf or Ti or Th In neat (no solvent) Cs2Cr2O7 was sealed in a capsule of the reduction agent which was then heated in a discharge tube;; | |
| With zirconium In neat (no solvent) a mixture of Cs2Cr2O7 and Zr powder (weight ratio = 1:4) was pressed to pellets and heated in high vacuum at 320 °C forming Cs, reaction explosive;; | |
| With zirconium In neat (no solvent) pressed powder mixture (Cs2Cr2O7:Zr = 1:10), heating in vac. to 380°C; mostly explosive reaction, with great excess of Zr smoothy;; further products;; | |
-

-
caesium(1+)
Conditions
| Conditions | Yield |
|---|
| Kinetics; at 3000 K; time- resolved absorption; | |
-

-
caesium(1+)
-
A

-
14700-96-0, 14835-24-6, 15723-23-6, 16547-50-5, 16885-04-4, 16887-00-6, 16904-11-3, 16904-12-4, 16999-02-3, 20337-89-7, 20499-73-4, 22537-15-1, 22537-27-5, 22541-73-7, 22541-74-8, 24203-47-2, 33944-49-9, 34937-26-3, 37352-90-2
chlorine
Conditions
| Conditions | Yield |
|---|
| Kinetics; at 3000 K; time- resolved absorption; | |
-

-
caesium(1+)
-
A

-
iodine
Conditions
| Conditions | Yield |
|---|
| Kinetics; at 3000 K; time- resolved absorption; | |
-

-
caesium(1+)
Conditions
| Conditions | Yield |
|---|
| In water Electrolysis; deposition potential in neutral, weakly acidic and in alkaline soln. at room temp.: -2.13 V (vers. normal calomel electrode);; polarography;; | |
| With iron In neat (no solvent) reduction of Cs-salts with Fe;; | |
| With Fe In neat (no solvent) reduction of Cs-salts with Fe;; | |
| In water Electrolysis; deposition potential in neutral, weakly acidic and in alkaline soln. at room temp.: -2.13 V (vers. normal calomel electrode);; polarography;; | |
-

-
cesium sulfate
Conditions
| Conditions | Yield |
|---|
| With zirconium In neat (no solvent) pressed powder mixture (Cs2SO4:Zr = 1:4), heating in vac. to 550°C; mostly explosive reaction, with great excess of Zr smoothy;; further products;; | |
| With iron In neat (no solvent) reduction of Cs2SO4 with Fe started at 700-800 °Cs; rapid reaction at 1000 °C;; the obtained metal is partially oxidized;; | |
| With Zr or Hf or Ti or Th In neat (no solvent) Cs2SO4 was sealed in a capsule of the reduction agent which was then heated in a discharge tube;; | |
-

-
cesium sulfate
-
A
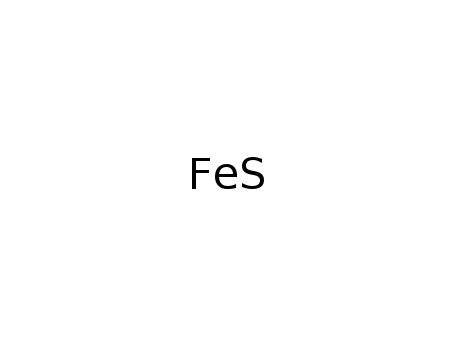
-
iron sulfide
-
B

-
iron(III) oxide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) in vacuum; high temp.;; | |
| In neat (no solvent) in vacuum; high temp.;; | |
Conditions
| Conditions | Yield |
|---|
| At a moderate starting temperature.; | |
| 0% |
| At a moderate starting temperature.; | |
| 0% |
-

-
cesium sulfide
-
A

-
nickel(II) sulfide
Conditions
| Conditions | Yield |
|---|
| Ni-powder in vac.; | |
| Ni-powder in vac.; | |
-
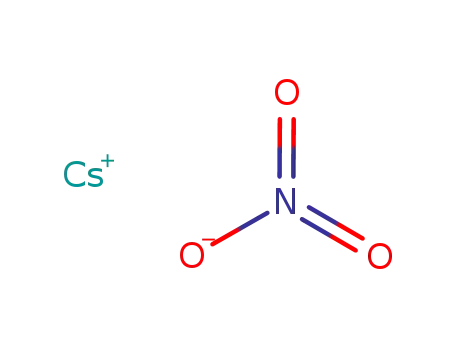
-
caesium nitrate
-
A

-
iron(III) oxide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) reaction under reduced pressure, no quantitative reaction;; | |
-
-
cesium hydroxide
-
A

-
iron(III) oxide
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) reaction in vacuum at 500 - 650°C;; | |
-
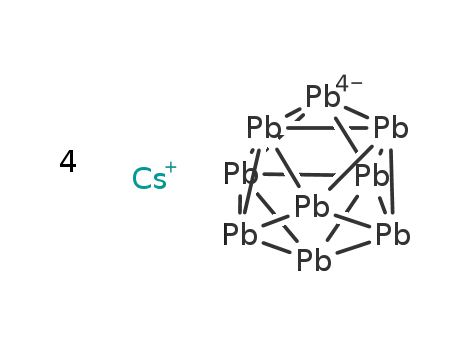
-
4Cs(1+)*Pb9(4-)=Cs4Pb9
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) stoich. amts., 650°C, 2 d; cooling to room temp. at 4°C/h; | 100% |
| With Hg In neat (no solvent) molar ratio Cs:Pb:Hg=4:8.5:12, 650°C, 12 h; cooling to room temp.at 5°C/h; | |
Conditions
| Conditions | Yield |
|---|
| In diethyl ether byproducts: H2; Ar atm.; cooling (0°C), excess of HCN; evapn. (vac.); | 100% |
| In benzene reaction of metallic Cs with an anhydrous soln. of HCN in benzene forming CsCN;; filtration under exclusion of air, removal of benzene in vacuum;; | |
| In benzene reaction of metallic Cs with an anhydrous soln. of HCN in benzene forming CsCN;; filtration under exclusion of air, removal of benzene in vacuum;; | |
-
-
bismuth(III) telluride
-
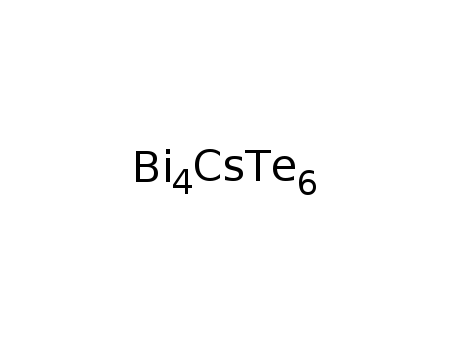
-
CsBi4Te6
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) under N2; Cs and Bi2Te3 (molar ratio 1:1.7) loaded separately in H-shaped tube; sealed (< 1E-4 Torr); heated to 250°C over 24 h; isothermed for 1 d; heated to 580°C over 24 h; held for 2 d; cooled to 50°C in 12 h; washed with degassed MeOH under N2; detd. by X-ray powder diffraction, energy dispersive spectroscopy and scanning electron microscopy; | 100% |
| In melt under N2; Cs and Bi2Te3 (molar ratio 1:1.9) mixed; sealed (< 1E-4 Torr);heated at 250°C for 24 h; melted slowly in flame torch for a min ; quenched in air; detd. by X-ray powder diffraction, energy dispersive spectroscopy and scanning electron microscopy; | 99% |
| vapor transport react.; | |
-

-
bis(tetra-n-butylammonium) dodecahydro-closo-dodecaborate
-
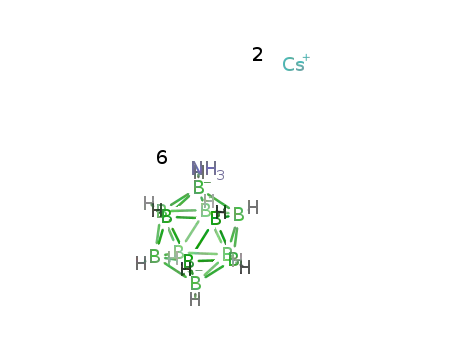
-
B12H12(2-)*2Cs(1+)*6H3N
Conditions
| Conditions | Yield |
|---|
| at -78 - -38℃; for 192h; Inert atmosphere; | 100% |
-
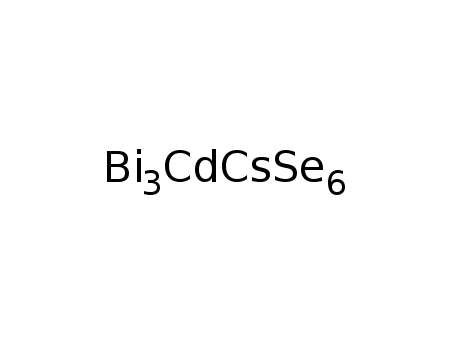
-
CsCdBi3Se6
Conditions
| Conditions | Yield |
|---|
| In melt mixt. heated at 750°C for 2 h with rocking in evacuated silica tubes; cooled to 550°C (20°/h), then to room temp. in 10 h; | 99% |
-

-
Bi3CsSe5, β
Conditions
| Conditions | Yield |
|---|
| In melt mixt. heated at 750°C for 2 h with rocking in evacuated silica tubes; cooled to 550°C (20°/h), then to room temp. in 10 h; | 99% |
-
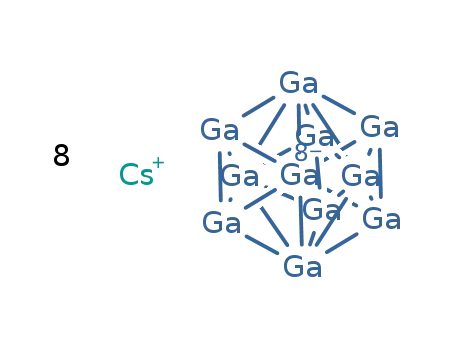
-
8Cs(1+)*Ga11(8-)=Cs8Ga11
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) absence of air and moisture; stoich. amts., welded Ta-container, 700°C, 24 h, cooling to room temp. at 3°C/h; | 99% |
-

-
caesium selenide
Conditions
| Conditions | Yield |
|---|
| In ammonia NH3 (liquid); N2-atmosphere; addn. of 0.5 equiv. of Se to Cs soln. (-78°C), stirring for 1 h; slow warming to room temp., drying (vac., overnight), drying in flame; | 98% |
| 300-330°C; vac.; | |
| In neat (no solvent) metallic Cs was treated slowly with Se-vapor in vacuum under shaking and knocking, the excess of Cs was distilled of at 200 °C; no formation of caesium polyselenide;; | |
-
![[Cs4(H2O)4][ortho-sulfidostannate]](//file1.lookchem.com/cas/reactions/2021/08/20/18812834.png_ms)
-
[Cs4(H2O)4][ortho-sulfidostannate]
Conditions
| Conditions | Yield |
|---|
| In melt (Ar or N2); heating a mixt. of metals in quartz ampule at 700°C for 10 min in vac., cooling to room temp., addn. of S, melting for 20 minin vac., grinding, mechanical sepn. of tin excess, dissolving in water at 20°C, stirring for 5 min; filtration, evapn. for 24 h; | 96% |
-
B
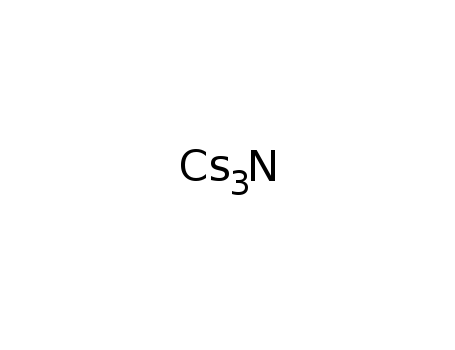
-
caesium nitride
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) reaction of Cs with activated N2 (electric discharge) with formation of CsN3 and some Cs3N;; | A 95%
B n/a |
| In neat (no solvent) reaction of Cs with activated N2 (electric discharge) with formation of CsN3 and some Cs3N;; | A 95%
B n/a |
-

-
tantalum
-
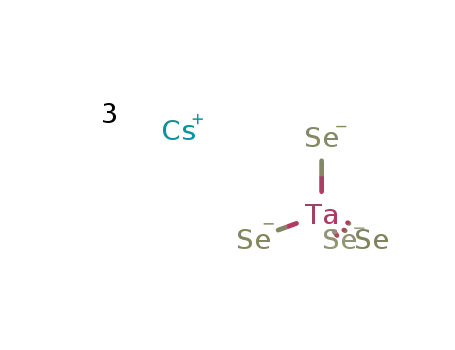
-
3Cs(1+)*TaSe4(3-)=Cs3TaSe4
Conditions
| Conditions | Yield |
|---|
| liquid Cs slowly added to mixt. of Ta and Se under Ar; tube sealed under vacuum (1E-4 Torr); heated in furnace at 800°C for 8 days;; purity determined by X-ray powder diffraction; chemical anal.; | 95% |
| silica tube under an Ar atmosphere, sealed under vac., heated at 800°C for 8 days; elem. anal.; | 95% |
-
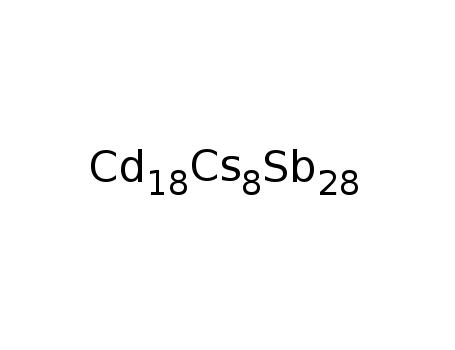
-
Cs8Cd18Sb28
Conditions
| Conditions | Yield |
|---|
| In melt stoich. amounts of Cs, Cd and Sb heated at 550°C for 10 h in welded niobium containers under vac., cooled (5°C/h); elem. anal. (ICP-OES); | 95% |
-
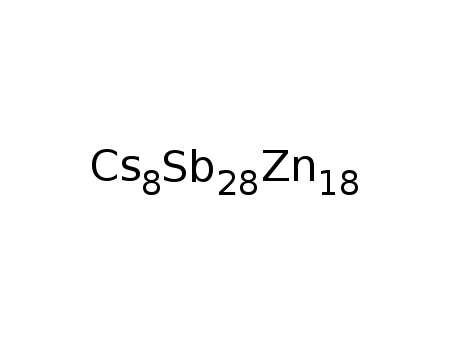
-
Cs8Zn18Sb28
Conditions
| Conditions | Yield |
|---|
| In melt stoich. amounts of Cs, Zn and Sb heated at 550°C for 10 h in welded niobium containers under vac., cooled (5°C/h); elem. anal. (ICP-OES); | 95% |
-
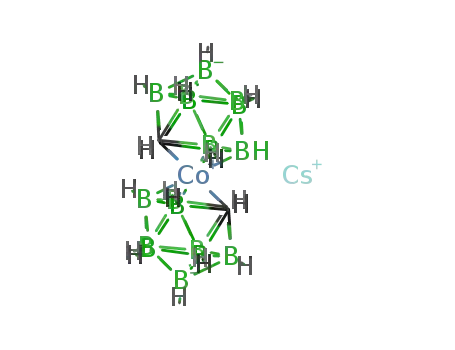
-
caesium cobalt(III)bis(1,2-dicarboilide)
-
![Cs2[Co(1,2-C2B9H11)2] * 0.9 DME](//file1.lookchem.com/cas/reactions/2021/08/20/16714300.png_ms)
-
Cs2[Co(1,2-C2B9H11)2] * 0.9 DME
Conditions
| Conditions | Yield |
|---|
| In 1,2-dimethoxyethane inert atmosphere; slight excess carbollide, warming and stirring for 15 min; crystn. (room temp., 3 d), collection (filtration), drying (vac.); elem.anal.; | 91% |
-
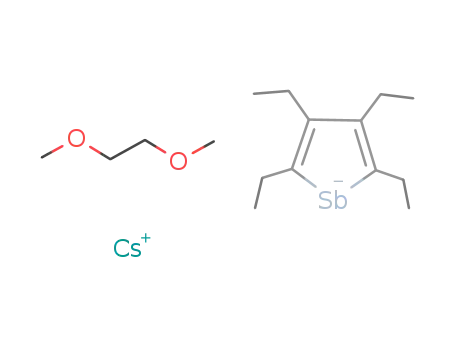
-
DME-cesium 2,3,4,5-tetraethylstibolide
Conditions
| Conditions | Yield |
|---|
| In 1,2-dimethoxyethane under Ar; Sb compd. dissolved in DME; Cs (2 equiv.) added; stirred at room temp. for 2 d; solids removed; vol. of the soln. reduced; crystd. at -20°C; elem. anal.; | 87% |
-
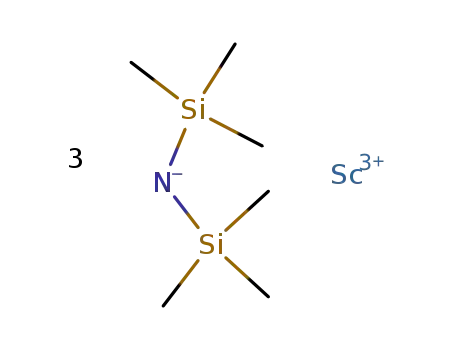
-
tris(bistrimethylsilylamine)scandium(III)
-
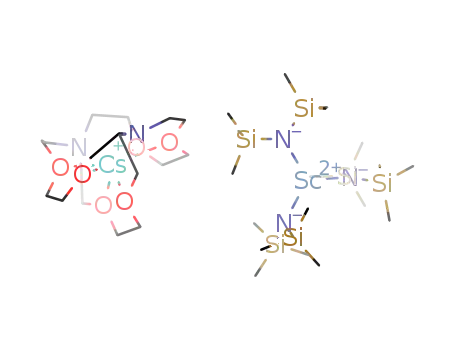
-
C18H54N3ScSi6(1-)*C18H36CsN2O6(1+)
Conditions
| Conditions | Yield |
|---|
| In diethyl ether at -35℃; Inert atmosphere; Schlenk technique; Glovebox; | 83% |
-
![[(CH3In)5(OCH2C6H5)8(O)]](//file1.lookchem.com/cas/reactions/2021/08/20/17599562.png_ms)
-
[(CH3In)5(OCH2C6H5)8(O)]
-
![[Cs(Cs(C4H8O))((CH3In(OCH2C6H5)2)4O)]](//file1.lookchem.com/cas/reactions/2021/08/20/17600258.png_ms)
-
[Cs(Cs(C4H8O))((CH3In(OCH2C6H5)2)4O)]
Conditions
| Conditions | Yield |
|---|
| In tetrahydrofuran; toluene Sonication; all manipulations under Ar atm.; to stirred mixt. of Cs and toluene added complex dropwise, stirred for 3 h by ultrasonication, refluxed for 24h, solvent evapd., added THF; filtered, added TMEDA, allowed to stand for 1 wk at 20°C, elem. anal.; | 81% |
-

-
praseodymium
-
![Pr5(11+)*C2(2-)*9Cl(1-)=[Pr5C2]Cl9](//file1.lookchem.com/cas/reactions/2021/08/20/16633096.png_ms)
-
Pr5(11+)*C2(2-)*9Cl(1-)=[Pr5C2]Cl9
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) byproducts: CsCl; Ar atm.; 900°C, 4 h; 800°C, 30 d; | 80% |
-
-
99685-96-8, 161105-99-3, 161106-00-9, 111138-12-6, 133318-63-5, 134053-11-5, 134931-35-4, 134931-36-5, 139703-76-7, 145633-27-8, 175414-73-0, 175414-74-1, 175414-75-2, 175519-12-7, 175519-13-8, 175519-14-9, 175519-15-0, 136376-46-0, 144906-37-6, 144906-38-7, 151767-00-9, 152882-97-8, 152882-98-9, 152882-99-0, 153062-34-1, 154171-74-1, 154171-75-2, 154333-99-0, 154334-00-6, 154397-63-4, 154460-59-0, 199456-56-9, 108739-25-9, 120329-57-9, 120329-58-0
fullerene-C60
-
-
cobalt(II) bromide
-
![[(fullerene)2Co(II)(dimethylformamide)3.3]](//file1.lookchem.com/cas/reactions/2021/08/20/19692530.png_ms)
-
[(fullerene)2Co(II)(dimethylformamide)3.3]
Conditions
| Conditions | Yield |
|---|
| In N,N-dimethyl-formamide under anaerobic conditions; C60 and Cs were dissolved at 50°C, after 6 h soln. was cooled to ca. -20°C, intermedite indicated by IR, to this soln. metal salt was added, stirring for 4 h at ca. -20°C; ppt. was filtered off, washed with MeCN, dried at ca. -20°C; elem. anal.; | 80% |
| In benzonitrile; acetonitrile under anaerobic conditions; C60 and Cs were dissolved in benzonitrile at50°C, after 4 h soln. was cooled to ca. -20°C, intermedit e indicated by IR, soln. metal salt in MeCN was added, intermediate was filtered off and soln. was bleached; ppt. was filtered off, washed with MeCN, dried; elem. anal.; | |
-

-
nickel(II) iodide
-

-
99685-96-8, 161105-99-3, 161106-00-9, 111138-12-6, 133318-63-5, 134053-11-5, 134931-35-4, 134931-36-5, 139703-76-7, 145633-27-8, 175414-73-0, 175414-74-1, 175414-75-2, 175519-12-7, 175519-13-8, 175519-14-9, 175519-15-0, 136376-46-0, 144906-37-6, 144906-38-7, 151767-00-9, 152882-97-8, 152882-98-9, 152882-99-0, 153062-34-1, 154171-74-1, 154171-75-2, 154333-99-0, 154334-00-6, 154397-63-4, 154460-59-0, 199456-56-9, 108739-25-9, 120329-57-9, 120329-58-0
fullerene-C60
-
![[(fullerene)2Ni(II)(dimethylformamide)4]](//file1.lookchem.com/cas/reactions/2021/08/20/19692526.png_ms)
-
[(fullerene)2Ni(II)(dimethylformamide)4]
Conditions
| Conditions | Yield |
|---|
| In N,N-dimethyl-formamide under anaerobic conditions; C60 and Cs were dissolved at 50°C, after 6 h soln. was cooled to ca. -20°C, intermedite indicated by IR, to this soln. metal salt was added, stirring for 4 h at ca. -20°C; ppt. was filtered off, washed with MeCN, dried at ca. -20°C; elem. anal.; | 80% |
-
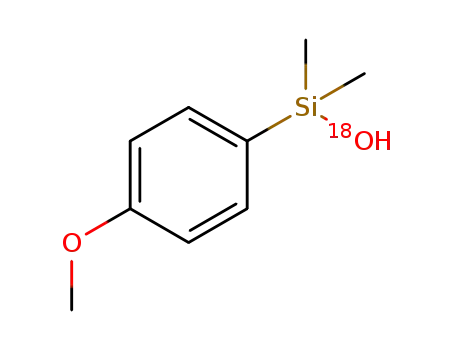
-
C9H14O(18)OSi
-
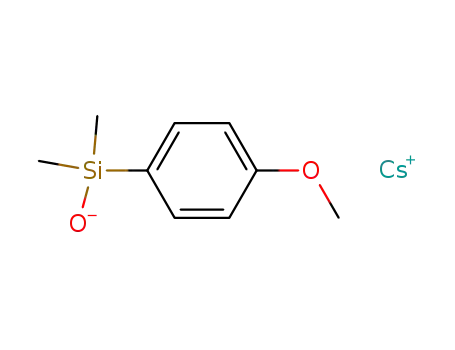
-
cesium (4-methoxyphenyl)dimethylsilanolate
Conditions
| Conditions | Yield |
|---|
| In benzene for 0.333333h; | 79% |
-
![Cs[{U(OSi(tert-butoxy)3)3}2(μ-N)]](//file1.lookchem.com/cas/reactions/2021/08/20/29414842.png_ms)
-
Cs[{U(OSi(tert-butoxy)3)3}2(μ-N)]
-
![Cs3[{U(OSi(tert-butoxy)3)3}2(μ-N)]](//file1.lookchem.com/cas/reactions/2021/08/20/29414844.png_ms)
-
Cs3[{U(OSi(tert-butoxy)3)3}2(μ-N)]
Conditions
| Conditions | Yield |
|---|
| In tetrahydrofuran at -40℃; for 3h; Inert atmosphere; Glovebox; | 77% |
-
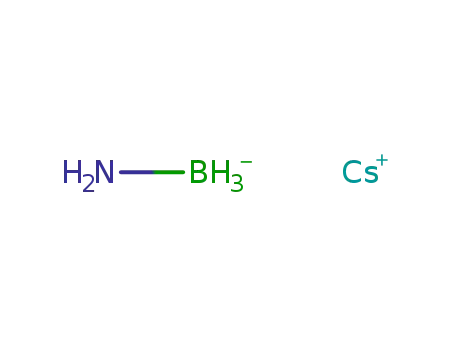
-
Cs(1+)*BH5N(1-)
Conditions
| Conditions | Yield |
|---|
| In tetrahydrofuran at 0℃; Inert atmosphere; | 77% |
-
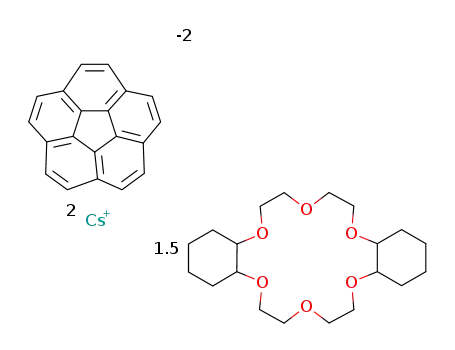
-
C20H10(2-)*1.5C20H36O6*2Cs(1+)
Conditions
| Conditions | Yield |
|---|
| In diethylene glycol dimethyl ether at 20℃; for 24h; Glovebox; Inert atmosphere; | 75% |
-
A
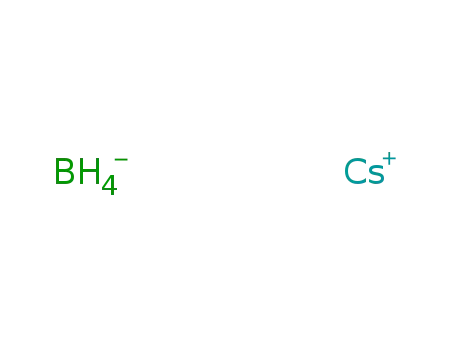
-
cesium borohydride
-
B
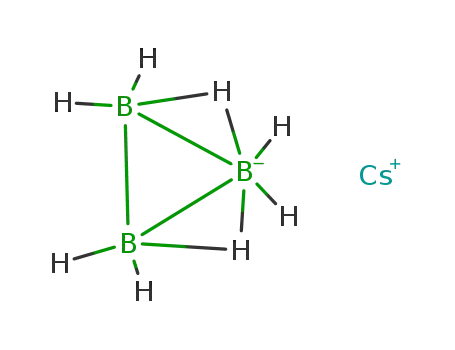
-
cesium octahydrotriborate
Conditions
| Conditions | Yield |
|---|
| at 20℃; for 2h; Schlenk technique; Inert atmosphere; | A n/a
B 73% |
-

-
caesium hydride
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) Special reactor, Cs metal, 1.6 equiv of H2 gas, 450-550°C, 44 h;; | 72% |
| With n-butyllithium; N,N,N,N,-tetramethylethylenediamine In hexane; tert-butyl alcohol addn. of t-BuOH to suspn. of Cs in hexane, diln. with hexane (stirring, 1 h), H2 atmosphere, addn. of tetramethylethylenediamine (-20°C), dropwise addn. of n-BuLi in hexane (-40°C), temp. raised to 5°C (under H2), stirred (1 h); only in soln.; yield was detected by measurement the volume of liberated H2 after decompn. with methanol;; | 67% |
| Irradiation (UV/VIS); heating (420°C, 20 torr), 457,9 nm argon laser illumination; | |
-
![La5(11+)*C2(2-)*9Cl(1-)=[La5C2]Cl9](//file1.lookchem.com/cas/reactions/2021/08/20/16633097.png_ms)
-
La5(11+)*C2(2-)*9Cl(1-)=[La5C2]Cl9
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) byproducts: CsCl; Ar atm.; 900°C, 4 h; 800°C, 40 d; | 70% |
Cesium Chemical Properties
Molecular structure:

IUPAC Name: Cesium
Synonyms of Cesium (CAS NO.7440-46-2): Cesium-133 ; Cesium Aa Single Element Standard ; Cesium, Aas Standard Solution ; Cesium Aa Standard ; Cesium Atomic Absorption Standard ; Cesium Atomic Spectroscopy Standard ; Cesium ; Cesium Metal
Product Categories: Alkali Metals Metal and Ceramic Science ; Cesium ; Metals ; Reduction ; Synthetic Reagents
Molecular Formula: Cs
Molecular Weight: 132.91
EINECS: 231-155-4
Density: 1.873 g/mL at 25 °C
Melting Point: 28.5 °C
Boiling Point: 705 °C
Vapor pressure: 1 mm Hg ( 279 °C)
Storage temp.: 2-8°C
Solubility in H2O: soluble
Merck: 13,2018
Stability: Flammable solid; highly flammable in powder form. Moisture-sensitive. Incompatible with chlorine, phosphorus, water.
Cesium History
Cesium (CAS NO.7440-46-2) (Latin caesius meaning "bluish gray") was spectroscopically discovered by Robert Bunsen and Gustav Kirchhoff in 1860 in mineral water from Dürkheim, Germany, as described below. Carl Setterberg first produced caesium metal in 1882 by electrolysis of caesium chloride.Setterberg received his PhD from Kekule and Bunsen for this work.
Historically, the most important use for caesium has been in research and development, primarily in chemical and electrical fields. It found no significant application until its was added into radio vacuum tubes in the 1920s as a getter, a scavenger of the trace amounts of oxygen remaining in the tube after manufacture, and as a coating on the heated cathode to increase the amount of electric current that could flow through the tube. Caesium became recognized as a functional, high-performance industrial metal in electronics in the 1950s.
Since 1967, the International System of Measurements has based its unit of time, the second, on the properties of caesium. The International System of Units (SI) defines the second as 9,192,631,770 cycles of the radiation, which corresponds to the transition between two hyperfine energy levels of the ground state of the 133Cs atom.
Cesium Production
Raw materials is Argon .
Most of the mined Cesium (CAS NO.7440-46-2) is directly converted into caesium formate (HCOO−Cs+) for applications such as oil drilling. Caesium metal can be isolated by electrolysis of fused caesium cyanide. Exceptionally pure and gas-free caesium can be made by the thermal decomposition of caesium azide. In vacuum applications, caesium dichromate can be reacted with zirconium forming pure caesium without other gaseous products.
Cs2Cr2O7 + 2 Zr → 2 Cs + 2 ZrO2+ Cr2O3 The primary commercial compounds of caesium are caesium chloride and its nitrate.
Cesium Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
| mouse | LD50 | intraperitoneal | 1700mg/kg (1700mg/kg) | | "Structure et Activite Pharmacodyanmique des Medicaments du Systeme Nerveux Vegetatif," Bovet, D., and F. Bovet-Nitti, New York, S. Karger, 1948Vol. -, Pg. 704, 1948. |
Cesium Consensus Reports
Reported in EPA TSCA Inventory.
Cesium Safety Profile
Moderately toxic by intraperitoneal route. Cesium is quite similar to potassium in its elemental state. It has been shown, however, to have pronounced physiological action in experimentation with animals. Hyper-irritability, including marked spasms, has been shown to follow the administration of cesium in amounts equal to the potassium content of the diet. It has been found that replacing the potassium in the diet of rats with cesium caused death after 10–17 days. Ignites spontaneously in air. Violent reaction with water, moisture, or steam releases hydrogen gas which explodes. Violent reaction with acids, halogens, and other oxidizing materials. Incandescent reaction with nonmetals (e.g., sulfur, phosphorus). See also SODIUM.
Hazard Codes: Xi,
Xi, C,
C, F+
F+
Risk Statements of Cesium (CAS NO.7440-46-2):
R36/38:Irritating to eyes and skin.
R34:Causes burns.
R14/15: Reacts violently with water and contact with water liberates extremely flammable gases.
R11:Highly flammable.
Safety Statements:
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S43:In case of fire use ... (there follows the type of fire-fighting equipment to be used.)
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S16:Keep away from sources of ignition.
S8:Keep container dry.
RIDADR: UN 3264 8/PG 3
WGK Germany: 3
RTECS: FK9225000
F 10: Keep under argon.
HazardClass: 4.3
PackingGroup: I
Cesium Standards and Recommendations
DOT Classification: 4.3; Label: Dangerous When Wet
Cesium Specification
Cesium (CAS NO.7440-46-2) is a silvery, light ductile metal. It causes burns to skin and eyes.It is highly flammable.Reacts violently with most acids. Reacts violently with fluorine, chlorine, bromine and iodine. Reacts with incandescence with sulfur and phosphorus. Burns vigorously in air.Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce corrosive solutions on contact with water. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.Produce flammable gases on contact with water. May ignite on contact with water or moist air. Some react vigorously or explosively on contact with water. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished. Some are transported in highly flammable liquids. Runoff may create fire or explosion hazard.





























































































![]()
 Xi,
Xi, C,
C, F+
F+