-
Name
D(+)-2-Octanol
- EINECS 228-213-6
- CAS No. 6169-06-8
- Article Data215
- CAS DataBase
- Density 0.822 g/mL at 25 °C(lit.)
- Solubility 1 g/L (20 °C) in water
- Melting Point -38oC
- Formula C8H18O
- Boiling Point 175 °C(lit.)
- Molecular Weight 130.23
- Flash Point 160 °F
- Transport Information
- Appearance Colorless to light yellow liquid
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms d-Octan-2-ol;octan-2-ol;2-Octanol, (S)-;2-Octanol, (2S)-;(S)-(+)-2-Octanol;D-2-Octanol;
- PSA 20.23000
- LogP 2.33760
Synthetic route
-
-
74403-68-2
(R)-2-octyl sulfate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With His-tagged Pseudomonas sp. metallo-β-lactamase-type alkylsulfatase Pisa1; water; tris hydrochloride at 30℃; for 6h; pH=8.2; Enzymatic reaction; optical yield given as %ee; stereoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With alcohol dehydrogenase from Lactobacillus kefir; alcohol dehydrogenase from Rhodococcus ruber DSM 44541; YcnD-oxidoreductase at 30℃; for 3h; pH=7.5; aq. buffer; Resolution of racemate; Enzymatic reaction; optical yield given as %ee; enantiospecific reaction; | 99% |
| With phosphate buffer; tributyrin for 140h; Ambient temperature; | 77% |

| Conditions | Yield |
|---|---|
| With NAD; Co2+immobilized acetophenone reductase from Geotrichum candidum In aq. phosphate buffer; isopropyl alcohol at 30℃; for 3h; pH=7.2; Green chemistry; Enzymatic reaction; enantioselective reaction; | 99% |
| With glucose dehydrogenase; D-Glucose; alcohol dehydrogenase from Thermoanaerobacter sp.; NADPH In aq. buffer at 30℃; for 24h; pH=7.5; Concentration; Time; Ionic liquid; Green chemistry; Enzymatic reaction; enantioselective reaction; | 85% |
| With acetophenone reductase from Geotrichum candidum NBRC 4597; NAD In isopropyl alcohol at 30℃; for 3h; pH=7.2; Enzymatic reaction; enantioselective reaction; | 85% |
-
-
74403-68-2
(R)-2-octyl sulfate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With His-tagged Pseudomonas sp. metallo-β-lactamase-type alkylsulfatase Pisa1; water; tris hydrochloride at 30℃; for 6h; pH=8.2; Enzymatic reaction; optical yield given as %ee; stereoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With alcohol dehydrogenase from Lactobacillus kefir; alcohol dehydrogenase from Rhodococcus ruber DSM 44541; YcnD-oxidoreductase at 30℃; for 3h; pH=7.5; aq. buffer; Resolution of racemate; Enzymatic reaction; optical yield given as %ee; enantiospecific reaction; | 99% |
| With phosphate buffer; tributyrin for 140h; Ambient temperature; | 77% |

| Conditions | Yield |
|---|---|
| With NAD; Co2+immobilized acetophenone reductase from Geotrichum candidum In aq. phosphate buffer; isopropyl alcohol at 30℃; for 3h; pH=7.2; Green chemistry; Enzymatic reaction; enantioselective reaction; | 99% |
| With glucose dehydrogenase; D-Glucose; alcohol dehydrogenase from Thermoanaerobacter sp.; NADPH In aq. buffer at 30℃; for 24h; pH=7.5; Concentration; Time; Ionic liquid; Green chemistry; Enzymatic reaction; enantioselective reaction; | 85% |
| With acetophenone reductase from Geotrichum candidum NBRC 4597; NAD In isopropyl alcohol at 30℃; for 3h; pH=7.2; Enzymatic reaction; enantioselective reaction; | 85% |
-
-
4128-31-8
rac-octan-2-ol

-
A
-
6169-06-8
(S)-2-Octanol

-
B
-
5978-70-1
(R)-Octan-2-ol

-
C
-
111-13-7
hexyl-methyl-ketone

| Conditions | Yield |
|---|---|
| With Arthrobacter atrocyaneus In N,N-dimethyl-formamide at 32℃; for 48h; Microbiological reaction; enantioselective reaction; | A 94% B n/a C n/a |
| With lyophilised cells of Escherichia coli overexpressing the solvent-tolerant alcohol dehydrogenase from Rhodococcus ruber DSM44541 In aq. buffer at 30℃; for 24h; pH=7.5; Reagent/catalyst; Resolution of racemate; Enzymatic reaction; | A n/a B n/a C n/a |
| With NADP In acetone at 50℃; for 22h; pH=8; Resolution of racemate; |

-
-
111221-79-5
(R)-(+)-2-hydroxyoctyl tosylate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran | 93% |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In methanol for 6h; Reflux; Inert atmosphere; | 87% |
| With potassium hydroxide Heating; | 3.4 g |
-
-
34760-88-8
rac-2-octyl sulfate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| Stage #1: rac-2-octyl sulfate With His-tagged Pseudomonas sp. metallo-β-lactamase-type alkylsulfatase Pisa1; water; tris hydrochloride at 30℃; for 24h; pH=8.2; Resolution of racemate; Enzymatic reaction; Stage #2: With water; toluene-4-sulfonic acid In 1,4-dioxane; tert-butyl methyl ether at 40℃; for 5h; optical yield given as %ee; stereoselective reaction; | 87% |
| Multi-step reaction with 2 steps 1: His-tagged Pseudomonas sp. metallo-β-lactamase-type alkylsulfatase Pisa1; water; tris hydrochloride / 6 h / 30 °C / pH 8.2 / Resolution of racemate; Enzymatic reaction 2: water; toluene-4-sulfonic acid / 1,4-dioxane; tert-butyl methyl ether / 2 h / 40 °C View Scheme |

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; Raney-Ni (W-2) In ethanol; water for 0.5h; Ambient temperature; pH=5.2; | 85% |
| With sodium hypophosphite; Raney Ni(W-2) In ethanol; water for 0.5h; Ambient temperature; | 75% |
-
-
1200233-22-2
(S)-N-(oct-2-yloxy)-4-(p-chlorophenyl)-thiazole-2(3H)-thione

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With tri-n-butyl-tin hydride In dichloromethane at 18℃; for 0.416667h; Inert atmosphere; Photolysis; enantioselective reaction; | 85% |
-
-
140421-08-5
(R)-1-Phenylsulfanyl-octan-2-ol

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; Raney Ni(W-2) In ethanol; water for 0.5h; Ambient temperature; | 84% |
| With sodium hypophosphite; Raney-Ni (W-2) In ethanol; water for 0.5h; Ambient temperature; pH=5.2; | 84% |

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; Raney Ni(W-2) In ethanol; water for 0.5h; Ambient temperature; | 82% |
| With sodium hypophosphite; Raney-Ni (W-2) In ethanol; water for 0.5h; Ambient temperature; pH=5.2; | 82% |

| Conditions | Yield |
|---|---|
| A 77% B 75% |

| Conditions | Yield |
|---|---|
| (1S,2R)-(-)-2-(N,N-di-n-butylamino)-1-phenylpropan-1-ol In hexane 1.) r.t., 20 min; 2.) 0 deg C, 16 h; | 70% |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; (R,R)-dihydroborate In hexane at -20℃; for 48h; Title compound not separated from byproducts; | A n/a B 68% |
| With hydrogen; tartaric acid-NaBr-mod. Raney nickel In tetrahydrofuran at 100℃; for 8h; Product distribution; enantioface hydrogenation with various carboxylic acids; | |
| With hydrogen; TA-NaBr-MRNi In tetrahydrofuran at 60℃; under 66195.7 Torr; for 96h; Product distribution; RNi(prepared from 3.8 g of the alloy(Ni/Al=42/58))modified with 200 ml of aq. solution containing (R,R)-TA(2 g) and NaBr(12 g) at pH 3.2, 100 deg C for 1 h. hydrogenations of var. 2-alkanones, reactions at var. temperatures; |
-
-
4128-31-8
rac-octan-2-ol

-
-
101-41-7
benzeneacetic acid methyl ester

-
A
-
6169-06-8
(S)-2-Octanol

-
B
-
5978-70-1
(R)-Octan-2-ol

-
C
-
92883-26-6
(R)-2-octyl phenylacetate

-
D
-
111-13-7
hexyl-methyl-ketone

| Conditions | Yield |
|---|---|
| With [(C4Ph4COHOCC4Ph4)(μ-H)][(CO)4Ru2]; Novozym(R) 435; 2,4-dimethylpentan-3-one In toluene at 70℃; under 142.511 Torr; for 40h; | A n/a B n/a C 63% D n/a |

-
-
1200233-00-6
(S)-N-(oct-2-yloxy)-pyridine-2(1H)-thione

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With tri-n-butyl-tin hydride In dichloromethane at 18℃; for 0.0333333h; Inert atmosphere; Photolysis; enantioselective reaction; | 59% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B; 1-(10-carboxydecyl)-3-methylimidazolium hexafluorophosphate; diisopropyl-carbodiimide In acetone at 40℃; for 2h; Resolution of racemate; Enzymatic reaction; enantioselective reaction; | A 51% B 35% |
| Title compound not separated from byproducts; | |
| With sodium acetate In water at 40℃; enantioselective reaction; | A n/a B n/a |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B In acetone at 35℃; for 3h; Resolution of racemate; Enzymatic reaction; enantioselective reaction; | A 51% B n/a |

-
-
108-05-4
vinyl acetate

-
-
4128-31-8
rac-octan-2-ol

-
A
-
6169-06-8
(S)-2-Octanol

-
B
-
54712-18-4
(R)-2-octyl acetate

| Conditions | Yield |
|---|---|
| With sol-gel encapsulated Candida antarctica lipase SP 525 at 20℃; for 17h; | A 50% B 47% |
| With 4 A molecular sieve; immobilized lipase PS In di-isopropyl ether at 30℃; for 15h; | A 49% B 48.7% |
| With immobilized lipase B from Candida antarctica In hexane at 32℃; for 1h; Resolution of racemate; Enzymatic reaction; | A 40% B 35% |

-
-
1200233-16-4
(S)-N-(oct-2-yloxy)-4-methylthiazole-2(3H)-thione

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With tri-n-butyl-tin hydride In dichloromethane at 18℃; for 0.416667h; Inert atmosphere; Photolysis; enantioselective reaction; | 50% |
-
-
56772-63-5
(R)-octan-2-yl methanesulfonate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrahydrofuran for 19h; Heating; | 46% |

| Conditions | Yield |
|---|---|
| With Lipase Amano AK at 20℃; for 72h; pH=7; aq. phosphate buffer; Enzymatic reaction; | A 42% B 43% |

| Conditions | Yield |
|---|---|
| With oleic acid hydratase from Elizabethkingia meningoseptica A248L mutant; water; hexanoic acid In dimethyl sulfoxide at 25℃; for 144h; pH=6; Reagent/catalyst; Enzymatic reaction; enantioselective reaction; | 22% |

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| bei der Verseifung; succinic acid methyl ester--ester; |

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| Hydrolysis; malonic acid ethyl ester--ester; |

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| bei der Verseifung; succinic acid ethyl ester--ester; |
-
-
89837-79-6
malonic acid bis-(1-methyl-heptyl ester)

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| Hydrolysis; malonic acid di--ester; |
-
-
29390-06-5
bis(1-methylheptyl)butanedioate

-
-
6169-06-8
(S)-2-Octanol

| Conditions | Yield |
|---|---|
| bei der Verseifung; succinic acid di--ester; |
-
-
6169-06-8
(S)-2-Octanol

-
-
39637-99-5, 20445-33-4
(S)-(+)-2-methoxy-2-trifluoromethyl-2-phenylacetyl chloride

-
-
20445-10-7
(R)-2-octyl (R)-α-methoxy-α-trifluoromethylphenylacetate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 0 - 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 13h; | 99% |
| With pyridine | 93.1% |
| for 1h; Heating; | 82% |
| With dmap at 30℃; for 18h; | |
| With dmap |
-
-
6169-06-8
(S)-2-Octanol

-
-
29117-48-4
(R)-(-)-2-iodooctane

| Conditions | Yield |
|---|---|
| With 1H-imidazole; iodine; triphenylphosphine In dichloromethane for 1h; Ambient temperature; | 98% |
| With iodine In Petroleum ether for 6h; Heating; | 25% |
| With hydrogen iodide |
-
-
6169-06-8
(S)-2-Octanol

-
-
124-63-0
methanesulfonyl chloride

-
-
62820-85-3
(S)-2-<(Methanesulfonyl)oxy>octane

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; | 98% |
| With pyridine for 2h; | 86% |
| With triethylamine In dichloromethane at 15 - 20℃; for 1h; | |
| With pyridine | |
| With pyridine |
-
-
6169-06-8
(S)-2-Octanol

-
-
5394-63-8
2,2,6-trimethyl-4H-1,3-dioxin-4-one

-
-
143913-51-3
(S)-2-octyl acetoacetate

| Conditions | Yield |
|---|---|
| for 0.1h; microwave irradiation; | 98% |
-
-
6169-06-8
(S)-2-Octanol

-
-
142-45-0
Acetylenedicarboxylic acid

-
-
1181217-27-5
bis[(1S)-1-methylheptyl] acetylenedicarboxylate

| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrahydrofuran for 14h; Reflux; | 97% |
-
-
6169-06-8
(S)-2-Octanol

-
-
65-85-0
benzoic acid

-
-
6938-51-8, 78687-05-5, 98819-31-9, 34881-29-3
(S)-benzoic acid 1-methylheptyl ester

| Conditions | Yield |
|---|---|
| With cyanomethylenetributyl-phosphorane In benzene at 100℃; for 24h; | 96% |

| Conditions | Yield |
|---|---|
| With azodicarboxylic acid bis(2-methoxyethyl) ester; triphenylphosphine In toluene at 20℃; Product distribution / selectivity; Mitsunobu reaction; | 96% |
| With azodicarboxylic acid bis(2-methoxyethyl) ester; triphenylphosphine In toluene at 20℃; for 3.5h; Mitsunobu reaction; Inert atmosphere; stereospecific reaction; | 96% |
| With ethyl 2-(3,4-dichlorophenyl)azocarboxylate; triphenylphosphine In toluene at 20℃; for 5h; Mitsunobu Displacement; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; toluene at 0 - 20℃; for 17.5h; Inert atmosphere; | 96% |
-
-
6169-06-8
(S)-2-Octanol

-
-
18651-57-5
(R)-2-chlorooctane

| Conditions | Yield |
|---|---|
| With 1-chloro-1-(dimethylamino)-2-methyl-1-propene In dichloromethane 1.) 0 deg C; 2.) rt., 3h; | 95% |
| With trichloroacetonitrile; triphenylphosphine In acetonitrile for 1h; Ambient temperature; | 70% |
| With diethyl ether; phosphorus trichloride |
-
-
6169-06-8
(S)-2-Octanol

-
-
245743-64-0
methyl 5-chloro-2-fluoro-4-hydroxy-benzoate

-
-
211172-74-6
(R)-5-chloro-2-fluoro-4-(2-octyloxy)benzoate

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In dichloromethane at 25℃; for 2h; Etherification; | 95% |

| Conditions | Yield |
|---|---|
| for 0.333333h; microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With trimethylamine hydrochloride; triethylamine In dichloromethane at 0℃; for 1h; Inert atmosphere; | 94% |
| With trimethylamine hydrochloride; triethylamine In dichloromethane at 0℃; for 2h; Inert atmosphere; | 94% |
| With pyridine at 4℃; Inert atmosphere; | 92% |
-
-
6169-06-8
(S)-2-Octanol

-
-
14180-11-1
4-(methoxycarbonyloxy)benzoic acid

-
-
271786-19-7
4-methoxycarbonyloxybenzoic acid (S)-1-methylheptyl ester

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20℃; for 48h; | 94% |
-
-
6169-06-8
(S)-2-Octanol

-
-
99-76-3
methyl 4-hydroxylbenzoate

-
-
138705-74-5
methyl 4-{[(1R)-1-methylheptyl]oxy} benzoate

| Conditions | Yield |
|---|---|
| Stage #1: (S)-2-Octanol; methyl 4-hydroxylbenzoate With triphenylphosphine In tetrahydrofuran at 0℃; for 0.25h; Mitsunobu reaction; Stage #2: With di-isopropyl azodicarboxylate In tetrahydrofuran at 0 - 20℃; for 8h; Mitsunobu reaction; | 94% |
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran |
-
-
6169-06-8
(S)-2-Octanol

-
-
65-85-0
benzoic acid

-
-
6938-51-8, 34881-29-3, 98819-31-9, 78687-05-5
(R)-benzoic acid 1-methylheptyl ester

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; toluene at 20℃; Product distribution / selectivity; Mitsunobu reaction; | 93% |
| With tributylphosphine; 1,1'-azodicarbonyl-dipiperidine In benzene at 60℃; for 24h; | 91% |
| With azodicarboxylic acid bis(2-methoxyethyl) ester; triphenylphosphine In diethyl ether at 20℃; for 2h; Mitsunobu reaction; Inert atmosphere; stereospecific reaction; | 89% |
-
-
6169-06-8
(S)-2-Octanol

-
-
666829-99-8
N,N-diethyl 4'-bromo-4-methoxybiphenyl-2-carboxamide

-
-
666830-01-9
(R)-N,N-diethyl 4'-bromo-4-(1-methylheptyloxy)biphenyl-2-carboxamide

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In dichloromethane at 25℃; for 4h; Mitsunobu reaction; | 93% |
-
-
6169-06-8
(S)-2-Octanol


| Conditions | Yield |
|---|---|
| In chlorobenzene for 0.5h; microwave irradiation; | 92% |

| Conditions | Yield |
|---|---|
| 92% |
-
-
6169-06-8
(S)-2-Octanol

-
-
586-76-5
4-Bromobenzoic acid

-
-
1160843-08-2
(S)-1-methylheptyl 4-bromobenzoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 92% |
-
-
100-02-7
4-nitro-phenol

-
-
6169-06-8
(S)-2-Octanol

-
-
850903-50-3
(S)-4-(1-methylheptyl)oxy-1-nitrobenzene

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 10 - 20℃; Inert atmosphere; | 91% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 18h; Mitsunobu Displacement; | 91% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In diethyl ether at 0 - 20℃; | 90% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| With triethylsilane; ytterbium(III) triflate In 1,2-dichloro-ethane for 4h; Reflux; | 91% |
| With triethylsilane; iron(III) chloride In nitromethane at 20℃; for 1h; | 81% |

| Conditions | Yield |
|---|---|
| With potassium fluoride In diethyl ether at 0℃; | 90% |
D(+)-2-Octanol Chemical Properties
The MF of (S)-(+)-2-Octanol(6169-06-8): C8H18O
The MW of (S)-(+)-2-Octanol(6169-06-8): 130.23
EINECS: 228-213-6
bp: 175 °C(lit.)
alpha: 9.5 °(neat)
density: 0.822 g/mL at 25 °C(lit.)
refractive index: n20/D 1.426(lit.)
Fp: 160 °F
Water Solubility: 1 g/L (20 °C)
Vapour Pressure: 0.306 mmHg at 25°C
Appearance: Colorless to light yellow liquid
Stability: Stable. Combustible. Incompatible with strong oxidizing agents
Categories: Alcohols, Hydroxy Esters and Derivatives;Chiral Compounds;chiral;Building Blocks for Liquid Crystals;Chiral Building Blocks;Chiral Compounds (Building Blocks for Liquid Crystals);Functional Materials;Simple Alcohols (Chiral);Synthetic Organic Chemistry;Chiral Compound;AlcoholsDerivatization Reagents;Chiral GC Derivatization Reagents;Chiral Building Blocks;Chiral Derivatization;ChiralDerivatization Reagents;Derivatization Reagents HPLC;Organic Building Blocks
Synonyms: D(+)-2-OCTANOL;D-2-OCTANOL;(s)-2-octano;(S)-octan-2-ol;(S)-(+)-2-HYDROXYOCTANE;(S)-2-HYDROXYOCTANE;(S)-(+)-2-OCTANOL;(S)-2-OCTANOL
The Structure of (S)-(+)-2-Octanol(6169-06-8):
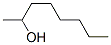
D(+)-2-Octanol Safety Profile
 Xi
Xi Risk Statements: 36/37/38
36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 26-36-37/39
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37/39: Wear suitable gloves and eye/face protection
WGK Germany: 3
HS Code: 29051620
D(+)-2-Octanol Specification
Ingestion:
If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid.
Inhalation:
Remove from exposure to fresh air immediately. Get medical aid if cough or other symptoms appear.
Skin:
Get medical aid if irritation develops or persists. Flush skin with plenty of soap and water.
Eyes:
Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
2. Handling and Storage
Storage:
Keep away from heat, sparks, and flame. Keep away from sources of ignition. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Handling:
Wash thoroughly after handling. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View