-
Name
Glutaric acid
- EINECS 203-817-2
- CAS No. 110-94-1
- Article Data476
- CAS DataBase
- Density 1.316 g/cm3
- Solubility 430 g/L (20 °C) in water
- Melting Point 95-98 °C(lit.)
- Formula C5H8O4
- Boiling Point 302.894 °C at 760 mmHg
- Molecular Weight 132.116
- Flash Point 151.2 °C
- Transport Information
- Appearance white or off-white crystals
- Safety 26-37/39-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Glutaricacid (8CI);1,3-Propanedicarboxylic acid;1,5-Pentanedioic acid;NSC 9238;
- PSA 74.60000
- LogP 0.32590
Synthetic route
-
-
13246-39-4
(+-)-1-(trimethylsilyl)ethanol

-
-
53715-97-2
5,5'-oxybis(5-oxopentanoic acid)

-
-
110-94-1
1,5-pentanedioic acid

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane | 100% |

| Conditions | Yield |
|---|---|
| With C24H33IrN4O3; water; sodium hydroxide for 18h; Catalytic behavior; Reagent/catalyst; Reflux; | 98% |
| In water for 48h; Ambient temperature; Gluconobacter roseus IAM 1841; | 97% |
| With sodium hydroxide In water at 20℃; Temperature; Concentration; Electrochemical reaction; | 91% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Na12[WZn3(H2O)2(ZnW9O34)2] at 75℃; for 7h; | 98% |

| Conditions | Yield |
|---|---|
| With ozone In tetrachloromethane; water at 20℃; for 1h; UV-irradiation; | 97% |
| With water; ozone at 25℃; under 760.051 Torr; for 1h; Irradiation; | 95% |
| With oxygen; sodium hydroxide In water at 90℃; under 3000.3 Torr; for 3h; pH=7; | 21% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium tungstate; phosphoric acid; dihydrogen peroxide at 90℃; for 5h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: Cyclopentanol With Oxone; ruthenium(III) chloride monohydrate In water at 70℃; for 8h; Stage #2: In ethanol Cooling; | 95% |
| With nitric acid at 70 - 100℃; | |
| With nitric acid; vanadia at 70 - 100℃; |

| Conditions | Yield |
|---|---|
| With oxygen; trifluoroacetic acid; sodium nitrite at 0 - 20℃; for 5.25h; Product distribution / selectivity; | 95% |
| Stage #1: cyclopentanone With Oxone; ruthenium(III) chloride monohydrate In water at 20℃; for 5h; Stage #2: In ethanol Cooling; | 90% |
| With oxygen; copper dichloride In acetic acid at 80℃; for 6h; Oxidation; | 84% |

| Conditions | Yield |
|---|---|
| With potassium metaperiodate; potassium aquapentachlororuthenate(III) In dichloromethane; water; acetonitrile at 20℃; for 2h; Catalytic behavior; Sonication; | 94% |
| With potassium permanganate; H-montmorillonite In water; benzene at 25 - 30℃; for 1h; | 90% |
| With periodic acid; cis-[RuCl2(bipy)2]*2H2O In tetrachloromethane; water; acetonitrile at 20℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| With triethylmethylammonium iodide; water; dihydrogen peroxide In acetonitrile at 55℃; for 23h; | 94% |
| With potassium carbonate In water at 25℃; Electrochemical reaction; | 81% |
| With iodine; oxygen In ethyl acetate for 10h; Mercury lamp irradiation; | 40% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium sulfate; OsO4 In ethyl acetate; N,N-dimethyl-formamide; tert-butyl alcohol | 92% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In water at 70℃; for 3.5h; Green chemistry; | 91% |
| With tert.-butylhydroperoxide; copper(l) chloride In acetonitrile at 20℃; for 3.5h; | 78% |
| With bis(acetylacetonate)oxovanadium; dihydrogen peroxide In acetonitrile at 60℃; for 8h; Reagent/catalyst; Time; | 42% |
-
-
56703-79-8
Glutaraminsaeureethylester

-
-
110-94-1
1,5-pentanedioic acid

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In 1,4-dioxane; water for 20h; Heating; | 90% |
-
-
1119-40-0
Dimethyl glutarate

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
1501-27-5
Pentanedioic acid, monomethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Dimethyl glutarate With potassium hydroxide In tetrahydrofuran; water at 0℃; for 0.166667h; Stage #2: With hydrogenchloride In tetrahydrofuran; water at 0℃; | A 13.2% B 86.8% |
| Stage #1: Dimethyl glutarate With lithium hydroxide; water In tetrahydrofuran at 0℃; for 0.333333 - 0.583333h; Stage #2: With water Product distribution / selectivity; Acidic conditions; | |
| Stage #1: Dimethyl glutarate With potassium hydroxide; water In tetrahydrofuran at 0℃; for 0.166667 - 0.416667h; Stage #2: With water Product distribution / selectivity; Acidic conditions; | |
| Stage #1: Dimethyl glutarate With cesium hydroxide; water In tetrahydrofuran at 0℃; for 0.166667 - 0.416667h; Stage #2: With water Product distribution / selectivity; Acidic conditions; | |
| Stage #1: Dimethyl glutarate With sodium hydroxide; water In tetrahydrofuran at 0℃; for 0.333333 - 0.583333h; Stage #2: With water Product distribution / selectivity; Acidic conditions; |
-
-
98-52-2
4-(1,1-dimethylethyl)-cyclohexanol

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
124-04-9
Adipic acid

-
C
-
110-15-6
succinic acid

-
D
-
10347-88-3
2-tert-butyl-1,4-butanedicarboxylic acid

| Conditions | Yield |
|---|---|
| With ammonium vanadate; nitric acid at 95 - 100℃; for 1h; | A n/a B n/a C n/a D 85% |


| Conditions | Yield |
|---|---|
| With oxygen; trifluoroacetic acid; sodium nitrite at 0 - 20℃; for 12.25h; Product distribution / selectivity; | A 85% B 10% |
-
-
17642-87-4
N,N,N',N'-tetraethyl glutaramide

-
-
110-94-1
1,5-pentanedioic acid

| Conditions | Yield |
|---|---|
| Stage #1: N,N,N',N'-tetraethyl glutaramide With sodium hydroxide In water at 110℃; for 12h; Stage #2: With hydrogenchloride; magnesium oxide In water; ethyl acetate pH=Ca. 4; Stage #3: With sulfuric acid In water; ethyl acetate pH=Ca. 4; | 85% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; hypophosphorous acid; sodium nitrite In water at -5 - 0℃; for 10 - 12h; Reagent/catalyst; | 80% |
-
-
110-83-8
cyclohexene

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
124-04-9
Adipic acid

-
C
-
1072-21-5
hexanedial

-
D
-
110-15-6
succinic acid

-
E
-
6140-65-4
1-cyclopentene-1-carboxaldehyde

-
F
-
931-17-9
1,2-Cyclohexanediol

-
G
-
533-60-8
cyclohexanone-2-ol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; ortho-tungstic acid In water at 140℃; for 0.333333h; Mechanism; Flow reactor; Microwave irradiation; Sealed tube; Green chemistry; chemoselective reaction; | A n/a B 74% C n/a D n/a E n/a F n/a G n/a |


| Conditions | Yield |
|---|---|
| With oxygen; sodium hydroxide In water at 90℃; for 3h; pH=> 13; | A 7% B 73% |
| With oxygen; H5PV2Mo10O40(1,11) In acetic acid at 60℃; under 750.06 Torr; for 10h; Yield given. Yields of byproduct given; | |
| With oxygen; H7*10H2O In acetic acid at 60℃; under 750.06 Torr; for 10h; Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With HeMaRaphos; water; toluene-4-sulfonic acid; palladium dichloride In tetrahydrofuran at 125℃; under 30003 Torr; for 24h; Autoclave; Green chemistry; regioselective reaction; | 73% |
-
-
19980-43-9
1-(trimethylsilyloxy)cyclopentene

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
99440-98-9
2-hydroxycyclopentanone

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; titanium silicate for 24h; Heating; | A 72% B 6% |

| Conditions | Yield |
|---|---|
| With water; nitrile hydratase SP361 at 30℃; for 40h; in potassium phosphate buffer (pH = 7); | A 9% B 70% |
| With water Product distribution; bacterial hydrolysis of aliphatic dinitriles with cells of Rhodococcus rhodochrous NCIB 11,216; 0.25 M phosphate buffer, pH 7; |
-
-
142-68-7
TETRAHYDROPYRANE

-
A
-
542-28-9
3,4,5,6-tetrahydro-2H-pyran-2-one

-
B
-
110-94-1
1,5-pentanedioic acid

| Conditions | Yield |
|---|---|
| With sodium bromate; potassium hydrogensulfate In water at 25 - 30℃; for 20h; Oxidation; | A 69% B 16% |
| With 2,6-dichloropyridine N-oxide; dichloro(5,10,15,20-tetrakis(pentafluorophenyl)porphyrinato)ruthenium(IV) In 1,2-dichloro-ethane at 40℃; for 20h; Reagent/catalyst; Inert atmosphere; | A 56% B 16% |

| Conditions | Yield |
|---|---|
| With ruthenium tetroxide In tetrachloromethane; water for 95h; | 68% |
| With lithium nitrate In water; acetonitrile at 25℃; anodic oxidation; | 66% |
| With hydrogenchloride | |
| With nitric acid at 25 - 35℃; |
-
-
1670-46-8
2-acetylcyclopentanaone

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
626-70-0
2-methylhexane-1,6-dioic acid

-
C
-
1262892-77-2
2-acetyl-2-hydroxycyclopentanone

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In acetic acid at 20℃; for 240h; | A 67% B 30% C 0.5% |


| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; nitric acid; trifluoroacetic acid at 20℃; under 760.051 Torr; for 18h; Catalytic behavior; Reagent/catalyst; | A 5 %Spectr. B 66% |
| With N-hydroxyphthalimide; bis(acetylacetonato)manganese(II); oxygen; acetic acid; cobalt acetylacetonate at 80℃; under 760.051 Torr; for 24h; | |
| Multi-step reaction with 2 steps 1: N-hydroxyphthalimide; oxygen; acetic acid; bis(acetylacetonato)manganese(II) / 6 h / 100 °C / 760.05 Torr 2: oxygen; acetic acid; bis(acetylacetonato)manganese(II) / 6 h / 100 °C / 760.05 Torr View Scheme |
-
-
108-93-0
cyclohexanol

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
124-04-9
Adipic acid

-
C
-
54812-72-5
monocyclohexyl adipate

-
D
-
108-94-1
cyclohexanone

| Conditions | Yield |
|---|---|
| With oxygen at 120℃; for 4.33333h; Further byproducts given; | A 3.1% B 12.4% C 8.9% D 65% |
| With oxygen at 120℃; for 4.33333h; Further byproducts given; | A 3.1% B 12.4% C 8.9% D 65% |

-
-
110-82-7
cyclohexane

-
A
-
110-94-1
1,5-pentanedioic acid

-
B
-
124-04-9
Adipic acid

-
C
-
110-15-6
succinic acid

-
D
-
108-94-1
cyclohexanone

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; bis(acetylacetonato)manganese(II); oxygen; acetic acid at 80℃; under 760.051 Torr; for 14h; Time; | A 9% B 65% C 6% D 19% |


| Conditions | Yield |
|---|---|
| With potassium superoxide; tetraethylammonium bromide In N,N-dimethyl-formamide at 25℃; for 12h; Ring cleavage; | 64% |
| Multi-step reaction with 2 steps 1: sodium; alcohol 2: chromic acid View Scheme |

| Conditions | Yield |
|---|---|
| With boron trifluoride at 65℃; for 0.333333h; | 100% |
| With phosphorus trichloride Cooling; | 92% |
| With hydrogenchloride |
-
-
110-94-1
1,5-pentanedioic acid

-
-
59565-09-2
2-(vinyloxy)ethyl isothiocyanate

| Conditions | Yield |
|---|---|
| trifluoroacetic acid at 55 - 60℃; | 100% |

| Conditions | Yield |
|---|---|
| In toluene (Ar) suspn. ZnO and glutaric acid in toiluene was heated at 80°C for 1 day; react. mixt. was cooled, ppt. was filtered, washed with acetone and dried in vacuo at 130°C; powder X-ray diffraction; | 100% |
| In ethanol; toluene at 60℃; for 10h; Solvent; | 97% |
| In toluene at 60℃; | 85% |
| In toluene byproducts: H2O; powdered ZnO added to soln. of org. compd., slurry stirred vigorously at55°C for 2 h then refluxed until no more H2O in trap; cooled to room temp., filtered, washed with acetone, powdered product dried in vac. at 100°C for 5 d; detn. FTIR, XANES EXAFS, (13)C NMR; | |
| In toluene heating (3 h, 45°C); filtration, drying (overnight, vac., 90°C); |
-
-
110-94-1
1,5-pentanedioic acid

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

-
-
151433-25-9
(R,R)-N,N'-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine

| Conditions | Yield |
|---|---|
| Stage #1: cobalt(II) acetate; (R,R)-N,N'-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine In ethanol for 5h; Heating / reflux; Stage #2: 1,5-pentanedioic acid With oxygen In dichloromethane; acetone at 20℃; for 3h; | 100% |


| Conditions | Yield |
|---|---|
| In chloroform for 0.5h; | 100% |
| In tetrahydrofuran Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With triethylamine at 160℃; for 5h; Autoclave; Green chemistry; | 99% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

-
-
7268-93-1
[Cu(pentanedioate)]n

| Conditions | Yield |
|---|---|
| With ozone In water for 6h; Time; | 98.97% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
95-54-5
1,2-diamino-benzene

-
-
7147-66-2
2,2'-(1,3-propanediyl)bis(1H-benzimidazole)

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid In water at 150℃; for 2h; | 98% |
| With polyphosphoric acid at 120℃; | 79% |
| With polyphosphoric acid at 180℃; | 65% |

| Conditions | Yield |
|---|---|
| With PPA at 80℃; for 12h; | 98% |

| Conditions | Yield |
|---|---|
| With PPA at 80℃; for 12h; | 98% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
99135-90-7
(3R,4R)-3,4-bis(diphenylphosphanyl)pyrrolidine

-
-
104351-44-2
δ-<(3R,4R)-3,4-Bis(diphenylphophino)pyrrolidino>-δ-oxopentansaeure

| Conditions | Yield |
|---|---|
| In diethyl ether; dichloromethane | 98% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
59721-28-7
camostat

| Conditions | Yield |
|---|---|
| In ethanol at 20 - 65℃; | 98% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
3171-45-7
4,5-dimethyl-1,2-phenylenediamine

-
-
1171054-64-0
5,6,5',6'-tetramethyl-2,2'-(1,3-propanediyl) bis-(1H-benzimidazole)

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid In water at 150℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 70℃; for 1h; | 98% |

-
-
110-94-1
1,5-pentanedioic acid

-
-
100-01-6
4-nitro-aniline

-
-
139776-02-6
1-(4-nitro-phenyl)-piperidine-2,6-dione

| Conditions | Yield |
|---|---|
| With PPA at 80℃; for 12h; | 97% |

| Conditions | Yield |
|---|---|
| With PPA at 80℃; for 12h; | 97% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
1002101-64-5
(C20H11CHO(OH)OCO)2C3H6

| Conditions | Yield |
|---|---|
| Stage #1: 1,5-pentanedioic acid; (S)-2,2'-dihydroxy-1,1'-binaphthyl-3-carbaldehyde With dmap In dichloromethane at 20℃; for 0.25h; Stage #2: With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0 - 20℃; Inert atmosphere; | 97% |
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane for 3h; | 96% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
1117-97-1
N,0-dimethylhydroxylamine

-
-
259236-21-0
N1,N5-dimethoxy-N1,N5-dimethylglutaramide

| Conditions | Yield |
|---|---|
| Stage #1: 1,5-pentanedioic acid; N,0-dimethylhydroxylamine In toluene at 0℃; for 0.166667h; Stage #2: With phosphorus trichloride In toluene at 20 - 60℃; for 0.5h; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: metformin hydrochloride With sodium hydroxide In methanol; chloroform; water at 20℃; Stage #2: 1,5-pentanedioic acid In methanol; chloroform at 20℃; Product distribution / selectivity; | 96.9% |
| Stage #1: metformin hydrochloride With sodium hydroxide In tetrahydrofuran; water at 20℃; Stage #2: 1,5-pentanedioic acid In tetrahydrofuran; water at 10℃; Product distribution / selectivity; | 96.7% |
| Stage #1: metformin hydrochloride With sodium hydroxide In methanol; chloroform; water at 20℃; Stage #2: 1,5-pentanedioic acid In methanol; chloroform; water at 20℃; Product distribution / selectivity; | 96.9% |

| Conditions | Yield |
|---|---|
| Stage #1: dimethylbiguanide In water; acetone at 40℃; Stage #2: 1,5-pentanedioic acid In water; acetone at 10℃; Product distribution / selectivity; | 95.3% |
| In water; acetone at 10 - 40℃; Product distribution / selectivity; | 95.3% |
| In ethanol at 10 - 20℃; Product distribution / selectivity; | 62.8% |
| With sodium hydroxide In ethanol at 70℃; Product distribution / selectivity; | 57.7% |
| In methanol at 40℃; Product distribution / selectivity; | 43.8% |
-
-
110-94-1
1,5-pentanedioic acid

-
-
104-76-7
2-Ethylhexyl alcohol

-
-
21302-20-5
glutaric acid bis-(2-ethyl-hexyl ester)

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene at 160℃; for 2h; | 95% |
| With Candida antarctica lipase B In cyclohexane at 45℃; for 24h; | 68% |
| With sulfuric acid; toluene |

| Conditions | Yield |
|---|---|
| With Isopropenyl acetate; Montmorillonite KSF for 0.0666667h; Irradiation; | 95% |
| With PEG-1000; sulfated zirconia at 40℃; for 1.5h; neat (no solvent); | 95% |
| With niobium(V) oxide hydrate In 1,3,5-trimethyl-benzene at 200℃; for 60h; Inert atmosphere; Molecular sieve; | 92% |
Glutaric acid Chemical Properties
The Molecular Structure of Glutaric acid (CAS NO.110-94-1):
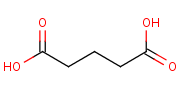
Empirical Formula: C5H8O4
Molecular Weight: 132.1146
IUPAC Name: pentanedioic acid
Appearance: white or off-white crystals
Nominal Mass: 132 Da
Average Mass: 132.1146 Da
Monoisotopic Mass: 132.042259 Da
Index of Refraction: 1.476
Molar Refractivity: 28.34 cm3
Molar Volume: 100.3 cm3
Surface Tension: 56.1 dyne/cm
Density: 1.316 g/cm3
Flash Point: 151.2 °C
Enthalpy of Vaporization: 59.74 kJ/mol
Boiling Point: 302.9 °C at 760 mmHg
Vapour Pressure: 0.000223 mmHg at 25°C
Product Categories: Pharmaceutical Intermediates;alpha,omega-Alkanedicarboxylic Acids;alpha,omega-Bifunctional Alkanes;Monofunctional & alpha,omega-Bifunctional Alkanes;C1 to C5;Carbonyl Compounds;Carboxylic Acids
Water Solubility: 430 g/L (20 ºC)
Stability: Stable. Incompatible with bases, oxidizing agents, reducing agents
Synonyms: 1,3-Propanedicarboxylic acid ; 1,5-Pentanedioic acid ; 4-02-00-01934 (Beilstein Handbook Reference) ; AI3-24247 ; BRN 1209725 ; EINECS 203-817-2 ; GLUTARIC ACID ; Pentandioic acid ; Pentanedioic acid ; n-Pyrotartaric acid ; Pentanedioic acid
Glutaric acid Uses
Glutaric acid Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 6gm/kg (6000mg/kg) | Biochemical Journal. Vol. 34, Pg. 1196, 1940. |
LD50/LC50: RTECS:
CAS# 110-94-1: Oral, mouse: LD50 = 6 gm/kg;
Carcinogenicity: Glutaric acid - Not listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65.
Other: See actual entry in RTECS for complete information. The toxicological properties have not been fully investigated.
Glutaric acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36-36/37/38
R36: Irritating to eyes
R36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 26-37/39-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S37/39: Wear suitable gloves and eye/face protection
S36: Wear suitable protective clothing
WGK Germany: 1
RTECS: MA3740000
HS Code: 29171990
Glutaric acid Specification
Personal Protection:
Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure.
Fire Fighting:Wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. To extinguish fire use water spray, dry chemical, carbon dioxide, or chemical foam.
Product Categories: Pharmaceutical Intermediates;alpha,omega-Alkanedicarboxylic Acids;alpha,omega-Bifunctional Alkanes;Monofunctional & alpha,omega-Bifunctional Alkanes;C1 to C5;Carbonyl Compounds;Carboxylic Acids
Related Products
- Glutaric acid
- Glutaric acid monomethyl ester
- Glutaric anhydride
- 110942-02-4
- 110945-00-1
- 11094-60-3
- 110-95-2
- 110960-73-1
- 11096-26-7
- 110-96-3
- 110963-29-6
- 110963-63-8
- 11096-37-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View