-
Name
Methylboronic acid
- EINECS 629-203-8
- CAS No. 13061-96-6
- Article Data41
- CAS DataBase
- Density 0.965 g/cm3
- Solubility Soluble in water.
- Melting Point 91-94 °C
- Formula CH5BO2
- Boiling Point 141.745 °C at 760 mmHg
- Molecular Weight 59.8605
- Flash Point 39.526 °C
- Transport Information
- Appearance White to light yellow crystal powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Methaneboronicacid (6CI,7CI,8CI);Boronicacid, methyl- (9CI);Methaneboronic acid;
- PSA 40.46000
- LogP -0.91100
Synthetic route
-
-
13292-87-0
dimethyl sulfide borane

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With carbon monoxide; water In tetrahydrofuran byproducts: S(CH3)2; a soln. of BH3*S(CH3)2 flushed with CO; a soln. of catalyst added, stirred rapidly and maintained at 25°C for 12-16 h; water added; evapd.; | 94% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether byproducts: LiCl, i-propanol; (N2); a soln. of B(O-i-Pr)3 cooled with a dry ice-acetone bath; CH3Li added slowly and stirred for 1 h; allowed to warm to room temp. with stirring for 3 h; cooled to 0°C; a soln. of HCl in ether added and stirred for 0.5 h; decanted; hydrolyzed with water; distd.; dried; | 79% |

| Conditions | Yield |
|---|---|
| With diethyl ether Zers. des Reaktionsproduktes durch angesaeuertes Wasser, wobei Verseifung des intermediaer gebildeten Methylborsaeureesters stattfindet; |

| Conditions | Yield |
|---|---|
| With diethyl ether und anschliessend mit wss.H2SO4; |

| Conditions | Yield |
|---|---|
| Zers. des entstandenen 'Methylborsaeuredimethylesters' CH3B(OCH3)2 durch angesaeuertes Wasser; |

-
-
17933-16-3
dibromomethylborane

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With water | |
| With water |
-
-
5314-85-2
2,4,6-Trimethyl-[1,3,5,2,4,6]triazatriborinane

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With water at 150℃; |
-
-
16321-94-1
1,2-Dimethylboran

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With water at 150℃; |
-
-
7647-01-0
hydrogenchloride

-
-
877-07-6
hexamethylborazine

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
593-51-1
methylamine hydrochloride

| Conditions | Yield |
|---|---|
| at 100℃; Hydrolysis; |


-
-
593-90-8
trimethylborane

-
-
7732-18-5
water

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
823-96-1
Trimethylboroxine

-
C
-
19163-04-3
Dimethylborinsaeure-anhydrid

| Conditions | Yield |
|---|---|
| at 320℃; |

-
-
5314-85-2
2,4,6-Trimethyl-[1,3,5,2,4,6]triazatriborinane

-
-
7732-18-5
water

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
7664-41-7
ammonia

| Conditions | Yield |
|---|---|
| at 150℃; |


| Conditions | Yield |
|---|---|
| at 200 - 250℃; |


| Conditions | Yield |
|---|---|
| at 200 - 250℃; |



-
A
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| In neat (no solvent) holding at room temp. in screw-cap vials, crystn. near the cap; identification by single-crystal X-ray analysis; |
-
-
917-64-6
methyl magnesium iodide

-
-
121-43-7
Trimethyl borate

-
A
-
593-90-8
trimethylborane

-
B
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| In diethyl ether | |
| In diethyl ether |

-
-
23777-55-1
methyl diborane

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With water byproducts: H2; at room temp.; | |
| With H2O byproducts: H2; at room temp.; |


| Conditions | Yield |
|---|---|
| With water hydrolysis;; | |
| With H2O hydrolysis;; |
-
-
16924-32-6
1.1-dimethyl borane(6)

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With water byproducts: H2; at room temp.; | |
| With H2O byproducts: H2; at room temp.; |

-
-
20436-23-1, 17156-88-6, 6808-37-3
dimethyldiborane

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With water byproducts: H2; at room temp.; | |
| With H2O byproducts: H2; at room temp.; |

-
-
21127-95-7
2-methyl-borazine

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
7664-41-7
ammonia

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; | |
| With H2O; HCl byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; |

-
-
21482-59-7
tetramethyl diborane

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With water byproducts: H2; at room temp.; | |
| With H2O byproducts: H2; at room temp.; |

-
-
21107-27-7
trimethyldiborane(6)

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With water byproducts: H2; | |
| With H2O byproducts: H2; |
-
-
21107-27-7
trimethyldiborane(6)

-
A
-
13061-97-7
hydroxy-dimethyl-borane

-
B
-
13061-96-6
dihydroxy-methyl-borane

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With water byproducts: H2; at room temp.; | |
| With H2O byproducts: H2; at room temp.; |

-
-
23208-27-7
B-dimethylborazole

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
7664-41-7
ammonia

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; | |
| With H2O; HCl byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; |

-
-
5314-85-2
2,4,6-Trimethyl-[1,3,5,2,4,6]triazatriborinane

-
A
-
13061-96-6
dihydroxy-methyl-borane

-
B
-
7664-41-7
ammonia

-
C
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; | |
| With H2O; HCl byproducts: H2; at 200-250°C for 6 h with 20% aq. HCl; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; water byproducts: NH3, CH3NH2, H2; by heating; | |
| With HCl; H2O byproducts: NH3, CH3NH2, H2; by heating; |
-
-
691857-51-9
ethyl 2-(4-fluorophenyl)-6-nitro-5-(trifluoromethylsulfonyloxy)benzofuran-3-carboxylate

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1378428-85-3
ethyl 2-(4-fluorophenyl)-5-methyl-6-nitro-1-benzofuran-3-carboxylate

| Conditions | Yield |
|---|---|
| With potassium fluoride; sodium bromide; tetrakis(triphenylphosphine) palladium(0) In water; toluene for 25h; Inert atmosphere; Reflux; | 100% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane for 12h; Inert atmosphere; Reflux; | 100% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In water; toluene at 85℃; for 6h; | 100% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| Stage #1: dihydroxy-methyl-borane; tert-butyl ((5-chloro-2-fluoro-4-((6-fluoro-2-((isopropyl(methyl)amino)methyl)-3-methoxybenzyl)amino)phenyl)sulfonyl)(thiazol-4-yl)carbamate With potassium phosphate In 1,4-dioxane for 0.333333h; Suzuki-Miyaura Coupling; Inert atmosphere; Stage #2: With palladium diacetate; tricyclohexylphosphine tetrafluoroborate In 1,4-dioxane at 105℃; | 100% |
-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; caesium carbonate In 2-methyltetrahydrofuran at 80℃; for 1h; | 100% |
| Stage #1: dihydroxy-methyl-borane; tert-butyl 2-bromo-6-[3-fluoro-2-methoxy-4-(trifluoromethoxy)phenoxy]-3-(trifluoromethyl)benzoate With caesium carbonate In 2-methyltetrahydrofuran for 0.5h; Inert atmosphere; Stage #2: With (bis-(2-methoxyethyl)amino)sulfur trufluoride In 2-methyltetrahydrofuran at 80℃; for 1h; | 100% |
-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With potassium dihydrogenphosphate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water at 100℃; for 16h; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; potassium phosphate; water In 2-methoxy-ethanol at 110℃; for 4h; | 91% |
-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); (R)-N-((S)-(3,5-di-tert-butyl-4-methoxyphenyl)(2-(dicyclohexylphosphanyl)phenyl)methyl)-N,2-dimethylpropane-2-sulfinamide; potassium hydroxide In toluene at 20 - 85℃; Inert atmosphere; Sealed tube; enantioselective reaction; | 99.2% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In tetrahydrofuran; toluene at 100℃; for 24h; Suzuki-Miyaura reaction; | 99% |
-
-
867380-21-0
2-(3-chloro-8-methyl-azulen-5-yl)-propionic acid methyl ester

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
111415-94-2
2-(3,8-Dimethyl-azulen-5-yl)-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium phosphate In toluene at 100℃; for 24h; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
111562-32-4
5-(2-aminophenyl)-1H-pyrazole

-
-
1350934-17-6
5-methyl-5,6-dihydrobenzo[e]pyrazolo[1,5-c][1,3,2]diazaborinine

| Conditions | Yield |
|---|---|
| In toluene for 1h; Inert atmosphere; Dean-Stark; Reflux; | 99% |
| In toluene for 1h; Inert atmosphere; Reflux; | 89% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1628799-95-0
2-formyl-4,6-dimethylphenyl trifluoromethanesulfonate

-
-
5779-71-5
2,3,5-trimethylbenzaldehyde

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium carbonate In tetrahydrofuran; water Suzuki Coupling; Reflux; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1403381-95-2
2-formyl-3,6-dimethylphenyl trifluoromethanesulfonate

-
-
34341-29-2
2,3,6-trimethylbenzaldehyde

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium carbonate In tetrahydrofuran; water Suzuki Coupling; Reflux; | 99% |
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium carbonate In tetrahydrofuran; water Suzuki Coupling; Enzymatic reaction; | 93% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
4408-64-4
N-methyliminodiacetic acid

| Conditions | Yield |
|---|---|
| In acetonitrile at 130℃; under 12929 Torr; for 0.0833333h; Solvent; Microwave irradiation; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1310708-89-4
3-methyl-1-(pyrimidin-2-yl)-1H-indole

| Conditions | Yield |
|---|---|
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; silver carbonate In neat (no solvent) at 46℃; for 1h; Milling; regioselective reaction; | 99% |
| With potassium fluoride; palladium diacetate; silver carbonate; p-benzoquinone In tert-Amyl alcohol at 110℃; for 6h; Sealed tube; regioselective reaction; | 95% |
-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In toluene at 100℃; for 3h; Inert atmosphere; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| Stage #1: (4S,5S)-2-{(1S,3S,4S,5R,6S)-1-chlor-5-(benzyloxy)-3-[(4-methoxybenzyl)oxy]-4,6-dimethyloctyl}-4,5-dicyclohexyl-1,3,2-dioxaborolane With dihydrogen peroxide; sodium carbonate In tetrahydrofuran; water at 20℃; for 24h; Stage #2: With Sodium thiosulfate pentahydrate; sodium iodide In tetrahydrofuran; water for 0.5h; Stage #3: dihydroxy-methyl-borane With magnesium sulfate at 20℃; for 19h; | A 64% B 99% |

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 1h; Molecular sieve; Inert atmosphere; Sealed tube; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
672-92-4
1,1',1''‐nitrilotris(propan‐2‐one)trioxime

-
-
100-39-0
benzyl bromide

| Conditions | Yield |
|---|---|
| Stage #1: dihydroxy-methyl-borane; 1,1',1''‐nitrilotris(propan‐2‐one)trioxime; benzyl bromide In methanol at 20℃; for 20h; Stage #2: With lithium carbonate In methanol at 20℃; for 4h; | 99% |

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1158974-93-6
(R)-(1-amino-3-methylbutyl)boronic acid hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; acetone at 20℃; | 99% |
-
-
269409-73-6
3-carboxyphenylboronic acid pinacol ester

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1218790-99-8
4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)piperidine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: dihydroxy-methyl-borane; 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)piperidine hydrochloride With trifluoroacetic acid In dichloromethane at 20℃; Stage #2: With hydrogenchloride In water at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); 3-(dicyclohexylphosphino)-2-(2,6-dimethoxyphenyl)-1-methyl-1H-indole In 1,4-dioxane at 100℃; for 6h; Suzuki-Miyaura Coupling; Schlenk technique; Sealed tube; Inert atmosphere; | 99% |
-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In toluene at 90℃; for 18h; Suzuki Coupling; Inert atmosphere; | 99% |
-
-
867380-15-2
1-chloro-4-methyl-azulene

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1127-69-1
1,4-dimethylazulene

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium phosphate In toluene at 100℃; for 24h; | 98% |

-
-
13061-96-6
dihydroxy-methyl-borane

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium diacetate; phosphine ligand In tetrahydrofuran; water; toluene at 100℃; for 0.166667h; microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; tri-tert-butyl phosphine; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 20℃; for 11h; Reflux; Inert atmosphere; | 98% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium diacetate; tricyclohexylphosphine tetrafluoroborate In 1,4-dioxane for 4h; Inert atmosphere; Reflux; | 98% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
210907-84-9
3-aminophenylboronic acid pinacolate

| Conditions | Yield |
|---|---|
| Stage #1: dihydroxy-methyl-borane; 3-aminophenylboronic acid pinacolate With trifluoroacetic acid In dichloromethane at 20℃; Stage #2: With hydrogenchloride In water at 20℃; Reagent/catalyst; Temperature; Solvent; | 98% |
-
-
13061-96-6
dihydroxy-methyl-borane


| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In 1,4-dioxane; water at 120℃; for 0.5h; Inert atmosphere; Sealed tube; Microwave irradiation; | 98% |
-
-
13061-96-6
dihydroxy-methyl-borane

-
-
1210419-44-5
methyl 6-(2,6-difluoro-3-(trifluoromethyl-sulfonyloxy)phenyl)-5-fluoropicolinate

-
-
1210419-49-0
6-(2,6-difluoro-3-methylphenyl)-5-fluoropicolinic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water at 100℃; for 3h; Inert atmosphere; | 97% |
Methylboronic acid Specification
Methylboronic acid is an organic compound with the formula CH5BO2, and its systematic name is the same with the product name. With the CAS registry number 13061-96-6, it is also named as Methaneboronic acid. It belongs to the product categories of Substituted Boronic Acids; API intermediates; Boronic acid; Organoborons; Chiral Chemicals; Alkyl. In addition, the molecular weight is 59.86. It is used as boron compounds intermediates in organic synthesis. This chemical is stable at common pressure and temperature, and it should be sealed and stored in a cool, ventilated and dry place. Moreover, it should be protected from oxides, bases heat and air.
Physical properties of Methylboronic acid are: (1)ACD/LogP: -0.212; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.21; (4)ACD/LogD (pH 7.4): -0.21; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 18.27; (8)ACD/KOC (pH 7.4): 18.22; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 40.46 Å2; (13)Index of Refraction: 1.345; (14)Molar Refractivity: 13.181 cm3; (15)Molar Volume: 62.004 cm3; (16)Polarizability: 5.225×10-24cm3; (17)Surface Tension: 27.77 dyne/cm; (18)Density: 0.965 g/cm3; (19)Flash Point: 39.526 °C; (20)Enthalpy of Vaporization: 44.145 kJ/mol; (21)Boiling Point: 141.745 °C at 760 mmHg; (22)Vapour Pressure: 2.35 mmHg at 25°C.
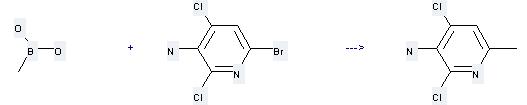
Uses of Methylboronic acid: it can be used to produce 2,4-dichloro-6-methyl-pyridin-3-ylamine at the temperature of 100 °C. It will need reagents K2CO3, Pd(PPh3)2Cl2 and solvent dimethylformamide with the reaction time of 24 hours. The yield is about 85%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need to wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: OB(O)C
(2)Std. InChI: InChI=1S/CH5BO2/c1-2(3)4/h3-4H,1H3
(3)Std. InChIKey: KTMKRRPZPWUYKK-UHFFFAOYSA-N
Related Products
- Methylboronic acid
- 1306-23-6
- 130623-81-3
- 1306-24-7
- 130624-89-4
- 1306-25-8
- 13062-76-5
- 13062-78-7
- 130628-75-0
- 13063-04-2
- 130631-59-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View