-
Name
4,4'-AZODIANILINE
- EINECS
- CAS No. 538-41-0
- Article Data50
- CAS DataBase
- Density 1.2046 (rough estimate)
- Solubility Very soluble in ethanol. Insoluble in water.
- Melting Point 245 °C
- Formula C12H12 N4
- Boiling Point 342.09°C (rough estimate)
- Molecular Weight 212.254
- Flash Point 213oC
- Transport Information
- Appearance
- Safety Poison by intravenous route. Mutation data reported. When heated to decomposition it emits toxic vapors of NOx.
- Risk Codes R40; R25
-
Molecular Structure
- Hazard Symbols 745553
- Synonyms Aniline,4,4'-azodi- (6CI,7CI,8CI); Benzenamine, 4,4'-azobis- (9CI); 4,4'-Azodianiline;4,4'-Diaminoazobenzene; NSC 17103; p,p'-Diaminoazobenzene; p-Azoaniline;p-Diaminoazobenzene; p'-Amino-p-aminoazobenzene
- PSA 76.76000
- LogP 4.42880
Synthetic route

| Conditions | Yield |
|---|---|
| With sodiumsulfide nonahydrate In ethanol at 80℃; for 20h; | 99% |
| With sodiumsulfide nonahydrate In methanol; water at 85℃; for 3h; | 90% |
| With sodiumsulfide nonahydrate In methanol; water at 85℃; for 4h; | 87% |
| With ammonium sulfide; ethanol | |
| With methanol; sodiumsulfide nonahydrate |

| Conditions | Yield |
|---|---|
| With sodium perborate; Dihydrate sodium molybdate In acetic acid at 20℃; for 1h; Kinetics; | 92% |
| With Dess-Martin periodane In dichloromethane at 20℃; for 0.666667h; | 84% |
| With [bis(acetoxy)iodo]benzene In dichloromethane at 20 - 25℃; for 1h; | 74% |

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butylbiphenyl; lithium; iron(II) chloride In tetrahydrofuran for 3h; Heating; | 83% |
| With ammonium persulfate; sulfuric acid ueber mehrere Stufen; | |
| Multi-step reaction with 2 steps 1: Oxone; sulfuric acid / water / 2 h / 60 - 65 °C 2: sodium sulfide / ethanol; water / 0.5 h / Reflux View Scheme |
-
-
100-01-6
4-nitro-aniline

-
A
-
61594-51-2
4,4'-diaminoazoxybenzene

-
B
-
106-50-3
1,4-phenylenediamine

-
C
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; nickel(II) nitrate; zinc In tert-butyl alcohol for 0.833333h; Heating; | A 54% B 40% C 2 % Chromat. |
| With hydrazine hydrate; nickel(II) nitrate; zinc In ethanol for 5h; Heating; | A 23% B 51% C 21% |


| Conditions | Yield |
|---|---|
| With titanium (0) reagent In tetrahydrofuran for 0.25h; Ambient temperature; | A 45% B 48% |

| Conditions | Yield |
|---|---|
| In toluene for 8h; Heating; | A 38% B 13% |
-
-
861596-22-7
N,N'-bis-[4-(4-amino-phenylazo)-phenyl]-hydrazine

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With ammonium hydrosulphide |
-
-
61893-92-3
acetic acid-[4-(4-amino-phenylazo)-anilide]

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With sulfuric acid |
-
-
906715-99-9
acetic acid-[4-(3-phenyl-triazenyl)-anilide]

-
-
142-04-1
aniline hydrochloride

-
A
-
60-09-3
aniline yellow

-
B
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With aniline anschliessend Kochen des Reaktionsproduktes mit verd. Schwefelsaeure; |


-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With ammonium hydrosulphide |
-
-
98-95-3
nitrobenzene

-
-
106-50-3
1,4-phenylenediamine

-
A
-
60-09-3
aniline yellow

-
B
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate |

-
-
906715-99-9
acetic acid-[4-(3-phenyl-triazenyl)-anilide]

-
-
142-04-1
aniline hydrochloride

-
-
62-53-3
aniline

-
A
-
60-09-3
aniline yellow

-
B
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| anschl. Kochen mit verd. H2SO4; | |
| Kochen des Reaktionsprodukts mit verd.Schwefelsaeure; |

-
-
3646-57-9, 89103-79-7
bis(4-nitrophenyl)diazene

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With sodium sulfide | |
| With tetrahydrofuran; lithium aluminium tetrahydride; diethyl ether | |
| With sodium sulfide In ethanol; water for 0.5h; Reflux; |
-
-
22719-28-4
4,4′-dinitrohydrazobenzene

-
A
-
21371-47-1
bis-[4-(4-amino-phenylazo)-phenyl]-diazene

-
B
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With sodium sulfide | |
| With sodium sulfide |

| Conditions | Yield |
|---|---|
| With sodium carbonate; nitrobenzene |
-
-
15446-39-6, 78752-51-9
N,N'-diacetyl-4,4'-diaminoazobenzene

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With hydrogenchloride | |
| With sodium hydroxide | |
| With hydrogenchloride; water In methanol for 1.5h; |

| Conditions | Yield |
|---|---|
| With methanol; iodine; magnesium for 1h; Ambient temperature; | A 49 % Chromat. B 50 % Chromat. |
-
-
14368-49-1
p-nitrobenzenediazonium

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| Reduktion der hierbei entstandenen Azoverbindung mit Glykose, Formaldehyd, Zink oder Zinn in alkal.Loesung; |
-
-
7647-01-0
hydrogenchloride

-
-
15446-39-6, 78752-51-9
N,N'-diacetyl-4,4'-diaminoazobenzene

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonia |

-
-
64-17-5
ethanol

-
-
22719-28-4
4,4′-dinitrohydrazobenzene

-
A
-
21371-47-1
bis-[4-(4-amino-phenylazo)-phenyl]-diazene

-
B
-
538-41-0
4,4'-Diaminoazobenzene

-
-
98-95-3
nitrobenzene

-
-
106-50-3
1,4-phenylenediamine

-
A
-
60-09-3
aniline yellow

-
B
-
538-41-0
4,4'-Diaminoazobenzene

-
-
98-95-3
nitrobenzene

-
-
106-50-3
1,4-phenylenediamine

-
A
-
60-09-3
aniline yellow

-
B
-
538-41-0
4,4'-Diaminoazobenzene

-
C
-
1161-45-1
1,4-bis-phenylazo-benzene

| Conditions | Yield |
|---|---|
| at 145℃; |

-
-
64-17-5
ethanol

-
-
614-25-5, 21650-71-5, 71297-95-5
4,4'-dinitroazoxybenzene

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| at 25℃; dann auf dem Wasserbad; |

-
-
100-25-4
para-dinitrobenzene

-
A
-
100-01-6
4-nitro-aniline

-
B
-
3646-57-9, 89103-79-7
bis(4-nitrophenyl)diazene

-
C
-
538-41-0
4,4'-Diaminoazobenzene

-
-
7664-93-9
sulfuric acid

-
-
61893-92-3
acetic acid-[4-(4-amino-phenylazo)-anilide]

-
-
538-41-0
4,4'-Diaminoazobenzene
-
-
64-17-5
ethanol

-
-
861596-22-7
N,N'-bis-[4-(4-amino-phenylazo)-phenyl]-hydrazine

-
-
538-41-0
4,4'-Diaminoazobenzene


| Conditions | Yield |
|---|---|
| Stage #1: bromocyane; 4,4'-Diaminoazobenzene With acetic acid In N,N-dimethyl-formamide at -15 - -10℃; for 0.5h; Stage #2: With sodium hydroxide In N,N-dimethyl-formamide at -15 - 20℃; for 25h; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 15 - 20℃; for 6h; | 93% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; Cooling with ice; | 91% |

| Conditions | Yield |
|---|---|
| With (2,2':6',2''-terpyridine)(pyridine-2-carboxylate)iron(II)chloride; sodium hydroxide In water; acetonitrile | 90% |
-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 4,4'-Diaminoazobenzene With hydrogenchloride; sodium nitrite In water Stage #2: With sodium azide In water | 90% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 12h; Reflux; Inert atmosphere; | 87% |
-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
494798-73-1
4-(4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-heptadecafluoroundecyloxy)-benzaldehyde

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl acetamide at 160℃; for 10h; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; ethyl acetate at 20℃; for 3h; Inert atmosphere; | 82% |
-
-
304865-77-8
N-(2,6-diisopropylphenyl)-acetimidoyl chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| In toluene for 4h; Reflux; | 81% |

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In N,N-dimethyl-formamide for 7h; Heating; | 80% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 50℃; for 12h; | 79% |

| Conditions | Yield |
|---|---|
| In ethanol | 75% |

| Conditions | Yield |
|---|---|
| In hexane at 50 - 60℃; for 3.16667h; Milling; | 75% |
-
-
64-18-6
formic acid

-
-
93614-64-3
2-triethylammonio acetic acid chloride chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-triethylammonio acetic acid chloride chloride; 4,4'-Diaminoazobenzene With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide; acetonitrile at 0 - 20℃; Inert atmosphere; Stage #2: formic acid With silica gel In water; acetonitrile | 73% |


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 73% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0℃; for 1h; Inert atmosphere; | 73% |
-
-
90-02-8
salicylaldehyde

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
16395-62-3
N,N'-bis(salicylidine)-4,4'-diaminoazobenzene

| Conditions | Yield |
|---|---|
| In methanol at 60 - 70℃; for 0.25h; Reflux; | 70% |
| With ethanol |
-
-
71190-35-7
1-(2,4-dinitrophenyl)-4,4'-bipyridinium chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| In ethanol | 70% |

| Conditions | Yield |
|---|---|
| Stage #1: C38H42N4O4 With benzotriazol-1-ol; O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; triethylamine In N,N-dimethyl-formamide at 4℃; for 1h; Inert atmosphere; Stage #2: 4,4'-Diaminoazobenzene In N,N-dimethyl-formamide at 4 - 24℃; for 24h; Inert atmosphere; | 70% |
-
-
39716-58-0
pent-4-enoyl chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
1204416-92-1
4-(pent-4-en)amido-4'-aminoazobenzene

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 68% |
-
-
79-03-8
propionyl chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
1204416-90-9
4-propionamido-4'-aminoazobenzene

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 68% |
-
-
71190-35-7
1-(2,4-dinitrophenyl)-4,4'-bipyridinium chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

-
B
-
97-02-9
2,4-Dinitroanilin

| Conditions | Yield |
|---|---|
| at 150 - 170℃; Zincke reaction; neat (no solvent); | A 68% B n/a |


-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In N,N-dimethyl-formamide for 10h; Heating; | 68% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 12h; Inert atmosphere; | 66% |
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In N,N-dimethyl-formamide for 18h; Inert atmosphere; | 58% |
-
-
141-75-3
butyryl chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
1204416-91-0
4-butyramido-4'-aminoazobenzene

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 65% |
-
-
74670-08-9
(3α,5β,7α,12α)-3,7,12-tris(formyloxy)cholan-24-oyl chloride

-
-
538-41-0
4,4'-Diaminoazobenzene

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0℃; | 63% |
-
-
13669-05-1
1,4-bis(thiophen-2-yl)butane-1,4-dione

-
-
538-41-0
4,4'-Diaminoazobenzene

-
-
425642-60-0
4,4'-bis-[(2,5-di-thien-2-yl)-pyrrol-1-yl]azobenzene

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 12h; Heating; | 60% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 12h; Inert atmosphere; | 58% |
P-Azoaniline Chemical Properties
Chemistry informtion about P-Azoaniline (CAS NO.538-41-0) is:
IUPAC Name: 4-(4-Aminophenyl)Diazenylaniline
Synonyms: 4,4'-Azobis-Benzenamin ; 4,4'-Azobisbenzenamine ; 4,4'-Azodi-Anilin ; P-Azoaniline ; 4,4'-Azodianiline ; 4,4'-Diaminoazobenzene ; P-Diaminoazobenzene ; 4,4'-Azobenzenediamine
MF: C12H12N4
MW: 212.25
EINECS: 208-690-7
Density: 1.23 g/cm3
Melting Point: 245 °C
Flash Point: 213 °C
Boiling Point: 428.5 °C at 760 mmHg
Vapour Pressure: 1.51E-07 mmHg at 25°C
Enthalpy of Vaporization: 68.36 kJ/mol
Merck: 14,2977
BRN: 745553
Following is the molecular structure of P-Azoaniline (CAS NO.538-41-0) is:
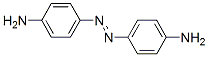
P-Azoaniline Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 180mg/kg (180mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#01451, |
P-Azoaniline Consensus Reports
Reported in EPA TSCA Inventory.
P-Azoaniline Safety Profile
Poison by intravenous route. Mutation data reported. When heated to decomposition it emits toxic vapors of NOx.
Hazard Codes:
 T
T
Risk Statements:
R40:Limited evidence of a carcinogenic effect.
R25 :Toxic if swallowed.
Safety Statements:
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S28:After contact with skin, wash immediately with plenty of soap-suds.
RIDADR: 2811
RTECS: BW7450000
HazardClass: 6.1
PackingGroup: III
P-Azoaniline Specification
P-Azoaniline (CAS NO.538-41-0) is a brown powder.
First Aid Measures
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin: Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion: Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Get medical aid immediately. Wash mouth out with water.
Inhalation: Get medical aid immediately. Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen.
Handling and Storage
Handling: Minimize dust generation and accumulation. Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Use only in a chemical fume hood.
Storage: Store in a cool, dry place. Store in a tightly closed container.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View