-
Name
Pentafluorophenol
- EINECS 212-235-8
- CAS No. 771-61-9
- Article Data79
- CAS DataBase
- Density 1.689 g/cm3
- Solubility soluble in water
- Melting Point 34-36 °C(lit.)
- Formula C6HF5O
- Boiling Point 146.3 °C at 760 mmHg
- Molecular Weight 184.065
- Flash Point 72.2 °C
- Transport Information
- Appearance white solid
- Safety 26-36-45-36/37/39-36/37-24/25-23-53
- Risk Codes 36/37/38-34-20/21/22-63-43-23/24/25-45
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, T,
T, C
C
- Synonyms Phenol,pentafluoro- (6CI,7CI,8CI,9CI);2,3,4,5,6-Pentafluorophenol;Hydroxypentafluorobenzene;NSC 21627;Pentafluorohydroxybenzene;Perfluorophenol;
- PSA 20.23000
- LogP 2.08770
Synthetic route
-
-
1256490-52-4
(S)-2-[(2,3,4,5,6-pentafluorophenoxy)phenoxyphosphorylamino]propionic acid isopropyl ester

-
A
-
56-41-7
L-alanin

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 25 - 60℃; for 4h; Reagent/catalyst; Temperature; Solvent; | A 94.2% B 95.7% C 98.2% |

-
-
848609-02-9
dimethyl pentafluorophenylboronate

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water; toluene at 10 - 20℃; for 14.08h; | 97.4% |

| Conditions | Yield |
|---|---|
| With water; potassium hydroxide at 160℃; under 4500.45 Torr; for 5h; Pressure; Reagent/catalyst; Temperature; | 97% |
| With water; sodium hydroxide at 120 - 125℃; for 2h; Temperature; Reagent/catalyst; Autoclave; | 95% |
| With N,N-didodecylamine; sodium hydroxide In water at 150℃; for 3h; Temperature; Autoclave; | 93.2% |

| Conditions | Yield |
|---|---|
| With tetraethoxy orthosilicate; dihydrogen peroxide; cetyltrimethylammonim bromide; copper(II) nitrate In acetonitrile at 65℃; for 3h; Solvent; Reagent/catalyst; Molecular sieve; | 96% |
| Stage #1: Pentafluorobenzene In 1,4-dioxane for 3h; Reflux; Stage #2: With dihydrogen peroxide; cobalt(II) acetate In 1,4-dioxane Reagent/catalyst; Reflux; | 94.9% |
| With sulfolane; potassium hydroxide In water at 120℃; under 6000.6 Torr; for 8h; Autoclave; Inert atmosphere; | 87.3% |
| Multi-step reaction with 3 steps 1.1: aluminum (III) chloride; bromine / 18 h / 45 °C 2.1: magnesium / tetrahydrofuran / 5 h / 40 - 45 °C / Inert atmosphere; Large scale 2.2: 2 h / -5 °C / Inert atmosphere; Large scale 2.3: 0.5 h / Inert atmosphere; Large scale 3.1: dihydrogen peroxide / water; tetrahydrofuran / 3 h / 70 °C View Scheme |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide at 20℃; for 14h; | 95.8% |
| With dihydrogen peroxide at 20℃; for 3h; | 92.6% |
-
-
267006-38-2
pentafluorophenylboronic acid isopropyl ester

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water; 1,3,5-trimethyl-benzene at 10 - 20℃; for 16.8h; | 95.7% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In ethanol at 40℃; for 0.0466667h; Temperature; Time; Solvent; Inert atmosphere; | 95.2% |
| With dihydrogen peroxide In tetrahydrofuran; water at 70℃; for 3h; Temperature; | 94.1% |
| With dihydrogen peroxide In toluene at 20 - 50℃; for 24h; Temperature; Solvent; Reagent/catalyst; | 85% |
| With dihydrogen peroxide at 20℃; Flow reactor; | 85.8% |
| With dihydrogen peroxide at 10 - 15℃; for 6h; Temperature; | 80% |
-
-
18595-35-2
2-(2,4-dinitrophenylthio)ethanol

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
1252553-78-8
2-(2,4-dinitrophenylthio)ethyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A n/a B 90% |


| Conditions | Yield |
|---|---|
| With copper(I) oxide; sodium carbonate; sodium hydroxide In water at 20 - 140℃; for 5h; Autoclave; | 89% |
| With sulfolane; palladium on activated charcoal; potassium hydroxide In water at 150℃; under 7500.75 Torr; for 10h; Reagent/catalyst; Temperature; Pressure; Autoclave; Inert atmosphere; | 84.7% |
| With potassium hydroxide In ammonia at -33℃; for 2h; | 8.2 mmol |
-
-
67-56-1
methanol

-
-
152342-45-5
2,4,5,6-tetrafluoro-3-(pentafluorophenoxy)-6-phenyl-2,4-cyclohexadienone

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| at 60℃; for 4h; | A n/a B 86% |


| Conditions | Yield |
|---|---|
| With copper(I) oxide; sodium carbonate; sodium hydroxide In water at 20 - 140℃; for 5h; Autoclave; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4,5,6-pentafluoroaniline With hydrogenchloride; tert.-butylnitrite In water at 0 - 5℃; for 1h; Stage #2: With copper(l) chloride In water; acetonitrile at 20 - 30℃; Reagent/catalyst; Solvent; Temperature; | 85% |
-
-
141-52-6
sodium ethanolate

-
-
140200-31-3
2,3,4,5,6-Pentafluoro-benzenesulfonic acid pentafluorophenyl ester

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
C
-
135736-88-8
Sodium; 2,4,6-triethoxy-3,5-difluoro-benzenesulfonate

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 10h; 1a:EtONa = 1:6; | A 81.5% B 50.1% C 61.9% |


| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In 1,2-dichloro-ethane at 50 - 60℃; for 4h; | 80% |
| With hydrogen iodide | |
| With aluminium trichloride at 120℃; | |
| With N,N,N,N-tetraethylammonium tetrafluoroborate In acetonitrile |
-
-
140200-31-3
2,3,4,5,6-Pentafluoro-benzenesulfonic acid pentafluorophenyl ester

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
135736-85-5
C6F4O4S(2-)*2Na(1+)

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 100℃; for 10h; | A 79.3% B 100 mg |
| With hydrogenchloride; sodium hydroxide 1) H2O, 100 degC, 10 h; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
10097-02-6
2,2-bis(hydroxymethyl)butanoic acid

-
-
59483-84-0
bis(pentafluorophenyl)carbonate

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
1252553-70-0
pentafluorophenyl 5-ethyl-2-oxo-1,3-dioxane-5-carboxylate

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran at 20℃; for 20h; | A n/a B 75% |

-
-
141-52-6
sodium ethanolate

-
-
140200-31-3
2,3,4,5,6-Pentafluoro-benzenesulfonic acid pentafluorophenyl ester

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 10h; 1a:EtONa = 1:1; | A 72.8% B 19% C 20.1% |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
627-18-9
1-bromo-3-propanol

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
1252553-75-5
3-bromopropyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A n/a B 70% |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
100-51-6
benzyl alcohol

-
A
-
247142-68-3
2-methyl-2-(benzyloxycarbonyl)-1,3-trimethylene carbonate

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A 68% B n/a |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
107-18-6
allyl alcohol

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
532424-75-2
5-methyl-5-allyloxycarbonyl-1 ,3-dioxane-2-one

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A n/a B 65% |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
107-19-7
propargyl alcohol

-
A
-
945841-51-0
propargyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran at 20℃; for 24h; | A 65% B n/a |

-
-
2162-31-4
2-(tetrahydropyran-2-yloxy)ethanol

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
1075204-22-6
2-(tetrahydro-2H-pyran-2-yloxy)ethyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A n/a B 65% |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
111625-28-6
2-(2-(Pyridin-2-yl)disulfanyl)ethanol

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A 65% B n/a |

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
-
26690-80-2
2-(N-tert-butoxycarbonylamino)ethanol

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A 65% B n/a |


-
-
188837-53-8
(2,3,4,5,6-pentafluorophenyl)pyridine-2-carboxylate

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; | A n/a B 64% |

-
-
64-17-5
ethanol

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
A
-
651718-45-5
2-ethoxycarbonyl-2-methyltrimethylene carbonate

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran | A 56% B n/a |

-
-
111-77-3
2-(2-methoxyethoxy)ethyl alcohol

-
-
1252553-69-7
pentafluorophenyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
A
-
1006903-36-1
2-(2-methoxyethoxy)ethyl 5-methyl-2-oxo-1,3-dioxane-5-carboxylate

-
B
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With cesium fluoride In tetrahydrofuran for 24h; | A 50% B n/a |

-
-
7722-84-1
dihydrogen peroxide

-
-
1076-44-4
pentafluorophenyl lithium

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With Trimethyl borate In tetrahydrofuran reaction in presence of (CH3O)3B at -70°C, 90 minutes;; | 39% |
| With (CH3O)3B In tetrahydrofuran reaction in presence of (CH3O)3B at -70°C, 90 minutes;; | 39% |
-
-
100-02-7
4-nitro-phenol

-
-
19220-93-0
pentafluorophenyl acetate

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
830-03-5
4-nitrophenol acetate

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 25℃; Rate constant; Mechanism; pH 7.00; |

-
-
392-56-3
Hexafluorobenzene

-
A
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
B
-
20994-04-1
4-hydroxy-2,3,5,6-tetrafluoronitrobenzene

| Conditions | Yield |
|---|---|
| With sodium nitrite In dimethyl sulfoxide at 160 - 170℃; for 5h; Yield given. Yields of byproduct given; |
-
-
700-16-3
Pentafluoropyridine

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
69585-26-8
4-pentafluorophenoxy-2,3,5,6-tetrafluoropyridine

| Conditions | Yield |
|---|---|
| With potassium fluoride; 18-crown-6 ether In acetonitrile at 80℃; for 1h; ratio of pentafluoropyridine and pentafluorophenol 1:0.1; | 100% |
| With cesium fluoride In N,N-dimethyl-formamide at 80℃; for 1h; Product distribution; other reagents, time; | 95% |
-
-
69620-40-2
2,6-bis-pentafluorophenoxy-3,4,5-trifluoropyridine

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With potassium fluoride; 18-crown-6 ether In acetonitrile at 80℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate at 20℃; for 1h; | 100% |
| With dicyclohexyl-carbodiimide In dichloromethane at 25℃; | 92% |
| With dicyclohexyl-carbodiimide In 1,4-dioxane for 2h; | 86.5% |
| With dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 1.75h; |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
64317-34-6
N-(pentafluorophenyl)carbonimidoyl dichloride

-
-
94055-40-0
C19F15NO2

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 80℃; for 5h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
71989-31-6
Fmoc-Pro-OH

-
-
125281-38-1, 86060-90-4
Fmoc-L-proline pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane for 1h; Esterification; | 100% |
| With dicyclohexyl-carbodiimide In ethyl acetate 0 deg C, 1 h; r.t., 1 h; | 82% |
| With dicyclohexyl-carbodiimide In ethyl acetate at 0℃; for 2h; Yield given; |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
74706-03-9
N-<1-(4-biphenylyl)-1-methylethoxycarbonyl>-2-(acetamidomethyl)-L-cysteine

-
-
73363-56-1
N<1-(4-biphenylyl)-1-methylethoxycarbonyl>-S-(acetamidomethyl)-L-cysteine pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate at 0℃; for 1h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
112471-83-7
Boc-Lys(Z)-Lys(Z)-Gly-OH

-
-
141914-54-7
Boc-Lys(Z)-Lys(Z)-Gly-OPFP

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide | 100% |
| With dicyclohexyl-carbodiimide | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
154627-63-1
5-tert-butoxycarbonylamino-2-phenyl-1,3-dioxane-5-carboxylic acid

-
-
154627-60-8
pentafluorophenyl 5-tert-butoxycarbonylamino-2-phenyl-1,3-dioxane-5-carboxylate

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at -20 - 20℃; for 16h; | 100% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -20 - 20℃; for 16h; | 99% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
141636-68-2
Fmoc-D-DMTP-Pro-OH

-
-
141636-69-3
Fmoc-D-DMPT-Pro-OPfp

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate; N,N-dimethyl-formamide at 5 - 20℃; for 3h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
138687-69-1
pentafluorophenyl diphenyl-phosphinate

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane for 1h; Ambient temperature; | 100% |
| With 1H-imidazole In dichloromethane at 25℃; for 3h; | 97% |
| With 1H-imidazole In dichloromethane Ambient temperature; | 92% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate for 2.5h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
69195-96-6
5-nitro-1-(phenylmethyl)-1H-imidazole-4-carboxylic acid

-
-
204527-99-1
1-Benzyl-5-nitro-1H-imidazole-4-carboxylic acid pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With ethyl acetate; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 0 - 20℃; for 3h; | 100% |
| With dicyclohexyl-carbodiimide In ethyl acetate; N,N-dimethyl-formamide 1) 0 deg C, 1 h; 2) rt, 1 h; | 95% |
| With dicyclohexyl-carbodiimide In ethyl acetate; N,N-dimethyl-formamide 1.) 0 deg C, 3 h, 2.) room temperature, 1 h; | 95% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
216481-09-3
2-((S)-1-{[2-({(S)-2-[(2-{6-Benzyloxymethyl-2-[2-((S)-1-tert-butoxycarbonylamino-ethyl)-5-methyl-oxazol-4-yl]-pyridin-3-yl}-thiazole-4-carbonyl)-amino]-3-methyl-butyrylamino}-methyl)-5-methyl-oxazole-4-carbonyl]-amino}-ethyl)-thiazole-4-carboxylic acid

-
-
216481-10-6
C51H50F5N9O10S2

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane | 100% |
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0 - 20℃; for 16h; Esterification; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide Esterification; | 100% |
-
-
35718-08-2
propargyl chloroformate

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
439912-32-0
propargyl pentafluorophenyl carbonate

| Conditions | Yield |
|---|---|
| With ethyl azide In diethyl ether at 0℃; for 7h; | 100% |
| With triethylamine In dichloromethane at -10℃; for 4h; | 98% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
683277-82-9
(2'-azido-biphenyl-2-yl)-acetic acid

-
-
683277-83-0
(2'-azido-biphenyl-2-yl)-acetic acid pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 100% |
-
-
683241-92-1
6-(4-(tert-butoxycarbonyl)piperazin-1-yl)-5-chloronicotinic acid

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
683241-96-5
tert-butyl 4-(3-chloro-5-((perfluorophenoxy)carbonyl)pyridin-2-yl)piperazine-1-carboxylate

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate at 20℃; for 16h; | 100% |
| With dicyclohexyl-carbodiimide In ethyl acetate at 0 - 20℃; for 16h; |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; | 100% |
| With 4-methyl-morpholine; dmap In dichloromethane at 0℃; for 0.55h; Inert atmosphere; | 89% |
| With pyridine In dichloromethane at 0 - 20℃; | 89% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 1h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
32857-63-9
4-t-butylphenylacetic acid

-
-
681810-54-8
pentafluorophenyl 2-(4-tert-butylphenyl)acetate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 16h; | 100% |
-
-
755014-13-2
3-methoxy-2-methylsulfanylmethyl-benzoic acid

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate for 1h; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
119222-62-7
Boc-D-Pen-OH

| Conditions | Yield |
|---|---|
| With 1,2-dichloro-ethane In dichloromethane | 100% |
-
-
914639-15-9
1,4,7,10-tetraazacyclododecane-1,7-bis(t-butyloxycarbonyl-methyl)-4,10-bis[2-(R)-pentanedioic acid, 1-t-butyl ester]

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
914639-12-6
1,4,7,10-tetraazacyclododecane-1,7-bis(t-butyloxycarbonyl-methyl)-4,10-bis[2-(R)-pentanedioic acid, 1-t-butyl-5-pentafluorophenyl ester]

| Conditions | Yield |
|---|---|
| With PS-CDI for 2h; Polystyrene; | 100% |
-
-
856223-02-4
(2S)-3-(4-benzyloxy-3'-{(2S)-2-[{(2S)-5-benzyloxycarbonylamino-2-tert-butoxycarbonylamino-pentanoyl}-(methyl)-amino]-3-methoxy-3-oxo-propyl}-4'-fluoro-biphenyl-3-yl)-2-benzyloxycarbonylamino-propionic acid

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
856223-15-9
pentafluorophenyl (2S)-3-(4-benzyloxy-3'-{(2S)-2-[{(2S)-5-benzyloxycarbonylamino-2-tert-butoxycarbonylamino-pentanoyl}-(methyl)-amino]-3-methoxy-3-oxo-propyl}-4'-fluoro-biphenyl-3-yl)-2-benzyloxycarbonylamino-propanoate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at -25 - 20℃; | 100% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
34439-17-3
bis[tris(trimethylsilylmethyl)molybdenum](Mo-Mo)

-
-
124461-95-6
Mo2(OC6F5)6

| Conditions | Yield |
|---|---|
| In benzene byproducts: SiMe4; under Ar atm.; soln. of C6F5OH added to complex, stirred for 48h; solvent removed (vac.), washed (hexane), dried; | 100% |

| Conditions | Yield |
|---|---|
| In hexane (inert atmosphere); -30°C slowly to room temp., stirring (2 h); evapn. to dryness, washing (hexane); | 100% |

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
90491-53-5
zirconocene bis(pentafluorophenoxide)

| Conditions | Yield |
|---|---|
| In diethyl ether; toluene (inert atmosphere); stirring (4 h); evapn. to dryness, washing (hexane), recrystn. (toluene/hexane); elem. anal.; | 100% |
-
-
12636-72-5
bis(cyclopentadienyl)dimethylzirconium(IV)

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
90491-53-5
zirconocene bis(pentafluorophenoxide)

| Conditions | Yield |
|---|---|
| In diethyl ether; toluene (inert atmosphere); stirring (2 h); evapn. to dryness, washing (hexane), recrystn. (toluene/hexane); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0 - 20℃; for 16h; Inert atmosphere; | 100% |
| With dicyclohexyl-carbodiimide In ethyl acetate at 20℃; for 12h; | |
| With diisopropyl-carbodiimide In ethyl acetate at 20℃; for 12h; |
-
-
919988-49-1
(2S)-3-[4-(benzyloxy)-3'-(2-{[(2S)-5-{[(benzyloxy)carbonyl]amino}-2-(tert-butoxycarbonylamino)pentanoyl]amino}-4-methoxy-4-oxobutyl)biphenyl-3-yl]-2-{[(benzyloxy)carbonyl]amino}propanoic acid

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
919988-50-4
methyl 4-{4'-(benzyloxy)-3'-[(2S)-2-{[(benzyloxy)carbonyl]amino}-3-oxo-3-(pentafluorophenoxy)propyl]biphenyl-3-yl}-3-({(2S)-5-{[(benzyloxy)carbonyl]amino}-2-[(tert-butoxycarbonyl)amino]pentanoyl}amino)butanoate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at -25 - 20℃; | 100% |
PENTAFLUOROPHENOL Chemical Properties
The density of Pentafluorophenol(771-61-9) is 1.757 and it has a melting point of 34-36 °C(lit.). The boiling point is 143 °C(lit.). The refractive index is about 1.4270. Its flash point is 162 °F.
The chemical synonyms of Pentafluorophenol(771-61-9) are PFP-OH;PERFLUOROPHENOL;PENTAFLUOROPHENOL;C6F5OH;pentafluoro-pheno;phenol,pentafluoro-;2,3,4,5,6-PENTAFLUOROHYDROXYBENZENE;2,3,4,5,6-PENTAFLUOROPHENOL
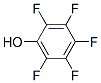
The molecular structure of Pentafluorophenol(771-61-9):
PENTAFLUOROPHENOL Uses
PENTAFLUOROPHENOL Toxicity Data With Reference
| 1. | scu-rat LD50:322 mg/kg | IZSBAI Izvestiya Sibirskogo Otdeleniya Akademii Nauk S.S.S.R., Seriya Biologomeditsinskikh Nauk. 3 (1965),91. | ||
| 2. | scu-mus LD50:283 mg/kg | IZSBAI Izvestiya Sibirskogo Otdeleniya Akademii Nauk S.S.S.R., Seriya Biologomeditsinskikh Nauk. 3 (1965),91. | ||
| 3. | orl-mam LD50:330 mg/kg | GISAAA Gigiena i Sanitariya. 45 (10)(1980),16. | ||
| 4. | skn-mam LD50:1120 mg/kg | GISAAA Gigiena i Sanitariya. 45 (10)(1980),16. |
PENTAFLUOROPHENOL Consensus Reports
PENTAFLUOROPHENOL Safety Profile
Hazard Codes Xi,T,C
Risk Statements 36/37/38-34-20/21/22-63-43-23/24/25-45
Safety Statements 26-36-45-36/37/39-36/37-24/25-23-53
RIDADR 2811
WGK Germany 3
RTECS SM6680000
F 3
Hazard Note Toxic/Irritant
TSCA T
HazardClass 8
PackingGroup III
HS Code 29081000
PENTAFLUOROPHENOL Specification
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Handling
Wash thoroughly after handling. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Wash clothing before reuse.
Incompatibilities
Oxidizing agents.
Stability
Stable under normal temperatures and pressures.
Decomposition
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen fluoride.
Combustion Products
Fire may produce irritating, corrosive and/or toxic gases.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View