-
Name
TRIMETHYLGALLIUM
- EINECS 215-897-6
- CAS No. 1445-79-0
- Article Data67
- CAS DataBase
- Density 1.151 g/cm3
- Solubility
- Melting Point -15.8 °C
- Formula C3H9Ga
- Boiling Point 55.7 °C
- Molecular Weight 114.827
- Flash Point -18 °C
- Transport Information UN 2845
- Appearance COA
- Safety 7-16-43-45
- Risk Codes 14-17-34
-
Molecular Structure
- Hazard Symbols F,C
- Synonyms Trimethylgallium ((CH3)3Ga);Trimethylgallane;
- PSA 0.00000
- LogP 1.37060
Synthetic route

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated slowly under 1E-2 mmHg in a trap-to-trap distillation apparatus (receiver flask at -196 °C), liberation of Me3Ga started at 50 °C, temp. was raised to 120 °C;; distillate contained benzene;; | 100% |

| Conditions | Yield |
|---|---|
| at 120 - 145℃; Solvent; Temperature; | A n/a B 98% |


| Conditions | Yield |
|---|---|
| With methylene chloride; sodium at 40 - 50℃; under 375.038 Torr; Inert atmosphere; Flow reactor; | A 93.7% B 96.8% |


| Conditions | Yield |
|---|---|
| With tridodecylamine In 5,5-dimethyl-1,3-cyclohexadiene at 100℃; for 1h; | 93.3% |
| Stage #1: gallium(III) trichloride With triethylaluminum Heating; Stage #2: trimethylaluminum at 160℃; | 85% |
| Stage #1: trimethylaluminum With triethylamine In toluene at 25 - 100℃; for 1h; Stage #2: gallium(III) trichloride In toluene Product distribution / selectivity; | 81% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated slowly under 1E-2 mmHg in a trap-to-trap distillation apparatus (receiver flask at -196 °C), liberation of Me3Ga started at 65 °C, proceeded rapidly at 120-130 °C;; contained traces of benzene;; | 93% |

| Conditions | Yield |
|---|---|
| Stage #1: gallium With magnesium at 60℃; for 1h; Inert atmosphere; Stage #2: methyl iodide In 1,3,5-trimethyl-benzene at 60 - 190℃; for 3.66h; | 92% |
| With magnesium In neat (no solvent) byproducts: MgI2; inert atm.; heating (autoclave, 120-160°C, 12 h); distn.; | 65% |
-
-
7440-55-3
gallium

-
-
917-64-6
methyl magnesium iodide

-
-
7553-56-2
iodine

-
-
74-88-4
methyl iodide

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| Stage #1: gallium; iodine; methyl iodide In toluene at 40 - 50℃; for 6h; Inert atmosphere; Sonication; Stage #2: methyl magnesium iodide In toluene at 100 - 110℃; for 5h; Inert atmosphere; | 92% |


| Conditions | Yield |
|---|---|
| at 20 - 140℃; Glovebox; Inert atmosphere; | 92% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated slowly under 1E-2 mmHg in a trap-to-trap distillation apparatus (receiver flask at -196 °C), liberation of Me3Ga started at 85 °C, the rate was more satisfactory at 110-130 °C;; | 91% |

| Conditions | Yield |
|---|---|
| Reflux; | 90% |
| In neat (no solvent) warming the mixt. of GaCl3 and trimethylaluminum from 233 K to room temp., stirring for 90 min; vaporization of volatiles in vac., fractional condensation at 178 K; IR; | |
| Stage #1: trimethylaluminum at 80 - 85℃; under 187.519 Torr; for 0.5h; Stage #2: Gallium trichloride at 80 - 90℃; for 0.5h; |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 110-120°C for 2-3 h; | 90% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 100-110°C for 2-3 h; | 88% |
| In neat (no solvent) heated slowly under 1E-2 mmHg in a trap-to-trap distillation apparatus (receiver flask at -196 °C), liberation of Me3Ga started at 80 °C, the rate was more satisfactory at 90 °C;; | 71% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 110-120°C for 2-3 h; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: gallium(III) trichloride; dimethylaluminum chloride In toluene Stage #2: With triethylamine In toluene at 100℃; for 1h; Product distribution / selectivity; | 82% |

| Conditions | Yield |
|---|---|
| In diethyl ether at -10 - -5℃; for 10h; Large scale; | 80% |

| Conditions | Yield |
|---|---|
| With potassium chloride; sodium chloride at 120℃; Inert atmosphere; | A 25.6% B 71.3% |
| With potassium chloride; sodium chloride at 120℃; Inert atmosphere; | A 25.6% B 69.6% |


| Conditions | Yield |
|---|---|
| With potassium chloride; sodium chloride at 120 - 155℃; Concentration; Temperature; Inert atmosphere; | A 24.7% B 69.6% |
| With potassium chloride; sodium chloride at 70 - 180℃; Concentration; Temperature; Inert atmosphere; | A 27.4% B 68% |

-
-
7440-55-3
gallium

-
-
7439-95-4
magnesium

-
-
74-88-4
methyl iodide

-
A
-
7786-30-3
magnesium chloride

-
B
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) inert gas atmosphere; mixing, heating in autoclave (100 - 160°C, stirring, 12 h); distillation; | A n/a B 65% |


| Conditions | Yield |
|---|---|
| In diethyl ether; pentane byproducts: LiCl; under Ar, excess of LiH in Et2O suspn. was added to Et2O soln. of Me2GaCl, reflux for 20 h, filtn., evapn. in vac. at room temp., drying at 45 °C (1E-3 Torr), solid was suspended in n-pentane, Me2GaCl was added in n-pentane, reflux for 16 h; soln. was filtered, residue was twice washed with n-pentane, combined filtrate was evapd., oily residue was distd. in vac. at room temp. (1E-3 Torr) with a cooled (-40 °C) collector, Me3Ga was collected at liq. N2 temp.; | A 49% B n/a |

-
-
348089-02-1
1,1'-bis(dimethylgallyl)ferrocene

-
A
-
561301-67-5
[(Fe(η5-C5H4)2)2(GaMe)2]

-
B
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In diethyl ether; toluene under Ar atm. Fe(C5H4GaMe2)2 was treated with Et2O and toluene at room temp. and cooled to +6°C; ppt. was washed with n-hexane and dried in vacuo; | A 46% B n/a |
| In chloroform under Ar atm. Fe(C5H4GaMe2)2 was dissolved in CHCl3 and cooled to +6°C; elem. anal.; |

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride at 80 - 120℃; |

| Conditions | Yield |
|---|---|
| With gallium at 110 - 130℃; |


| Conditions | Yield |
|---|---|
| With methyl magnesium iodide; methyl iodide In 1,4-dioxane byproducts: MgI2; excess of CH3MgI and CH3I, heated at 101°C; | |
| With methyl magnesium iodide; methyl iodide In tetrahydrofuran byproducts: MgI2; excess of CH3MgI and CH3I, heated at 65°C; | |
| With methyl magnesium iodide; methyl iodide In di-isopropyl ether byproducts: MgI2; excess of CH3MgI and CH3I, heated at 69°C; |

| Conditions | Yield |
|---|---|
| according to A. Storr, B. S. Thomas, Can. J. Chem. 48 (1970) 3667; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) vigorously starting reaction, completed at 80 - 120 °C;; |

| Conditions | Yield |
|---|---|
| In not given reaction of excess GaCl3 with Zn(CH3)2;; |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| With methyl iodide In further solvent(s) under N2, MeI added slowly to stirred suspn. of Mg5Ga2 in di-isopentyl ether, react. initiated with I2, stirred for 18 h, refluxed for 24 h, cooled to ambient temp.; distn. at bath temp. of 190°C; | 30-50 |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| With methylmagnesium bromide GaCl3 is treated with a three-fold excess of MeMgBr; elem. anal.; |

| Conditions | Yield |
|---|---|
| In not given inert atm., PhMe or hexane as solvent, equimolar ratio; | 100% |
| In toluene (under dry nitrogen); t-butyl amine added to stirred soln. of trimethylgallium at -78°C; warmed to 25°C; stirred for 24 h; evapd. (vac.); sublimed (65-67°C; E-3 Torr); |
-
-
1445-79-0
trimethyl gallium

-
-
66622-03-5
dimethyl(methylperoxy)gallium

| Conditions | Yield |
|---|---|
| With oxygen In n-heptane | 100% |
| With oxygen In not given according to: Yu. A. Aleksandrov, G. I. Makin et al., Zh. Obshch. Khim.48 (1978) 467; oxidn. of Ge-compd. with molecular oxygen in hydrocarbonmedium at -78°C; | 100% |
| With O2 In neat (no solvent) heated from -120°C -70°C; |
-
-
1453-58-3
3-Methylpyrazole

-
-
1445-79-0
trimethyl gallium

-
-
79422-26-7
dimethyl-(3-methyl-pyrazol-1-yl)-gallium

| Conditions | Yield |
|---|---|
| In benzene byproducts: CH4; under argon atm., reflux, 1 h; evapn. of solvent; | 100% |

| Conditions | Yield |
|---|---|
| In not given N2-atmosphere; equimolar amts.; can be recrystallized (pentane, low temp.) or sublimated (55°C, 1E-2 mbar); | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) all manipulations under N2 atm.; mixed equimolar amts. of Ga and Bi compds. at ambient temp.; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) all manipulations under N2 atm.; mixed equimolar amts. of Ga and Bi compds. at ambient temp.; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) under N2 atm. react. Me3Ga and Et4Sb2; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In benzene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 64% |
-
-
32318-29-9
tris(diethylamino)difluorophosphorane

-
-
1445-79-0
trimethyl gallium

-
-
222190-85-4
[FP(N(C2H5)2)3](1+)*[(CH3)3GaF](1-)=[FP(N(C2H5)2)3][(CH3)3GaF]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Ar atm.; equimolar amts., stirring (20°C, 24 h); evapn. (vac.); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In benzene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 73% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In benzene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 67% |
-
-
234443-33-5
[(CH3)2SnN(CH2CH(CH3)2)]3

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In benzene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 76% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In not given inert atm., PhMe or hexane as solvent, equimolar ratio; | 100% |
-
-
163631-37-6
(2,6-iPr2C6H3)N(SiMe3)Si(OH)3

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; hexane byproducts: CH4; molar ratio Ga:ligand 2:1, ratio n-hexane/1,4-dioxane 10/1, addn. (room temp.), refluxing (1 h), pptn.; crystn. (12 h), concn.; elem. anal.; | 100% |
-
-
163631-37-6
(2,6-iPr2C6H3)N(SiMe3)Si(OH)3

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; hexane byproducts: CH4; equimolar ratio, ratio n-hexane/1,4-dioxane 10/1, stirring (10 min, roomtemp.), refluxing (1 h); evapn. (vac.); elem. anal.; | 100% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; hexane byproducts: CH4; molar ratio Ga:ligand 2:1, ratio n-hexane/1,4-dioxane 10/1, addn. (room temp.), refluxing (1 h), pptn.; crystn. (12 h), concn.; elem. anal.; | 100% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; hexane byproducts: CH4; equimolar ratio, ratio n-hexane/1,4-dioxane 10/1, stirring (10 min, roomtemp.), refluxing (1 h); evapn. (vac.); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In benzene byproducts: CH4; under argon atm., reflux, 1 h; evapn. of solvent; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane (inert atm.), Me3Ga injected to a stirred suspn. of ligand in CH2Cl2 at -76°C, slowly warmed to room temp.; evapd.(vac.), elem. anal.; | 100% |
-
-
37486-48-9
N,N'-di-tert-butyloxamide

-
-
1445-79-0
trimethyl gallium

-
-
1309357-34-3, 1309357-37-6
(N,N-di-tert-butyloxamidate)Ga2Me4

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (inert atm.), Me3Ga injected to a stirred suspn. of ligand in THF at room temp., refluxed for 1 h; evapd.(vac.), elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at -50℃; for 1h; Inert atmosphere; Schlenk technique; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at -70 - 20℃; for 1h; Inert atmosphere; Schlenk technique; | 100% |
-
-
724794-67-6
1-(2,6-diisopropylphenyl)-3-(2-hydroxyphenyl)-4,5-dihydro-imidazolyl chloride

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In dichloromethane at -70 - 20℃; for 1h; Inert atmosphere; Schlenk technique; | 100% |

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 0.166667h; Inert atmosphere; Schlenk technique; | 100% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 42℃; for 24h; Temperature; Schlenk technique; Inert atmosphere; | 100% |

-
-
1445-79-0
trimethyl gallium

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; for 72h; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at -50 - 20℃; Inert atmosphere; Schlenk technique; | 100% |
Trimethylgallium Specification
The Trimethyl gallium with CAS registry number of 1445-79-0 is also called Gallium, trimethyl-. Its EINECS registry number is 215-897-6. The IUPAC name is trimethylgallane. In addition, the molecular formula is C3H9Ga and the molecular weight is 114.83.
Physical properties about this chemical are: (1)Exact Mass: 113.996006; (2)MonoIsotopic Mass: 113.996006; (3)Heavy Atom Count: 4; (4)Complexity: 8; (5)Covalently-Bonded Unit Count: 1.
Preparation of Trimethyl gallium: The methylmagnesium iodide can react with gallium trichloride to give trimethylgallium ether. Then trimethylgallium ether can disintegrate to get the product. And you get the high purity trimethyl gallium through the operation of distillation, separation, distillation and purification.
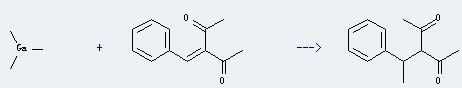
Uses of Trimethyl gallium: this chemical can be used for metal organic chemical vapor deposition (MOCVD) and in preparation of semiconductor compounds such as GaAs and AsGaAl. Besides, it can be used in the manufacture of solar cells. In addition, it can react with benzylideneacetone to get 3-(1-phenylethyl)pentane-2,4-dione. This reaction will need solvent diethyl ether. The yield is about 71%.
When you are using this chemical, please be cautious about it as the following:
It is spontaneously flammable in air and can react violently with water. And it can cause burns. In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk add - Never use water). And in case of accident or if you feel unwell, seek medical advice immediately (show label where possible). You should keep container tightly closed and keep away from sources of ignition - No smoking.
You can still convert the following datas into molecular structure:
(1)SMILES: [Ga](C)(C)C
(2)InChI: InChI=1/3CH3.Ga/h3*1H3;/rC3H9Ga/c1-4(2)3/h1-3H3
(3)InChIKey: XCZXGTMEAKBVPV-YHXBHQJBAF
Related Products
- Trimethylgallium
- 144584-29-2
- 144584-65-6
- 144584-66-7
- 144584-67-8
- 1445-91-6
- 14459-29-1
- 14459-33-7
- 144598-75-4
- 14459-95-1
- 144599-95-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View