-
Name
Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate
- EINECS
- CAS No. 120801-75-4
- Article Data8
- CAS DataBase
- Density 1.333 g/cm3
- Solubility Reacts with water.
- Melting Point
- Formula C5H9F3O4SSi
- Boiling Point 160.4 °C at 760 mmHg
- Molecular Weight 250.271
- Flash Point 50.8 °C
- Transport Information UN 2920 8/PG 2
- Appearance
- Safety 16-26-27-36/37/39
- Risk Codes 10-34
-
Molecular Structure
-
Hazard Symbols
 C,
C,  F,
F,  Xi
Xi
- Synonyms Trimethylsilyl2-fluorosulfonyl-2,2-difluoroacetate;Aceticacid, difluoro(fluorosulfonyl)-, trimethylsilyl ester (9CI);Trimethylsilyl(fluorosulfonyl)difluoroacetate;Trimethylsilyl2,2-difluoro-2-(fluorosulfonyl)acetate;
- PSA 68.82000
- LogP 2.33740
Synthetic route
-
-
107-46-0
Hexamethyldisiloxane

-
-
677-67-8
fluorosulfonyldifluoroacetyl fluoride

-
A
-
420-56-4
trimethylsilyl fluoride

-
B
-
1717-59-5
2,2-difluoro-2-(fluorosulfonyl)acetic acid

-
C
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| Stage #1: Hexamethyldisiloxane; fluorosulfonyldifluoroacetyl fluoride at 0 - 100.6℃; under 4350.44 - 5925.59 Torr; for 18h; Stage #2: With chloro-trimethyl-silane at 20 - 107.8℃; Product distribution / selectivity; Heating / reflux; | A n/a B n/a C 85% |

-
-
107-46-0
Hexamethyldisiloxane

-
-
677-67-8
fluorosulfonyldifluoroacetyl fluoride

-
A
-
420-56-4
trimethylsilyl fluoride

-
B
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane at 105℃; under 4200.42 Torr; for 6h; | A n/a B 84% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
1717-59-5
2,2-difluoro-2-(fluorosulfonyl)acetic acid

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| at 0 - 20℃; for 26h; Inert atmosphere; | 83% |
| at 20℃; | 78% |
| at 0℃; Esterification; | 78% |
| for 12h; Heating; | |
| at 0 - 65℃; for 19h; Inert atmosphere; |
-
-
16029-98-4
trimethylsilyl iodide

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| for 24h; -196 deg C upto room temp.; | 29% |
-
-
53300-46-2
N-(cyclohexylthiocarbonyl)aniline

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene at 80℃; for 0.166667h; Temperature; Solvent; Optical yield = 58 %de; | 100% |
-
-
22007-40-5
4-Methoxyphenylthiocarbamidsaeure-O-isopropylester

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene at 50℃; for 0.166667h; | 97% |
-
-
73568-25-9
2-chloro-3-quinoline carboxaldehyde

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-3-quinoline carboxaldehyde With sodium fluoride; triphenylphosphine In ethyl acetate at 90℃; for 0.0166667h; Wittig Olefination; Schlenk technique; Inert atmosphere; Stage #2: trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate In ethyl acetate at 90℃; for 1h; Schlenk technique; Inert atmosphere; | 96% |

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene at 60℃; for 0.25h; | 96% |
| With 1,1,1,3,3,3-hexafluoro-2,2-di(p-tolyl)propane; N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In para-xylene at 60℃; for 0.25h; regioselective reaction; |
-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
20442-66-4
3-(4-methoxyphenyl)-1-phenylprop-2-yn-1-one

-
-
882182-82-3
[3,3-difluoro-2-(4-methoxy-phenyl)-cycloprop-1-enyl]-phenyl-methanone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 3h; | 95% |
-
-
50555-04-9
cinnamyl benzoate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With sodium fluoride In various solvent(s) at 120℃; | 94% |

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene for 0.5h; Barton-Kellog Olefination; Schlenk technique; Reflux; | 94% |
-
-
4891-38-7
phenylpropynoic acid methyl ester

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; | 94% |
-
-
4265-16-1
2-formylbenzo[b]furan

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1449521-09-8
2-(2,2-difluorovinyl)benzo[b]furan

| Conditions | Yield |
|---|---|
| Stage #1: 2-formylbenzo[b]furan With sodium fluoride; triphenylphosphine In ethyl acetate at 90℃; for 0.0166667h; Wittig Olefination; Schlenk technique; Inert atmosphere; Stage #2: trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate In ethyl acetate at 90℃; for 1h; Schlenk technique; Inert atmosphere; | 93% |
-
-
13509-41-6
methyl N-phenylthiocarbamate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene at 50℃; for 0.166667h; | 93% |
-
-
139225-17-5
1-pentyl-1-hexenyl acetate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
74667-07-5
5,6-difluoromethylene-6-acetoxyundecane

| Conditions | Yield |
|---|---|
| With sodium fluoride In various solvent(s) at 120℃; | 92% |
-
-
57179-69-8
3,6-dimethyl-5-nitropyridin-2-ol

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1188407-31-9
2-(difluoromethoxy)-3,6-dimethyl-5-nitropyridine

| Conditions | Yield |
|---|---|
| Stage #1: 3,6-dimethyl-5-nitropyridin-2-ol With sodium hydride In acetonitrile at 20℃; for 0.5h; Stage #2: trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate With cesium fluoride In acetonitrile at 23 - 30℃; | 92% |
| With sodium hydride; cesium fluoride In acetonitrile at 23 - 30℃; for 1.66667h; Inert atmosphere; | 92% |
-
-
57179-69-8
3,6-dimethyl-5-nitropyridin-2-one

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1188407-31-9
2-(difluoromethoxy)-3,6-dimethyl-5-nitropyridine

| Conditions | Yield |
|---|---|
| With sodium hydride; cesium fluoride In acetonitrile; mineral oil at 20℃; for 1.16667h; | 92% |

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene at 60℃; for 0.25h; | 92% |
| With 1,1,1,3,3,3-hexafluoro-2,2-di(p-tolyl)propane; N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In para-xylene at 60℃; for 0.25h; regioselective reaction; |
-
-
90982-74-4
phenyl m-chlorobenzenedithiocarboxylate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In toluene for 0.5h; Barton-Kellog Olefination; Schlenk technique; Reflux; | 92% |
-
-
28489-45-4
6-methyl-5-nitropyridin-2-ol

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1224432-76-1
6-(difluoromethoxy)-2-methyl-3-nitropyridine

| Conditions | Yield |
|---|---|
| With sodium hydride; cesium fluoride In acetonitrile | 91% |
-
-
1429215-66-6
C17H14O

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1429215-44-0
phenyl 2,2-difluoro-3-(3,5-dimethylphenyl)cyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 91% |
-
-
3132-99-8
m-bromobenzoic aldehyde

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
84750-92-5
1-bromo-3-(2,2-difluorovinyl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: m-bromobenzoic aldehyde With sodium fluoride; triphenylphosphine In ethyl acetate at 90℃; for 0.0166667h; Wittig Olefination; Schlenk technique; Inert atmosphere; Stage #2: trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate In ethyl acetate at 90℃; for 1h; Schlenk technique; Inert atmosphere; | 91% |
-
-
873-66-5
1-propenylbenzene

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With sodium fluoride In various solvent(s) at 105℃; | 90% |
-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
671796-41-1
1-(4-(tert-butyl)phenyl)-3-phenylprop-2-yn-1-one

-
-
882182-78-7
4-t-butylphenyl 2,2-difluoro-3-phenylcyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 90% |
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 3h; | 85% |
-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
14939-05-0
1-phenyl-3-(4-methylphenyl)prop-2-yn-1-one

-
-
882182-83-4
phenyl 2,2-difluoro-3-(4-methylphenyl)cyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 3h; | 90% |
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 87% |
-
-
530-93-8
1,2,3,4-tetrahydronaphthalen-2-one

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
712-79-8
2-(difluoromethoxy)naphthalene

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,3,4-tetrahydronaphthalen-2-one; trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate With sodium carbonate; 1,3-bis(mesityl)imidazolium chloride In toluene at 100℃; for 1h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In toluene at 100℃; | 90% |
-
-
583-04-0
vinyl benzoate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With sodium fluoride at 105℃; | 89% |
-
-
18203-32-2
3-butenyl benzoate

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
117388-09-7
2-(2,2-difluorocyclopropyl)ethyl benzoate

| Conditions | Yield |
|---|---|
| With sodium fluoride at 105℃; | 89% |
| With sodium fluoride at 110℃; for 2h; Inert atmosphere; | 51% |
| With sodium fluoride In neat (no solvent) at 110℃; for 2h; | 51% |
| With sodium fluoride at 110℃; for 12h; | 50% |
| With sodium fluoride at 125℃; for 12h; Cycloaddition; |
-
-
7338-94-5
1,3-diphenyl-2-propynone

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
882182-77-6
phenyl 2,2-difluoro-3-phenylcyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 3h; | 89% |
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 85% |
-
-
14674-99-8
1-phenyl-3-(3-methylphenyl)prop-2-yn-1-one

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1429215-43-9
phenyl 2,2-difluoro-3-(3-methylphenyl)cyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 89% |

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

| Conditions | Yield |
|---|---|
| With sodium hydride; cesium fluoride In acetonitrile; mineral oil at 0℃; for 1h; | 89% |
-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
39833-48-2
3-(4-bromophenyl)-1-phenylprop-2-yn-1-one

-
-
882182-84-5
phenyl 2,2-difluoro-3-(4-bromophenyl)cyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 3h; | 88% |
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 85% |
-
-
91873-75-5
1-<<2-cyclohexylideneethenyl>sulfonyl>-4-methylbenzene

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
926033-49-0
1,1-difluoro-2-[(tolyl-4-sulfonyl)methylidene]spiro[2.5]octane

| Conditions | Yield |
|---|---|
| With sodium fluoride In xylene for 3.16667h; Heating; | 88% |
-
-
1429215-67-7
C23H26O

-
-
120801-75-4
trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate

-
-
1429215-45-1
phenyl 2,2-difluoro-3-(3,5-di-t-butylphenyl)cyclopropenyl ketone

| Conditions | Yield |
|---|---|
| With sodium fluoride In diethylene glycol dimethyl ether at 120℃; for 1h; Inert atmosphere; | 88% |
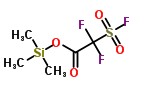
Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate Chemical Properties
Molecular structure of Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate (CAS NO.120801-75-4) is:
Product Name: Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate
CAS Registry Number: 120801-75-4
Empirical Formula: C5H9F3O4SSi
Molecular Weight: 250.2683
Surface Tension: 25.4 dyne/cm
Density: 1.333 g/cm3
Flash Point: 50.8 °C
Enthalpy of Vaporization: 39.7 kJ/mol
Boiling Point: 160.4 °C at 760 mmHg
Vapour Pressure: 2.39 mmHg at 25°C
Refractive index: n20/D 1.367(lit.)
Product Categories: C-C Bond Formation;Others;Synthetic Reagents
Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate Safety Profile
Safty information about Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate (CAS NO.120801-75-4) is:
Hazard Codes:  C;
C;  F;
F;  Xi
Xi
Risk Statements: 10-34
R10:Flammable.
R34:Causes burns.
Safety Statements: 16-26-27-36/37/39
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S27:Take off immediately all contaminated clothing.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
RIDADR: UN 2920 8/PG 2
WGK Germany: 2
Hazard Note: Flammable/Corrosive
HazardClass: 8
PackingGroup: II
Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate Specification
Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate , its cas register number is 120801-75-4. It also can be called Trimethylsilyl 2,2-difluoro-2-(fluorosulfonyl)acetate .
Related Products
- Trimethylsilyl 2-(fluorosulfonyl)difluoroacetate
- Trimethylsilyl acetate
- Trimethylsilyl bromoacetate
- Trimethylsilyl chlorosulphate
- Trimethylsilyl cyanide
- Trimethylsilyl isobutyrate
- Trimethylsilyl methacrylate
- Trimethylsilyl methanesulfonate
- Trimethylsilyl perchlorate
- Trimethylsilyl propionate
- 12080-32-9
- 1208-03-3
- 1208082-24-9
- 120-80-9
- 12081-16-2
- 1208-12-4
- 120812-48-8
- 120815-05-6
- 12082-08-5
- 120-82-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View