-
Name
Trimethylsilylacetylene
- EINECS 213-919-9
- CAS No. 1066-54-2
- Article Data65
- CAS DataBase
- Density 0.752 g/cm3
- Solubility reacts with water
- Melting Point >0 °C
- Formula C5H10Si
- Boiling Point 53 °C at 760 mmHg
- Molecular Weight 98.2199
- Flash Point <?30°F
- Transport Information UN 1993 3/PG 2
- Appearance clear colorless liquid
- Safety 16-26-36
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Silane, ethynyltrimethyl-;
- PSA 0.00000
- LogP 1.49700
Synthetic route

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride In pentane at 25℃; for 0.5h; | A n/a B 100% |
-
-
13683-41-5
1-bromo-1-(trimethylsilyl)ethene

-
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dimethyl sulfoxide at 40 - 110℃; for 2h; Solvent; | 94% |
| tetrakis(triphenylphosphine) palladium(0) | |
| tetrakis(triphenylphosphine) palladium(0) Mechanism; |

| Conditions | Yield |
|---|---|
| Stage #1: chloro-trimethyl-silane; lithium acetylide; 4,4'-di-tert-butylbiphenyl In tetrahydrofuran at 0℃; for 2h; Stage #2: With water In tetrahydrofuran at 20℃; | 91.4% |

| Conditions | Yield |
|---|---|
| With diethylene glycol diethyl ester at 50℃; for 2h; | 80% |
| In tetrahydrofuran; xylene for 24h; | 75% |

| Conditions | Yield |
|---|---|
| Stage #1: acetylene With ethidium Bromide; magnesium; ethylene dibromide at 5 - 10℃; Grignard reaction; Stage #2: chloro-trimethyl-silane at 15 - 35℃; for 1h; Substitution; | 80% |
| Stage #1: acetylene With butyl magnesium bromide In tetrahydrofuran at -5 - 20℃; for 1.5h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran at 15 - 20℃; for 1h; Inert atmosphere; Reflux; | 75% |
| With n-butyllithium In tetrahydrofuran; hexane at -78 - 25℃; for 10h; | |
| With n-butyllithium 1.) THF, hexane, -78 deg C, 10 min, 2.) -78 deg C, 2.5 h; room temp., 2 h; Multistep reaction; | |
| With sodium 1.) anisole, 110 deg C, 2.) 24 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; | 77% |
-
-
19061-00-8
bis(trimethylsilyl)ketene

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; flash vacuum pyrolysis; | A 22% B 16% C 76% |

-
-
98991-82-3
(trimethylsilyl)(dimethylsilyl)ketene

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; | A 22% B 16% C 76% |


| Conditions | Yield |
|---|---|
| In acetonitrile at 100℃; for 1h; | 74% |
-
-
16029-98-4
trimethylsilyl iodide

-
-
994-39-8
triethylethynylstannane

-
A
-
2943-86-4
triethyltin iodide

-
B
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| In acetonitrile mixt. heating for 1 h at 100°C; solvent washing out (water), Si-compd. isolation (vac. distn.); | A n/a B 74% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
1066-26-8
sodium acetylide

-
A
-
74-86-2
acetylene

-
B
-
1066-54-2
trimethylsilylacetylene

-
C
-
14630-40-1
Bis(trimethylsilyl)ethyne

| Conditions | Yield |
|---|---|
| In various solvent(s) at 100℃; | A 0.232 mol B 29% C 50% |

-
-
54655-07-1
lithium trimethylsilylacetylenide

-
A
-
4526-07-2
1,4-bis(trimethylsilyl)-1,3-butadiyne

-
B
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide In diethyl ether at -78℃; for 1h; Product distribution; | A 44% B 25% |

| Conditions | Yield |
|---|---|
| In chloroform for 4h; Heating; Yields of byproduct given; | A 41% B n/a |


| Conditions | Yield |
|---|---|
| In (2)H8-toluene at 20℃; for 1.25h; Equilibrium constant; Inert atmosphere; | A 38% B n/a |

-
-
52673-44-6
Dimethyl-oxypropyl-ethinyl-silan

-
A
-
4180-02-3
1,3-diethynyltetramethyldisiloxane

-
B
-
57914-14-4
Methyl-di-oxypropyl-ethinyl-silan

-
C
-
57914-15-5
Tri-oxypropyl-ethinyl-silan


-
F
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With hydrogen sulfide for 8h; Product distribution; Irradiation; var. time, traces of HCl and H2O; | A n/a B n/a C n/a D 30.1% E n/a F n/a |

-
-
32957-37-2
dimethylethoxyethynylsilane

-
A
-
5700-28-7
triethoxyethynylsilane

-
B
-
4180-02-3
1,3-diethynyltetramethyldisiloxane


-
E
-
103084-89-5
diethoxyethynylmethylsilane

-
F
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With hydrogen sulfide for 4h; Product distribution; Irradiation; var. time, traces of HCl and H2O; | A n/a B n/a C 13.7% D n/a E n/a F n/a |

-
-
14630-40-1
Bis(trimethylsilyl)ethyne

-
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With iron(III) chloride In octane at 25℃; Product distribution; | 5.5% |

| Conditions | Yield |
|---|---|
| With phenyllithium |
-
-
62827-97-8
Bis(trimethylsilyl)thioketene

-
A
-
7796-59-0
tetramethylcyclodisilthiane

-
C
-
1066-54-2
trimethylsilylacetylene

-
D
-
14630-40-1
Bis(trimethylsilyl)ethyne

| Conditions | Yield |
|---|---|
| at 768℃; flash vacuum pyrolysis; | A 4 % Chromat. B 17 % Chromat. C 11 % Chromat. D 17 % Chromat. |

-
-
13139-83-8
[3H]ethynyl-trimethyl-silane

-
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With morpholine buffer; water at 25℃; Rate constant; |
-
-
93641-74-8
N,N-dimethyl-3-(trimethylsilyl)propiolamide

-
A
-
124-40-3
dimethyl amine

-
B
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With water; Triethylgermyllithium Product distribution; 1.) -78 deg C, 1.5 h; |
-
-
98991-82-3
(trimethylsilyl)(dimethylsilyl)ketene

-
A
-
47956-45-6
dimethylsilanone

-
B
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; Mechanism; |
-
-
87514-35-0
N,N-diethyl-3-trimethylsilyl-2-propiolamide

-
A
-
109-89-7
diethylamine

-
B
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With water; Triethylgermyllithium Product distribution; 1.) -78 deg C, 1.5 h; |

-
A
-
3574-04-7
hexamethylcyclotrisilathiane

-
B
-
70740-65-7
2,2,4,4,5,5-Hexamethyl-1,3-dithia-2,4,5-trisilacyclopentan

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; flash vacuum pyrolysis; | A 7 % Chromat. B n/a C 92 % Chromat. |
| at 700℃; flash vacuum pyrolysis; | |
| With flash vacuum pyrolysis at 700℃; |

-
-
75-77-4
chloro-trimethyl-silane

-
-
925-90-6
ethylmagnesium bromide

-
-
74-86-2
acetylene

-
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 1h; |


| Conditions | Yield |
|---|---|
| With water; lithium 1.) hexane, 0 deg C, 12 h; |
-
-
38274-16-7
2-bromo-1-(trimethylsilylethynyl)benzene

-
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol for 1h; Ambient temperature; Yield given; |
-
-
210471-93-5
1,1-diethynyl-2,2,2-trimethyldisilane

-
A
-
460-12-8
Butadiyne

-
B
-
100899-98-7
1-silacyclopropenylidene

-
E
-
74-86-2
acetylene

-
F
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| In gaseous matrix at 1200℃; Product distribution; pulsed flash pyrolysis study; |

-
-
536-74-3
phenylacetylene

-
-
14630-40-1
Bis(trimethylsilyl)ethyne

-
A
-
2170-06-1
1-Phenyl-2-(trimethylsilyl)acetylene

-
B
-
74-86-2
acetylene

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| (Me2SiCp''2UBu)2O*2THF In tetrahydrofuran at 66℃; for 4h; Product distribution; Further Variations:; reaction time; | A 172 mg B n/a C 31 mg |

-
-
3437-95-4
2-Iodothiophene

-
-
1066-54-2
trimethylsilylacetylene

-
-
40231-03-6
2-(trimethylsilylethynyl)thiophene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 2h; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; trimethylamine for 0.5h; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; Reflux; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; | 99% |
-
-
109-04-6
2-bromo-pyridine

-
-
1066-54-2
trimethylsilylacetylene

-
-
86521-05-3
2-(trimethylsilylethynyl)pyridine

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 70℃; for 3h; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine at 20℃; | 97% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 50℃; Sonogashira Cross-Coupling; Inert atmosphere; | 97% |
-
-
626-05-1
2,6-Dibromopyridine

-
-
1066-54-2
trimethylsilylacetylene

-
-
75867-44-6
2,6-bis(2-(trimethylsilyl)ethynyl)pyridine

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine In toluene at 20℃; Sonogashira Cross-Coupling; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine In tetrahydrofuran at 50℃; for 6h; Inert atmosphere; | 98% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); N-ethyl-N,N-diisopropylamine In toluene for 90h; Sonogashira Cross-Coupling; Inert atmosphere; Darkness; | 98% |
-
-
5332-24-1
3-bromoquinoline

-
-
1066-54-2
trimethylsilylacetylene

-
-
86521-08-6
3-((trimethylsilyl)ethynyl)-quinoline

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine; triphenylphosphine In tetrahydrofuran at 50℃; for 24h; Inert atmosphere; | 100% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran at 20℃; for 24h; | 96% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In N,N-dimethyl-formamide at 20℃; for 18h; Sonogashira coupling; Inert atmosphere; | 87% |
-
-
286-20-4
cyclohexane-1,2-epoxide

-
-
1066-54-2
trimethylsilylacetylene

-
-
142636-23-5
(1R*,2S*)-2-(trimethylsilanylethynyl)cyclohexanol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.166667h; Inert atmosphere; Stage #2: With boron trifluoride diethyl etherate In tetrahydrofuran; hexane at -78℃; for 0.166667h; Inert atmosphere; Stage #3: cyclohexane-1,2-epoxide In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium; boron trifluoride diethyl etherate In tetrahydrofuran at -78℃; for 0.5h; Stage #2: cyclohexane-1,2-epoxide In tetrahydrofuran at -78℃; for 4h; | 95% |
| Stage #1: trimethylsilylacetylene With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium In tetrahydrofuran; hexane at 0℃; Stage #2: cyclohexane-1,2-epoxide In tetrahydrofuran; hexane at 0℃; for 12h; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane | 77% |
| With boron trifluoride | |
| Yield given. Multistep reaction; |
-
-
108-94-1
cyclohexanone

-
-
1066-54-2
trimethylsilylacetylene

-
-
17962-22-0
1-((trimethylsilyl)ethynyl)cyclohexanol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran; hexane at -78℃; Stage #2: cyclohexanone In tetrahydrofuran; hexane at -78℃; Further stages.; | 100% |
| With n-butyllithium In diethyl ether; hexane 1.) -78 deg C, 1 h, 2.) -78 deg C, 1 h; 0 deg C, 1 h; | 99% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.75h; Inert atmosphere; Stage #2: cyclohexanone In tetrahydrofuran; hexane at -78 - 20℃; for 1.5h; Inert atmosphere; | 95% |
-
-
99-90-1
para-bromoacetophenone

-
-
1066-54-2
trimethylsilylacetylene

-
-
75883-03-3
4'-(2-trimethylsilylethynyl)acetophenone

| Conditions | Yield |
|---|---|
| With copper diacetate; triethylamine; triphenylphosphine; palladium dichloride at 100℃; for 1.5h; | 100% |
| With copper diacetate; triethylamine; triphenylphosphine; palladium dichloride at 100℃; for 1.5h; | 100% |
| With copper(l) iodide; palladium diacetate; triethylamine; triphenylphosphine at 60℃; for 12h; | 98% |
-
-
112671-55-3
ethyl 2-bromo-3-pyridinecarbamate

-
-
1066-54-2
trimethylsilylacetylene

-
-
112671-57-5
(2-Trimethylsilanylethynyl-pyridin-3-yl)carbamic acid ethyl ester

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 100℃; for 4h; Sealed tube; | 100% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 100℃; for 4h; | 100% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 100 - 110℃; |
-
-
100-52-7
benzaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
89530-34-7
1-phenyl-3-trimethylsilylprop-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran at 0℃; for 1h; Stage #2: benzaldehyde In tetrahydrofuran at 0℃; for 1h; | 100% |
| With n-butyllithium In tetrahydrofuran at -78℃; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1.33333h; Inert atmosphere; Stage #2: benzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane | 100% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
159351-36-7
1-(4-methoxyphenyl)-3-(trimethylsilyl)prop-2-yn-1-ol

| Conditions | Yield |
|---|---|
| With methyllithium In tetrahydrofuran; diethyl ether 1 h, RT; -30 deg C to RT; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran at 0℃; for 1h; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran at 0℃; for 1h; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In diethyl ether at -78℃; Stage #2: 4-methoxy-benzaldehyde In diethyl ether at -78 - -10℃; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: hexanal In tetrahydrofuran; hexane at -78 - 20℃; for 3h; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: hexanal In tetrahydrofuran; hexane at 0℃; for 4h; | 96% |
| With n-butyllithium In hexane at 25℃; for 1.5h; | 80% |
-
-
1122-91-4
4-bromo-benzaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
77123-57-0
4-[(trimethylsilyl)ethynyl]bezaldehyde

| Conditions | Yield |
|---|---|
| With copper(l) iodide; diisopropylamine; polymer-supp. 11-(2-[Pd(OAc)2]-1-Mes-3-imidazolyl)undecanol In tetrahydrofuran at 20℃; for 2h; Sonogashira coupling; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 20℃; Inert atmosphere; | 100% |
| Stage #1: 4-bromo-benzaldehyde With 5% Pd/C; triethylamine; triphenylphosphine for 0.0833333h; Sonogashira Cross-Coupling; Sealed tube; Sonication; Stage #2: trimethylsilylacetylene for 3h; Sealed tube; | 100% |
-
-
135-02-4
ortho-anisaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
153889-90-8, 153889-99-7
1-(2-methoxyphenyl)-3-(trimethylsilyl)prop-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1.33333h; Inert atmosphere; Stage #2: ortho-anisaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In diethyl ether at -78℃; Stage #2: ortho-anisaldehyde In diethyl ether at -78 - -10℃; | 90% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In diethyl ether; hexane at -78℃; for 0.5h; Stage #2: ortho-anisaldehyde In diethyl ether; hexane at -78 - 20℃; Further stages.; | 90% |
-
-
6630-33-7
ortho-bromobenzaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
77123-58-1
2-[2-(trimethylsilyl)ethynyl]benzaldehyde

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | 100% |
| Stage #1: ortho-bromobenzaldehyde; trimethylsilylacetylene With bis-triphenylphosphine-palladium(II) chloride; triethylamine at 20℃; for 0.25h; Sonogashira coupling; Inert atmosphere; Stage #2: With copper(l) iodide at 50℃; for 2.5h; Sonogashira coupling; Inert atmosphere; | 99% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 50℃; Inert atmosphere; | 99% |
-
-
2043-61-0
cyclohexanecarbaldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
133216-88-3, 112497-25-3
(+/-)-1-cyclohexyl-3-(trimethylsilyl)-2-propynol

| Conditions | Yield |
|---|---|
| With n-butyllithium Inert atmosphere; | 100% |
| With ethylmagnesium bromide In tetrahydrofuran; diethyl ether at -15 - 20℃; for 3h; Inert atmosphere; | 100% |
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.333333h; Stage #2: cyclohexanecarbaldehyde In tetrahydrofuran; hexane at -78 - 24℃; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane | 92% |
-
-
108-86-1
bromobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
2170-06-1
1-Phenyl-2-(trimethylsilyl)acetylene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; diisopropylamine; polymer-supp. 11-(2-[Pd(OAc)2]-1-Mes-3-imidazolyl)undecanol In tetrahydrofuran at 20℃; for 2.5h; Sonogashira coupling; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine for 24h; Sonogashira coupling; Inert atmosphere; Reflux; | 99% |
| With potassium carbonate In water at 60℃; for 2h; | 95% |
-
-
401-78-5
3-bromo-1-trifluoromethylbenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
40230-93-1
3-<(trimethylsilyl)ethynyl>benzotrifluoride

| Conditions | Yield |
|---|---|
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In ethyl acetate at 50℃; for 18h; Sonogashira Coupling; | 100% |
| With copper(l) iodide; palladium diacetate; triethylamine; phenylphosphane at 80℃; for 16h; | 66.9% |

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: heptanal In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Inert atmosphere; | 100% |
| With n-butyllithium 1.) THF, -78 deg C, 1 h, 2.) THF, room temp., 1 h; Multistep reaction; | |
| With n-butyllithium 1) THF, -78 deg C, 1 h, 2) THF, r. t., 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In hexanes; diethyl ether at -78℃; for 1.16667h; Stage #2: Cyclopropyl methyl ketone In hexanes; diethyl ether at 20℃; for 48h; | 100% |
| With methyllithium 1.) ether, room temperature, 2 h; Multistep reaction; |
-
-
637-87-6
1-Chloro-4-iodobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
78704-49-1
1-(4-chlorophenyl)-2-trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran Sonogashira Cross-Coupling; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine for 2h; Ambient temperature; | 98% |
| With piperidine; copper(l) iodide; MCM-41-supported bidentate phosphine palladium(0) at 20℃; for 1.5h; Sonogashira coupling reaction; | 97% |
-
-
3132-99-8
m-bromobenzoic aldehyde

-
-
1066-54-2
trimethylsilylacetylene

-
-
77123-55-8
3-((trimethylsilyl)ethynyl)benzaldehyde

| Conditions | Yield |
|---|---|
| With copper(l) iodide; bis(benzonitrile)palladium(II) dichloride; tri-tert-butyl phosphine; diisopropylamine In 1,4-dioxane Inert atmosphere; | 100% |
| With copper(l) iodide; bis(benzonitrile)palladium(II) dichloride; tri-tert-butyl phosphine; diisopropylamine In 1,4-dioxane for 7h; Inert atmosphere; | 100% |
| With palladium diacetate; triethylamine; triphenylphosphine at 100℃; for 6h; Inert atmosphere; | 95% |
-
-
352-34-1
4-fluoro-1-iodobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
130995-12-9
1-(4-fluorophenyl)-2-trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 50℃; for 6h; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 50℃; for 5h; Sonogashira Cross-Coupling; Inert atmosphere; | 99% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In tetrahydrofuran at 20℃; for 18h; | 95% |
-
-
583-55-1
1-Bromo-2-iodobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
38274-16-7
2-bromo-1-(trimethylsilylethynyl)benzene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 100% |
| With copper(l) iodide; trans-bis(triphenylphosphine)palladium dichloride; triethylamine at 20℃; Inert atmosphere; | 100% |
-
-
580-13-2
2-bromonaphthalene

-
-
1066-54-2
trimethylsilylacetylene

-
-
40231-00-3
trimethyl(2-(naphthalen-2-yl)ethynyl)silane

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 60℃; for 72h; | 100% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 20 - 40℃; Inert atmosphere; | 99% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 40℃; for 5h; Sonogashira coupling; | 99% |
-
-
124-38-9
carbon dioxide

-
-
1066-54-2
trimethylsilylacetylene

-
-
5683-31-8
3-(trimethylsilyl)prop-2-ynoic acid

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at 78℃; for 0.5h; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran; hexane at 20℃; for 1h; | 100% |
| With hydrogenchloride; C54H44Cu2N2P4S2; caesium carbonate In N,N-dimethyl-formamide at 25℃; for 12h; Inert atmosphere; | 93% |
| With [CuI(1,1′-bis(di-tert-butylphosphino)ferrocene)]; caesium carbonate In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 24h; Inert atmosphere; | 90% |
-
-
625-99-0
3-iodochlorobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
227936-62-1
1-chloro-3-[2-(trimethylsilyl)ethynyl]benzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine; triphenylphosphine at 80℃; for 4h; Sonogashira coupling; Inert atmosphere; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine In toluene at 20 - 60℃; for 19h; Inert atmosphere; | 92% |
| With copper(l) iodide; palladium on activated charcoal; choline chloride; triethylamine; glycerol at 60℃; for 3h; Sonogashira Cross-Coupling; | 63% |
-
-
589-87-7
1,4-bromoiodobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
16116-78-2
(4-bromophenyl)(trimethylsilyl)acetylene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 100℃; for 0.5h; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran Sonogashira Cross-Coupling; Inert atmosphere; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine at 0℃; Schlenk technique; Inert atmosphere; | 99% |
-
-
624-38-4
para-diiodobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
17938-13-5
1,4-Bis(trimethylsilylethynyl)benzene

| Conditions | Yield |
|---|---|
| Sonogashira cross-coupling; | 100% |
| With copper(l) iodide; N-ethyl-N,N-diisopropylamine; bis-triphenylphosphine-palladium(II) chloride Castro-Stephens/Sonogashira coupling; Heating; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine In tetrahydrofuran at 50℃; for 21h; Sonogashira Cross-Coupling; Inert atmosphere; | 98% |
-
-
615-41-8
2-iodochlorobenzene

-
-
1066-54-2
trimethylsilylacetylene

-
-
105273-33-4
((2-chlorophenyl)-ethynyl)trimethylsilane

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine at 80℃; for 4h; Sonogashira coupling; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 60℃; for 22h; Sonogashira Cross-Coupling; Inert atmosphere; | 64% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In triethylamine Yield given; |
Trimethylsilylacetylene Specification
The Trimethylsilylacetylene, with the CAS registry number 1066-54-2 and EINECS registry number 213-919-9, has the systematic name of ethynyl(trimethyl)silane. It is a kind of clear colorless liquid, and belongs to the following product categories: Acetylenes; Diyne Compounds (LB Films); Ethynylsilanes; Functional Materials; Functionalized Acetylenes; LB Films; Si (Classes of Silicon Compounds); Silicon Compounds (for Synthesis); Synthetic Organic Chemistry. And the molecular formula of this chemical is C8H9FN2O. What's more, it is usually used as silicon agent and anisomerous common intermediates. And it should be stored at 2-8°C.
The physical properties of Trimethylsilylacetylene are as followings: (1)ACD/LogP: 2.16; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.16; (4)ACD/LogD (pH 7.4): 2.16; (5)ACD/BCF (pH 5.5): 25.64; (6)ACD/BCF (pH 7.4): 25.64; (7)ACD/KOC (pH 5.5): 354.88; (8)ACD/KOC (pH 7.4): 354.88; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 0 Å2; (13)Index of Refraction: 1.401; (14)Molar Refractivity: 31.75 cm3; (15)Molar Volume: 130.5 cm3; (16)Polarizability: 12.58×10-24cm3; (17)Surface Tension: 18 dyne/cm; (18)Density: 0.752 g/cm3; (19)Enthalpy of Vaporization: 28.39 kJ/mol; (20)Boiling Point: 53 °C at 760 mmHg; (21)Vapour Pressure: 273 mmHg at 25°C.
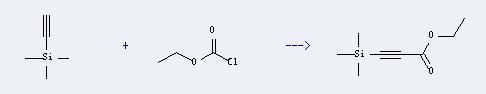
Uses of Trimethylsilylacetylene: It can react with carbonochloridic acid ethyl ester to produce trimethylsilanyl-propiolic acid ethyl ester. This reaction will need reagent n-BuLi. And the yield is about 95%.
You should be cautious while dealing with this chemical. It is a kind of flammble chemical which irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Keep away from sources of ignition-No smoking; Wear suitable protective clothing, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: C#C[Si](C)(C)C
(2)InChI: InChI=1/C5H10Si/c1-5-6(2,3)4/h1H,2-4H3
(3)InChIKey: CWMFRHBXRUITQE-UHFFFAOYAI
Related Products
- Trimethylsilylacetylene
- 106658-09-7
- 106658-10-0
- 106670-34-2
- 106681-15-6
- 106-68-3
- 106685-40-9
- 106685-41-0
- 106689-24-1
- 106-69-4
- 106697-24-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View