-
Name
trans-Anethole
- EINECS 224-052-0
- CAS No. 4180-23-8
- Article Data158
- CAS DataBase
- Density 0.959 g/cm3
- Solubility practically insoluble in water
- Melting Point 20-21 °C(lit.)
- Formula C10H12O
- Boiling Point 237.5 °C at 760 mmHg
- Molecular Weight 148.205
- Flash Point 88.4 °C
- Transport Information
- Appearance clear colorless to pale yellow liquid
- Safety 36/37
- Risk Codes 43
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Anisole,p-propenyl-, (E)- (8CI);Benzene, 1-methoxy-4-(1-propenyl)-, (E)-;Benzene,1-methoxy-4-(1E)-1-propenyl- (9CI);(E)-1-(4-Methoxyphenyl)propene;(E)-1-p-Methoxyphenylpropene;(E)-Anethol;(E)-Anethole;1-Methoxy-4-[(1E)-1-propenyl]benzene;NSC 209529;trans-1-(4-Methoxyphenyl)-1-propene;trans-1-(p-Methoxyphenyl)-1-propene;trans-1-(p-Methoxyphenyl)propene;trans-1-p-Anisylpropene;trans-4-(1-Propenyl)anisole;trans-p-Anethole;trans-p-Methoxy-b-methylstyrene;Benzene,1-methoxy-4-(1E)-1-propen-1-yl-;
- PSA 9.23000
- LogP 2.72830
Synthetic route

| Conditions | Yield |
|---|---|
| With Polystyrene-supported 4-tert-butyl-2-(diisopropylphosphino)-1H-imidazole and ruthenium complex In [(2)H6]acetone at 25℃; for 0.166667h; Reagent/catalyst; Inert atmosphere; Glovebox; stereoselective reaction; | 99% |
| With PdClMe(2,9-dimethyl-1,10-phenanthroline); sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In chloroform-d1 at 0℃; for 16h; Sealed tube; | 99% |
| With nickel(II) iodide; 6,6'-dimethyl-2,2'-bipyridine; phosphonic acid diethyl ester; zinc In N,N-dimethyl acetamide at 35℃; for 24h; | 97% |
-
-
25679-28-1
cis-Anethole

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: cis-Anethole With cobalt(II) chloride; 2,2'-bis(diphenylphosphino)diphenylamine In toluene at 20℃; for 0.0833333h; Schlenk technique; Inert atmosphere; Stage #2: With sodium triethylborohydride In toluene at 20℃; for 1h; Catalytic behavior; Reagent/catalyst; Schlenk technique; Inert atmosphere; diastereoselective reaction; | 99% |
| With sodium hydrogen sulfate at 100 - 155℃; for 1h; Temperature; | 95.4% |
| cadmium(II) sulphide In dichloromethane Product distribution; Irradiation; |

| Conditions | Yield |
|---|---|
| With potassium carbonate; dichloro[1,1'-bis(di-t-butylphosphino)ferrocene]palladium(II) In water; isopropyl alcohol at 120℃; for 0.5h; Inert atmosphere; Microwave irradiation; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: (E)-phenyl(prop-1-en-1-yl)silane With tetrabutyl ammonium fluoride In tetrahydrofuran for 0.05h; Glovebox; Inert atmosphere; Schlenk technique; Stage #2: para-iodoanisole With tris-(dibenzylideneacetone)dipalladium(0) In tetrahydrofuran for 10h; Hiyama Coupling; Glovebox; Inert atmosphere; Schlenk technique; | 97% |
-
-
1530-32-1
ethyltriphenylphosphonium bromide

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: ethyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at -78 - 0℃; Inert atmosphere; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran at -78 - 25℃; for 3.83333h; Wittig reaction; Inert atmosphere; Stage #3: With dichloro bis(acetonitrile) palladium(II) In chloroform at 25℃; for 72h; | 94% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 0.166667h; | A n/a B 91% |


-
A
-
25679-28-1
cis-Anethole

-
B
-
99685-96-8, 161105-99-3, 161106-00-9, 111138-12-6, 133318-63-5, 134053-11-5, 134931-35-4, 134931-36-5, 139703-76-7, 145633-27-8, 175414-73-0, 175414-74-1, 175414-75-2, 175519-12-7, 175519-13-8, 175519-14-9, 175519-15-0, 136376-46-0, 144906-37-6, 144906-38-7, 151767-00-9, 152882-97-8, 152882-98-9, 152882-99-0, 153062-34-1, 154171-74-1, 154171-75-2, 154333-99-0, 154334-00-6, 154397-63-4, 154460-59-0, 199456-56-9, 108739-25-9, 120329-57-9, 120329-58-0
fullerene-C60

-
C
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| In toluene Mechanism; Product distribution; Ambient temperature; Irradiation; | A 10% B n/a C 90% |


| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II) In dichloromethane at 20℃; for 14h; | 90% |

| Conditions | Yield |
|---|---|
| With 1-phenyl-2-(2-pyridyl)-1,2-disodiumethane In tetrahydrofuran at 0℃; for 10h; | 90% |
-
-
676-58-4
methylmagnesium chloride

-
-
15197-86-1
1-(2-fluorovinyl)-4-methoxybenzene

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With 1,2-bis(diphenylphosphino)ethane nickel(II) chloride In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| With 1-hexyl-3-methyl-1-imidazolium bromide at 140℃; for 0.116667h; Reactivity; Reagent/catalyst; Time; Microwave irradiation; Combinatorial reaction / High throughput screening (HTS); chemoselective reaction; | 87% |
| With silica gel In 1,4-dioxane for 0.05h; microwave irradiation; | 67% |
| With trichlorophosphate In pyridine for 2h; Heating; |
-
-
53484-50-7
(E)-p-methoxy-cinnamyl alcohol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With bis(tricyclohexylphosphine)nickel(II) dichloride; n-hexylmagnesium chloride; 1,2-bis-(dicyclohexylphosphino)ethane In toluene at 100℃; for 12h; Inert atmosphere; Schlenk technique; | 86% |
-
-
16031-55-3
1-(4-methoxyphenyl)prop-1-yl acetate

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With 1-hexyl-3-methyl-1-imidazolium bromide at 140℃; for 0.166667h; Microwave irradiation; chemoselective reaction; | 82% |
-
-
696-62-8
para-iodoanisole

-
-
6336-44-3, 7547-96-8, 88982-39-2, 7547-97-9
(E)-propenylboronic acid

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With orotic acid; caesium carbonate In acetone at 100℃; for 24h; Suzuki coupling; Inert atmosphere; | 82% |
-
-
88533-70-4, 88533-77-1, 103785-85-9, 103785-86-0
(1RS,2RS)-1-(4'-methoxyphenyl)-2-(diphenylphosphinoyl)propan-1-ol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide | 81.3% |
-
-
88533-70-4, 88533-77-1, 103785-85-9, 103785-86-0
2-(Diphenyl-phosphinoyl)-1-(4-methoxy-phenyl)-propan-1-ol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide | 81% |
-
-
1106676-03-2
C17H18O3

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With 1-hexyl-3-methyl-1-imidazolium bromide at 140℃; for 0.166667h; Microwave irradiation; chemoselective reaction; | 80% |
-
-
4792-39-6
1,1-bis(4-methoxyphenyl)propane

-
A
-
100-66-3
methoxybenzene

-
B
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With silica gel In water at 400℃; Inert atmosphere; | A 77.3% B 45.1% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
123-38-6
propionaldehyde

-
-
109-77-3
malononitrile

-
A
-
1338442-76-4
3-amino-4'-methoxy-6-methyl-[1,1'-biphenyl]-2,4-dicarbonitrile

-
B
-
55525-92-3
2-amino-4-ethyl-5-methylbenzene-1,3-dicarbonitrile

-
C
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With morpholine In N,N-dimethyl-formamide at 80℃; | A 77% B 8% C 10% |

-
-
88533-70-4, 88533-77-1, 103785-85-9, 103785-86-0
(1R*,2S*)-2-Diphenylphosphinoyl-1-(4-methoxyphenyl)propan-1-ol

-
A
-
25679-28-1
cis-Anethole

-
B
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide | A 75% B 6% |
-
-
51410-44-7
1-(4-methoxylphenyl)-2-propen-1-ol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With bis(tricyclohexylphosphine)nickel(II) dichloride; n-hexylmagnesium chloride; 1,2-bis-(dicyclohexylphosphino)ethane In toluene at 100℃; for 12h; Inert atmosphere; Schlenk technique; | 75% |
-
-
599-70-2
ethyl phenyl sulfone

-
-
105-13-5
4-Methoxybenzyl alcohol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With C24H20ClN2OPRu; potassium tert-butylate In 1,4-dioxane at 125℃; for 5h; Inert atmosphere; Schlenk technique; Glovebox; | 71% |

| Conditions | Yield |
|---|---|
| With chloro(1,5-cyclooctadiene)rhodium(I) dimer; potassium carbonate; bis(pinacol)diborane; bis[2-(diphenylphosphino)phenyl] ether In hexane at 70℃; for 10h; Glovebox; Sealed tube; Inert atmosphere; stereoselective reaction; | 68% |
-
-
140-67-0
Estragole

-
-
201230-82-2
carbon monoxide

-
A
-
104-45-0
4-n-propylanisole

-
B
-
5462-06-6
3-(p-methoxyphenyl)-2-methylpropionaldehyde

-
C
-
56047-51-9
4-(4-methoxyphenyl)butyraldehyde

-
D
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With hydrogen; N-dodecyl-N-(2-hydroxyethyl)-N,N-dimethylammonium bromide; {Rh(cod)[μ-S(CH2)3Si(OMe)3]}2; triphenylphosphine In water; butan-1-ol at 80℃; under 10350.8 Torr; for 6h; microemulsion/sol-gel; Further byproducts given; | A 1.4% B 27.4% C 63.8% D 6.7% |

-
-
623-12-1
4-chloromethoxybenzene

-
-
105494-65-3, 66680-85-1, 66680-84-0
(E)-propenyl-tri-n-butylstannane

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With palladium diacetate; orotic acid; cesium fluoride In 1,4-dioxane at 100℃; for 18h; Stille Cross Coupling; Sealed tube; Inert atmosphere; Schlenk technique; | 62% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
24850-33-7
allyltributylstanane

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; triphenylphosphine; lithium chloride In diethylene glycol dimethyl ether at 130℃; Stille Cross Coupling; Inert atmosphere; regioselective reaction; | 62% |
-
-
14846-66-3
trans-1-methoxy-4-(2-methylsulfanylvinyl)benzene

-
-
75-16-1
methylmagnesium bromide

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| bis(triphenylphosphine)nickel(II) chloride In diethyl ether; benzene Heating; | 60% |
-
-
513-35-9
2-methyl-but-2-ene

-
-
123-11-5
4-methoxy-benzaldehyde

-
A
-
67-64-1
acetone

-
B
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With iron(III) chloride; silver tetrafluoroborate In dichloromethane at 25℃; for 3h; Inert atmosphere; | A n/a B 60% |

-
-
4333-56-6
cyclopropyl bromide

-
-
113379-13-8
3-(4-methoxyphenyl)-1-iodopropane

-
B
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: cyclopropyl bromide With iodine; magnesium In 2-methyltetrahydrofuran for 1.5h; Inert atmosphere; Reflux; Stage #2: 3-(4-methoxyphenyl)-1-iodopropane With N,N,N,N,-tetramethylethylenediamine; cobalt(II) chloride In tetrahydrofuran; 2-methyltetrahydrofuran at 0℃; for 1h; Catalytic behavior; Temperature; Inert atmosphere; chemoselective reaction; | A 56% B 17% |

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
82947-15-7, 82947-17-9
2-Bromo-1-(4-methoxy-phenyl)-propan-1-ol

| Conditions | Yield |
|---|---|
| With hypobromous acid In 1,4-dioxane at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; benzamidin for 3h; | 100% |
-
-
59089-57-5
pyridin-2-yl sulfenyl chloride

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: pyridin-2-yl sulfenyl chloride With sulfuryl dichloride In dichloromethane at 20℃; for 0.166667h; Stage #2: E-1-(4'-methoxyphenyl)prop-1-ene In dichloromethane at 20℃; for 20h; regioselective reaction; | 100% |
-
-
59957-75-4
2,2'-dipyridyldiselenide

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: 2,2'-dipyridyldiselenide With sulfuryl dichloride In chloroform at 20℃; for 0.333333h; Stage #2: E-1-(4'-methoxyphenyl)prop-1-ene In chloroform at 20℃; for 5h; Reflux; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With magnesium; nickel dichloride In water at 20℃; for 12h; Sealed tube; Photolysis; Inert atmosphere; | 99% |
| With oxygen; hydrazine hydrate In propan-1-ol at 120℃; under 15001.5 Torr; for 0.5h; | 91% |
| With oxygen; hydrazine hydrate In propan-1-ol at 120℃; under 15001.5 Torr; for 0.166667h; Flow reactor; | 91% |
-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
4705-34-4, 15638-14-9, 34480-01-8, 112246-67-0, 2510-75-0
1,2-bis(4-methoxyphenyl)ethene

| Conditions | Yield |
|---|---|
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride; acrylic acid methyl ester In dichloromethane for 2h; Reflux; Inert atmosphere; | 99% |
| With C33H43Cl2N3O3RuS In toluene at 110℃; for 2h; Inert atmosphere; | 90% |
| With silica-supported Hoveyda-Grubbs II catalyst In hexane at 70℃; for 14h; Inert atmosphere; Glovebox; | 74% |
| (1,3-dimesitylimidazolin-2-ylidene)(C2H4)RuCl2; (p-cymene)RuCl2 In toluene at 85℃; for 2h; | 90 % Turnov. |
-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Ru(2,2':6',2''-terpyridine)(2,6-pyridinedicarboxylate) In tert-Amyl alcohol at 20℃; for 12h; Conversion of starting material; | 99% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
54815-45-1
ethyl benzenesulfenate

-
-
540-72-7
sodium thiocyanide

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: chloro-trimethyl-silane; sodium thiocyanide In chloroform at 20℃; for 1h; Stage #2: ethyl benzenesulfenate; E-1-(4'-methoxyphenyl)prop-1-ene In chloroform | 99% |

-
-
292638-84-7
styrene

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With 9-(2-methylphenyl)-1,3,6,8-tetramethoxythioxanthylium trifluoromethanesulfonate In nitromethane at 20℃; for 2h; Catalytic behavior; Solvent; Reagent/catalyst; Irradiation; regioselective reaction; | 99% |
| With iron perchlorate hexahydrate In ethyl acetate at 40℃; for 24h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; diastereoselective reaction; | 88% |
| With iron perchlorate hexahydrate In ethyl acetate at 40℃; under 760.051 Torr; for 24h; Solvent; Reagent/catalyst; | 88% |
| With Fe(3,4,7,8-tetramethyl-1,10-phenanthroline)3(PF6)3 In 2,2,2-trifluoroethanol; 1,2-dichloro-ethane at 20℃; for 3h; Catalytic behavior; Reagent/catalyst; Solvent; | 86% |
-
-
132524-91-5
t-butyl 2-diazobut-3-enoate

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| With tris(2,2′-bipyrazine-N1,N1′)ruthenium(II) hexafluorophosphate In nitromethane; dichloromethane at 35℃; for 1h; Reagent/catalyst; Irradiation; diastereoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With cross-linked copolymer based on polymethyl methacrylate and 4,7-diphenylbenzothiadiazole; air In nitromethane at 20℃; for 8h; Diels-Alder Cycloaddition; UV-irradiation; | 99% |
| With 9-fluorenone In nitromethane at 20℃; for 6h; Catalytic behavior; Mechanism; Reagent/catalyst; Concentration; Solvent; Diels-Alder Cycloaddition; Irradiation; | 74% |

| Conditions | Yield |
|---|---|
| With cross-linked copolymer based on polymethyl methacrylate and 4,7-diphenylbenzothiadiazole; air In nitromethane at 20℃; for 4h; Solvent; Reagent/catalyst; Diels-Alder Cycloaddition; UV-irradiation; | 99% |
| With 9-fluorenone In nitromethane at 20℃; for 23h; Diels-Alder Cycloaddition; Irradiation; | 88% |

| Conditions | Yield |
|---|---|
| With cross-linked copolymer based on polymethyl methacrylate and 4,7-diphenylbenzothiadiazole; air In nitromethane at 20℃; for 48h; Diels-Alder Cycloaddition; UV-irradiation; | 99% |
-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
112150-19-3
(±)-(1S,2S)-4'-methoxy-2,4,5-trimethyl-1,2,3,6-tetrahydro-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With tris(2,2’--bipyrazyl)ruthenium(II) bis(tetrakis(3,5-bis(trifluoromethyl)phenyl)borate) In dichloromethane at 20℃; for 1h; Diels-Alder Cycloaddition; Irradiation; regioselective reaction; | 98% |
| With iron perchlorate hexahydrate In acetonitrile at 20℃; for 24h; Diels-Alder Cycloaddition; regioselective reaction; | 98% |
| With iron(III) chloride In acetonitrile at 20℃; for 1h; Time; Reagent/catalyst; Solvent; | 98% |
-
-
84-85-5
4-methoxynaphth-1-ol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
139016-16-3
(+/-)-trans-2-(4-methoxyphenyl)-2,3-dihydro-5-methoxy-3-methylnaphtho<1,2-b>furan

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate In nitromethane for 12h; Irradiation; Inert atmosphere; | 98% |
| With tetrabutylammonium tetrafluoroborate In acetonitrile Electrolysis; | 96% |
| With tetrabutylammonium tetrafluoroborate In acetonitrile Electrolysis; | 96% |
| With ammonium peroxydisulfate; tris(2,2′-bipyrazine-N1,N1′)ruthenium(II) hexafluorophosphate In acetonitrile for 12h; Irradiation; Schlenk technique; | 92% |
| With lithium perchlorate; acetic acid In acetonitrile for 3h; Ambient temperature; electrolysis, platinum electrodes; | 64% |
-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
78-79-5
isoprene

-
-
112150-17-1
(1SR,2SR)-4'-methoxy-2,4-dimethyl-1,2,3,6-tetrahydro-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With tris(2,2’--bipyrazyl)ruthenium(II) bis(tetrakis(3,5-bis(trifluoromethyl)phenyl)borate) In dichloromethane at 20℃; for 1h; Diels-Alder Cycloaddition; Irradiation; regioselective reaction; | 98% |
| With lithium perchlorate In nitromethane at 20℃; Concentration; Diels-Alder Cycloaddition; Electrolysis; | 98% |
| With iron perchlorate hexahydrate In acetonitrile at 20℃; for 24h; Reagent/catalyst; Diels-Alder Cycloaddition; regioselective reaction; | 98% |
-
-
60556-87-8
2-carbonyl-3-butenenitrile

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 168h; hetero-Diels-Alder cycloaddition; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: benzaldehyde; 4-nitro-aniline In acetonitrile at 20℃; for 1h; Povarov reaction; Stage #2: E-1-(4'-methoxyphenyl)prop-1-ene With copper(II) bis(trifluoromethanesulfonate) In acetonitrile at 20℃; for 24h; Povarov reaction; diastereoselective reaction; | 98% |
| With boron trifluoride diethyl etherate In acetonitrile at 70℃; for 10h; imino Diels-Alder reaction; | 95% |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
51901-85-0
bromocatecholborane

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
1217435-96-5
(Z)-2-(1-(4-methoxyphenyl)prop-1-en-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| Stage #1: bromocatecholborane With N-Methyldicyclohexylamine; [(bis(3,5-di-tert-butylphenyl)(tert-butyl)phosphine)PdI2]2 In toluene at 70℃; for 0.0833333h; Heck Reaction; Schlenk technique; Inert atmosphere; Sealed tube; Stage #2: E-1-(4'-methoxyphenyl)prop-1-ene In toluene at 70℃; for 4h; Heck Reaction; Schlenk technique; Sealed tube; Inert atmosphere; Stage #3: 2,3-dimethyl-2,3-butane diol In toluene at 20℃; for 0.5h; stereoselective reaction; | 98% |


| Conditions | Yield |
|---|---|
| With iron(III) chloride In dichloromethane at 20℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With 9-(2-methylphenyl)-1,3,6,8-tetramethoxythioxanthylium trifluoromethanesulfonate In nitromethane at 20℃; for 2h; Irradiation; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With tris(trimethylsilyl)amine; tetramethylammonium fluoride at 60℃; for 5h; Inert atmosphere; Sealed tube; Glovebox; | 98% |
-
-
150-76-5
4-methoxy-phenol

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
139016-13-0
(+/-)-trans-2-(4-methoxyphenyl)-2,3-dihydro-5-methoxy-3-methylbenzofuran

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate In nitromethane for 22h; Catalytic behavior; Solvent; Reagent/catalyst; Irradiation; Inert atmosphere; | 97% |
| With ammonium peroxydisulfate; tris(2,2′-bipyrazine-N1,N1′)ruthenium(II) hexafluorophosphate In acetonitrile for 27h; Irradiation; Schlenk technique; | 88% |
| With lithium perchlorate; acetic acid In acetonitrile for 2h; Ambient temperature; electrolysis, platinum electrodes; | 80% |
-
-
50-00-0
formaldehyd

-
-
4180-23-8
E-1-(4'-methoxyphenyl)prop-1-ene

-
-
5689-72-5
4-p-methoxyphenyl-5-methyl-1,3-dioxane

| Conditions | Yield |
|---|---|
| resine C35O; hydrogen cation In benzene at 60℃; for 12h; | 97% |

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; tris(2,2′-bipyrazine-N1,N1′)ruthenium(II) hexafluorophosphate In acetonitrile for 30h; Irradiation; Schlenk technique; | 97% |

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; tris(2,2′-bipyrazine-N1,N1′)ruthenium(II) hexafluorophosphate In acetonitrile for 30h; Irradiation; Schlenk technique; | 97% |

| Conditions | Yield |
|---|---|
| With iron perchlorate hexahydrate In acetonitrile at 20℃; for 24h; Diels-Alder Cycloaddition; regioselective reaction; | 97% |
| With C26H18N10Ru(2+)*2C32H12BF24(1-) In dichloromethane for 1.5h; Diels-Alder Cycloaddition; Irradiation; | 88% |
| With 9-(2-trifluoromethylphenyl)-1,3,6,8-tetramethoxythioxanthylium trifluoromethanesulfonate In nitromethane at 20℃; for 1h; Diels-Alder Cycloaddition; UV-irradiation; | 84% |
| With iron(III) chloride In dichloromethane; acetonitrile at 0℃; for 3h; | 76% |
trans-Anethole Consensus Reports
trans-Anethole Specification
1. Introduction of Trans-Anethole
Trans-Anethole, with the CAS registry number 4180-23-8, is also known as Anisole, p-propenyl-, (E)-. Its EINECS registry number is 224-052-0. This chemical's molecular formula is C10H12O and molecular weight is 148.20168. Its IUPAC name is called 1-methoxy-4-[(E)-prop-1-enyl]benzene. This chemical's classification codes are Drug / Therapeutic Agent; Mutation Data; Pharmaceutic aid [flavor]; Reproductive Effect; Tumor Data. Meanwhile, the product should be sealed and stored in cool and dry place.
2. Properties of Trans-Anethole
Physical properties of trans-Anethole: (1)ACD/LogP: 3.17; (2)ACD/LogD (pH 5.5): 3.17; (3)ACD/LogD (pH 7.4): 3.17; (4)ACD/BCF (pH 5.5): 150.59; (5)ACD/BCF (pH 7.4): 150.59; (6)ACD/KOC (pH 5.5): 1260.3; (7)ACD/KOC (pH 7.4): 1260.3; (8)#H bond acceptors: 1; (9)#Freely Rotating Bonds: 2; (10)Index of Refraction: 1.545; (11)Molar Refractivity: 48.82 cm3; (12)Molar Volume: 154.4 cm3; (13)Surface Tension: 31.8 dyne/cm; (14)Density: 0.959 g/cm3; (15)Flash Point: 88.4 °C; (16)Enthalpy of Vaporization: 45.51 kJ/mol; (17)Boiling Point: 237.5 °C at 760 mmHg; (18)Vapour Pressure: 0.0687 mmHg at 25°C.
3. Structure Descriptors of Trans-Anethole
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CC=CC1=CC=C(C=C1)OC
(2)Isomeric SMILES: C/C=C/C1=CC=C(C=C1)OC
(3)InChI: InChI=1S/C10H12O/c1-3-4-9-5-7-10(11-2)8-6-9/h3-8H,1-2H3/b4-3+
(4)InChIKey: RUVINXPYWBROJD-ONEGZZNKSA-N
4. Toxicity of Trans-Anethole
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 2167mg/kg (2167mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Food and Cosmetics Toxicology. Vol. 2, Pg. 327, 1964. |
| mouse | LD50 | intraperitoneal | 650mg/kg (650mg/kg) | Therapie. Vol. 22, Pg. 309, 1967. | |
| mouse | LD50 | oral | 3050mg/kg (3050mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: COMA | Food and Cosmetics Toxicology. Vol. 2, Pg. 327, 1964. |
| rat | LD50 | intraperitoneal | 900mg/kg (900mg/kg) | Therapie. Vol. 22, Pg. 309, 1967. | |
| rat | LD50 | oral | 2090mg/kg (2090mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: COMA | Food and Cosmetics Toxicology. Vol. 2, Pg. 327, 1964. |
5. Preparation of Trans-Anethole
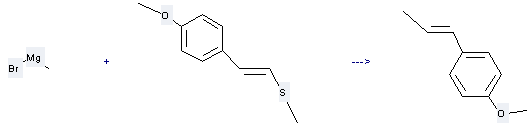
Trans-Anethole can be prepared by trans-p-Methoxystyrylmethylsulfid and methylmagnesium bromide. This reaction will need catalyst <(C6H5)3P>2NiCl2 and solvents benzene, diethyl ether. The yield is about 60%.
6. Use of Trans-Anethole
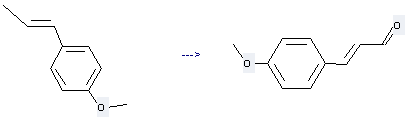
Trans-Anethole can be used to produce 3t-(4-methoxy-phenyl)-propenal at temperature of 50 - 60 °C. This reaction will need reagent SeO2.
7. Other details of Trans-Anethole
When you are using Trans-Anethole, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. In addition, it may cause sensitisation by skin contact. Whenever you will contact it, please wear suitable protective clothing and gloves.
Related Products
- trans-Anethole
- 4180-26-1
- 41804-89-1
- 4180-62-5
- 41806-40-0
- 41808-35-9
- 4181-05-9
- 4181-20-8
- 41814-78-2
- 4181-95-7
- 41820-22-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View