Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:563-78-0
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis
Cas:563-78-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:563-78-0
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:563-78-0
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:563-78-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Items Specifications Appearance Colorless to light yellow transparent liquid Color ≤50Hazen Purity
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:563-78-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Clear colourless to light yellow liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Appl
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:563-78-0
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:563-78-0
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
2,3-Dimethyl-1-butene Basic information Product Name: 2,3-Dimethyl-1-butene
Cas:563-78-0
Min.Order:100 Gram
Negotiable
Type:Trading Company
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
Cas:563-78-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen BaiFuchem Co.,Ltd
BaiFuChem is a Professional chemical raw material supplier in China, our main products include Biochemical , Pharma Intermediate and Organic chemical etc. BaiFuChem have wealth of products,experience , expertise and state-of-the-art equ
Hangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
XinChem Corporation
We have our own research and development center, provide reliable quality products for you. Port:shanghai
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Shanghai AngewChem Co., Ltd.
Shanghai AngewChemCo., Ltd. is an innovative enterprise on fine chemicals and pharmaceuticals. Based on Shanghai R&D center and Hunan chemical manufacturing plant, we offer chemical research, process development, and large-scale production. Complete
Finetech Industry Limited
FINETECH INDUSTRY LIMITED is a LONDON based CRO company providing drug discovery & development services to worldwide clients. FINETECH INDUSTRY LIMITED supplies the 2,3-Dimethyl-1-butene, CAS:563-78-0 with the most competitive price and the best qual
Skyrun Industrial Co.,Ltd
SkyRun Industrial CO.,LTD is a leading chemical producer in China. We supply Intermediate, Agro-Chemical and Petrol Chemicals for different usage.SkyRun Industrial CO.,LTD is a leading chemical producer in China. We supply Intermediate, Agro-Chemic
Nanjing Raymon Biotech Co., Ltd.
1-Butene, 2,3-dimethyl- Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Hangzhou Yierdechem Co. Ltd
With about ten years experiences in the field of pharmaceutical chemicals, Yierdechem has established solid business cooperation relationships with many large companys worldwide.As a leading supplier of API and pharmaceutical intermediates, holds its
Hebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Changzhou Foreign Trade Corp
CFTC?PharmaChem?is?a?service?based?company?in?China,?a?pharmachem?division?of?Changzhou?Foreign?Trade?Corp.,supplying?raw?materials,?intermediates,?APIs?and?fine?chemicals?for?the?pharmaceutical?and?specialty?chemical?industries?worldwide. Applicatio
Synthetic route
-
-
187737-37-7
propene

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With diethyl aluminiumcholoride; trioctyl phosphite In toluene at -10℃; under 1500.15 Torr; for 5h; Reagent/catalyst; Temperature; Inert atmosphere; Autoclave; | 90% |
| With In toluene at 70℃; for 50h; | 9% |
| With In benzene-d6 at 49℃; Rate constant; | |
| With (C6H5O)2P-N(C4H9)2; diethylaluminium chloride; Ni(β-C10H7CO2)2 In toluene at 18 - 23℃; under 750.06 Torr; Product distribution; influence of organophosphorus ligand L on the activity and selectivity of catalytic systems; | |
| With C30H38FeN3 In benzene-d6 at 23℃; for 48h; Diels-Alder Cycloaddition; |
-
-
187737-37-7
propene

-
A
-
763-29-1
2-Methyl-1-pentene

-
B
-
691-38-3
(Z)-4-methyl-2-pentene

-
C
-
691-37-2
4-Methyl-1-pentene

-
D
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With ethylaluminum dichloride; π-C3H5NiP(C6H11-cyclo)3Br In chlorobenzene at -75 - -45℃; for 0.5h; Product distribution; | A 4% B 1% C 18% D 76% |
| With VCl3((C5H3N)(C(CH3)N(C6H2)Br2Me)2) In toluene at 20℃; for 1h; | |
| VCl3((C5H3N)(C(CH3)N(C6H2)Br2Me)2) In toluene at 20℃; for 1h; | A 6 %Chromat. B 45 %Chromat. C 24 %Chromat. D 25 %Chromat. |

-
-
187737-37-7
propene

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
625-27-4
2-methyl-2-pentene

-
C
-
763-29-1
2-Methyl-1-pentene

-
D
-
4461-48-7
4-methyl-2-pentene

-
E
-
691-37-2
4-Methyl-1-pentene

-
F
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| diethylaluminium chloride In benzene at 50℃; under 6840 Torr; for 2h; Product distribution; 0 - 60 deg C, 1 - 32 h, influence of the catalysts on the yields; | A 5.4% B 3.2% C 18.4% D 58.2% E 10.2% F 2.1% G n/a |

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
79-29-8
2,3-dimethylbutane

-
C
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.25h; Product distribution; Ambient temperature; various time; | A 39.3% B 0.05% C 52.2% |
| With hydrogen; 1,5-hexadienerhodium(I)-chloride dimer In various solvent(s) for 2h; Ambient temperature; pH=7.6; | A 27% B 25% C 22% |
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; hydrogen; disodium salt of the bis(m-sulfonatophenyl)-n-butylphosphane at 60℃; under 22502.3 Torr; for 6h; Ionic liquid; chemoselective reaction; |

-
-
115982-92-8
tantalum(η5-Cp)(2,3-dimethylbutadiene){C5H6O(isopropyl)2}

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
38443-89-9
2-methyl-3-isopropyl-6-hepten-3-ol

-
C
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| Hydrolysis of starting compd.; Gas chromy.; | A 35% B 48% C 17% |


-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
38023-36-8
1,4-diphenyl-2,3-dimethylbutadiene-1,3

-
B
-
21564-72-7
1,4-Dihydro-2,3-dimethylnaphthalene

-
G
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| In benzene at 120℃; for 16h; | A n/a B n/a C 30% D n/a E 20% F 45% G n/a |

-
-
64-17-5
ethanol

-
-
125881-10-9
2,3,4,5,6-Pentamethyl-benzenesulfonic acid 1,2,2-trimethyl-3-trimethylsilanyl-propyl ester

-
A
-
558-37-2
tert-butylethylene

-
B
-
31397-75-8
(2,3-Dimethyl-but-3-enyl)-trimethyl-silane

-
E
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With water at 25℃; Rate constant; Product distribution; Mechanism; other alcohol, var. EtOH conc.; deuterium kinetic isotopic effect; | A 9% B 32% C 6% D 39% E 14% |

-
-
187737-37-7
propene

-
A
-
625-27-4
2-methyl-2-pentene

-
B
-
13269-52-8
trans-3-Hexene

-
C
-
4050-45-7
trans-2-hexene

-
D
-
691-38-3
(Z)-4-methyl-2-pentene

-
E
-
674-76-0
(E)-4-methylpent-2-ene

-
F
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With tetrabutylammonium perchlorate; bis(triphenylphosphine)nickel(II) chloride In various solvent(s) at 50℃; under 3750.3 Torr; for 24h; Product distribution; electrochemical reduction process, effect of catalyst, anode, supporting electrolyte, ligand, temperature and pressure; | A 23% B 13.5% C 4.2% D 6.1% E 32.7% F 14.8% |


| Conditions | Yield |
|---|---|
| With NaX zeolite at 300℃; | 27% |
| silver (I) ion In acetonitrile Irradiation; Yield given; | |
| With hydrogenchloride; water In decalin at 165℃; for 20h; Product distribution; in sealed tube; also with DCl/D2O, deuteration investigated; | 20.1 % Chromat. |
-
-
35913-82-7
cyclopentadienylirondicarbonyl hydride

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
38117-54-3
cyclopentadienyl iron(II) dicarbonyl dimer

-
D
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| In pentane under N2, stirring at room temp, the react. was complete within about 1 h; evapn. (vac.), chromy. on alumina (eluent pentane for the hydrometalated products, and more polar solvents for the dimer), elem. anal., (the org. products identified by GC and NMR); | A n/a B 20% C 8% D n/a |

-
-
68258-20-8
2-(2,2-dimethylcyclopropyl)acetic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
558-37-2
tert-butylethylene

-
C
-
691-37-2
4-Methyl-1-pentene

-
D
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| at 496.9℃; for 0.25h; Product distribution; Thermodynamic data; Rate constant; | A 11% B n/a C n/a D n/a E n/a |

-
-
85-44-9
phthalic anhydride

-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

-
-
98-11-3
benzenesulfonic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
558-37-2
tert-butylethylene

-
C
-
563-78-0
2,3-Dimethyl-1-butene


| Conditions | Yield |
|---|---|
| With zinc(II) chloride |

| Conditions | Yield |
|---|---|
| With magnesium oxide at 265℃; | |
| With lead(II) oxide at 280 - 290℃; |

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide; silica gel at 300℃; bei der Destillation; |
-
-
119-64-2
tetralin

-
-
594-57-0
2-chloro-2,3-dimethylbutane

-
-
557-20-0
diethylzinc

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
560-21-4
2,3,3-Trimethyl-pentane

-
C
-
79-29-8
2,3-dimethylbutane

-
D
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| at 3℃; |


| Conditions | Yield |
|---|---|
| With aluminum(III) sulfate at 275℃; | |
| With sodium permutite at 310℃; | |
| With phosphorus pentoxide; silica gel at 300℃; |
-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With oxalic acid at 110 - 120℃; | |
| With ethylmagnesium bromide at 340℃; | |
| ueber das Gleichgewicht zwischen den verschiedenen Dimethylbutenen; |
-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With aluminum oxide at 300℃; | |
| With aluminum(III) sulfate at 275℃; | |
| With phosphoric acid at 300℃; under 128714 Torr; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic anhydride | |
| With aluminum oxide at 300℃; | |
| Multi-step reaction with 2 steps 1: 14 percent Chromat. / γ-Al2O3 / 325 °C / variation of temperature and reagent 2: 27 percent / NaX zeolite / 300 °C View Scheme | |
| Multi-step reaction with 2 steps 1: β-naphthalene-sulfonic acid View Scheme |

| Conditions | Yield |
|---|---|
| With iodine | |
| With oxalic acid at 100℃; | |
| With naphthalene-2-sulfonate |
-
-
594-52-5
2-bromo-2,3-dimethylbutane

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With triethanolamine; 2-Butoxyethanol | |
| With potassium carbonate | |
| With potassium acetate; acetic acid | |
| With potassium hydroxide |
-
-
859183-56-5
2-ethoxy-1-bromo-2,3-dimethyl-butane

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With propan-1-ol; zinc |
-
-
5857-68-1
di-t-butyl-1,1 ethylene

-
-
162109-20-8
1-bromo-4-naphthalenesulfonic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
558-37-2
tert-butylethylene

-
C
-
563-78-0
2,3-Dimethyl-1-butene

-
D
-
115-11-7
isobutene


| Conditions | Yield |
|---|---|
| at 400℃; |
-
-
4806-33-1
2,3-dimethyl-2-butyl acetate

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| at 400℃; |
-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

-
-
144-62-7
oxalic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

-
-
594-60-5
2,3-dimethylbutan-2-ol

-
-
144-62-7
oxalic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| ein Gleichgewichtsgemisch ist erhalten worden bei der Dehydratisierung; |

-
-
594-60-5
2,3-dimethylbutan-2-ol

-
-
104-15-4
toluene-4-sulfonic acid

-
A
-
563-79-1
2,3-Dimethyl-2-butene

-
B
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| ein Gleichgewichtsgemisch ist erhalten worden bei der Dehydratisierung; |


| Conditions | Yield |
|---|---|
| With sulfuric acid; α,α′-bis(2-pyridyl(tert-butyl)phosphino)-o-xylene; water; palladium(II) acetylacetonate; acetic acid at 20 - 100℃; under 30003 Torr; for 20h; Inert atmosphere; Autoclave; | 99% |
-
-
1631-73-8
trimethylstannane

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
15095-89-3
1-Trimethylstannyl-2,3-dimethyl-butan

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); UV-irradiation at 10°C for 80 h in a sealed tube;; | 98% |
| Irradiation (UV/VIS); UV-irradiation at 10°C for 80 h in a sealed tube;; | 98% |

| Conditions | Yield |
|---|---|
| aluminum oxide at -0.1℃; for 0.166667h; | 95% |
| x In nitromethane at 25℃; for 0.25h; | 88% |
| NiX(21) zeolite at -0.1℃; Rate constant; or ZnX(21) zeolite; |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1172581-29-1
2-(2,3-dimethylbutyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With C20H22AlN2PSe In neat (no solvent) at 30℃; for 8h; Schlenk technique; Glovebox; Inert atmosphere; chemoselective reaction; | 90% |
| With chlorocarbonylbis(triphenylphosphine)rhodium(I) In dichloromethane at 20 - 40℃; for 3.5h; Inert atmosphere; | 88% |
| With C17H19Cl2CoN3O; sodium t-butanolate In tetrahydrofuran at 25℃; for 1h; Inert atmosphere; regioselective reaction; | 78 mg |
-
-
12176-06-6
hydrido(tricarbonyl)(cyclopentadienyl)molybdenum

-
-
1493-13-6
trifluorormethanesulfonic acid

-
-
563-78-0
2,3-Dimethyl-1-butene

-
B
-
79-29-8
2,3-dimethylbutane

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 react. at -75°C for 5 min; quenching by pyridine, detecting by n.m.r. spectroscopy; | A n/a B 86% |


| Conditions | Yield |
|---|---|
| With C39H60N4O4; nickel(II) tetrafluoroborate hexahydrate In 1,2-dichloro-ethane at 60℃; for 32h; optical yield given as %ee; | 86% |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
62127-35-9
2,3-dimethyl-1-buthyl fluoride

| Conditions | Yield |
|---|---|
| With HF-melamine; hydrogen fluoride In tetrahydrofuran at 0℃; for 1h; | 85% |
-
-
13081-18-0
ethyl-3,3,3-trifluoropyruvate

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With C39H60N4O4; magnesium triflate In dichloromethane at 30℃; for 24h; optical yield given as %ee; enantioselective reaction; | 83% |
-
-
75-25-2
Bromoform

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
22715-57-7
2,2,3,3-tetramethyl-1,1-dibromocyclopropane

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In pentane at 0 - 20℃; for 2.33333h; Inert atmosphere; | 82% |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With phosgene; potassium tert-butylate In toluene at 20℃; for 12h; Inert atmosphere; | 82% |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
72221-03-5
2,3-dimethyl-1,2-epoxybutane

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; 3-chloro-benzenecarboperoxoic acid In dichloromethane for 12h; Ambient temperature; | 81% |
| With sodium periodate; >*1.5H2O In dichloromethane; water at 2℃; for 15h; | 59% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0 - 22℃; | 49% |
-
-
55962-05-5
[N-(p-tolylsulfonyl)imino]phenyliodinane

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
956102-58-2
2-isopropyl-2-methyl-1-[(4-methyl-phenyl)sulfonyl]aziridine

| Conditions | Yield |
|---|---|
| With Cu(C6H5NC6H2(C(CH3)3)2O)2 In acetonitrile at 20℃; for 5h; Schlenk technique; Inert atmosphere; | 81% |
-
-
99-87-6
4-methylisopropylbenzene

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
2084-69-7
1,1,2,4,4,7-hexamethyl-1,2,3,4-tetrahydro-naphthalene

| Conditions | Yield |
|---|---|
| Stage #1: 4-methylisopropylbenzene; 2,3-Dimethyl-1-butene With isobutyryl chloride for 0.5h; Stage #2: With antimony(III) chloride In dichloromethane at 25℃; for 2h; Reagent/catalyst; Solvent; Temperature; | 80.24% |
| With aluminium trichloride; tertiary butyl chloride; Aliquat 336 In cyclohexane at 20℃; | 47.5% |
| With aluminum (III) chloride; tertiary butyl chloride In dichloromethane; cyclohexane at 10℃; for 0.416667h; Temperature; Solvent; Reagent/catalyst; | 44.22% |
| With ion-exchange resin + form> | |
| With aluminum (III) chloride; tertiary butyl chloride In cyclohexane |
-
-
102690-46-0
phenyl buta-2,3-dienoate

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| With ethylaluminum dichloride In hexane; dichloromethane at 20℃; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; water In acetonitrile at 20℃; for 48h; Irradiation; Inert atmosphere; Sealed tube; | 75% |
-
-
16312-79-1
4-methylurazole

-
-
563-78-0
2,3-Dimethyl-1-butene

| Conditions | Yield |
|---|---|
| In ethyl acetate Ambient temperature; | A 73% B 15% |
| In ethyl acetate for 12h; Ambient temperature; | A 73% B 15% |

-
-
50-00-0
formaldehyd

-
-
563-78-0
2,3-Dimethyl-1-butene

-
A
-
74126-47-9
3,4-dimethylpent-3-en-1-ol

-
B
-
76019-22-2
3-methylene-4-methylpentan-1-ol

| Conditions | Yield |
|---|---|
| With dimethylaluminum chloride In dichloromethane at 25℃; for 12h; | A 24% B 72% |


| Conditions | Yield |
|---|---|
| Stage #1: 2,3-Dimethyl-1-butene With n-butyllithium; potassium tert-butylate In tetrahydrofuran at -78 - -45℃; for 1.08333h; Stage #2: epichlorohydrin In tetrahydrofuran at -78℃; for 0.5h; | 72% |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 8h; Heating; | 71% |
-
-
109976-00-3
1,2,3-tri-tert-butylazadiboridine

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
367941-22-8
1,2,5,-tri-tert-butyl-3-isopropyl-3-methyl-1-aza-2,5-diboracyclopentane

| Conditions | Yield |
|---|---|
| In pentane soln. of compds. stirred at room temp. for 3 h; volatiles removed in vac., pure liquid product condensed into cooled receiver at 60°C/0.001 mbar; elem. anal.; | 71% |
-
-
872513-93-4
(2S)-methyl 2-N-benzoylamino-5-methylhex-4-enoate

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
1440507-70-9
(S,E)-methyl 2-benzamido-5,6-dimethylhept-4-enoate

| Conditions | Yield |
|---|---|
| With Hoveyda-Grubbs catalyst second generation In benzene at 100℃; for 24h; Inert atmosphere; Sealed tube; | 71% |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
228861-07-2
(22R,5'ξ)-22-acetoxy-22-(5'-isopropyl-5'-methylisoxazolin-3'-yl)-6,6-ethylenedioxy-3α,5-cyclo-23,24-dinorcholane

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; triethylamine In chloroform for 2h; | 70% |

| Conditions | Yield |
|---|---|
| With 2,4,6-Triisopropylthiophenol; C36H16F16IrN4(1+)*F6P(1-) In 1,4-dioxane at 35℃; Glovebox; Irradiation; chemoselective reaction; | 70% |

| Conditions | Yield |
|---|---|
| With acetylacetonatodicarbonylrhodium(l); C50H59Fe2NP2; hydrogen In toluene at 120℃; under 56255.6 Torr; for 16h; | 69% |
| With hydrogen; carbonylhydridetris(triphenylphosphine)rhodium(I); (-)-Diop at 95℃; under 40 Torr; for 96h; Yield given; | |
| With carbonylhydridetris(triphenylphosphine)rhodium(I); hydrogen In toluene at 60℃; under 11400 Torr; for 2h; Product distribution; Further Variations:; Solvents; Reagents; Pressures; |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate In chloroform for 24h; | 68% |
-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
684-16-2
Hexafluoroacetone

-
-
16203-14-8
1,1,1-trifluoro-4,5-dimethyl-2-trifluoromethyl-hex-4-en-2-ol

| Conditions | Yield |
|---|---|
| With 3 Å molecular sieves at 100℃; for 0.166667h; Microwave irradiation; | 67% |

-
-
563-78-0
2,3-Dimethyl-1-butene

-
-
1356386-29-2
C2B10H11CH2CH(CH3)CH(CH3)2

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: thallium(I) chloride; (Ar); using Schlenk techniques; addn. of soln. of HBCl2*dioxane in CH2Cl2 to stirred soln. of 2,2-dimethyl-1-butene in CH2Cl2 for 10 min, stirring at room temp. for 2 h, addn. to suspn. of Tl2C2B9H11 in CH2Cl2 for 5 min, stirring at room temp. for 18 h; filtration through paper filter, washing of ppt. on filter with CH2Cl2, evapn. with silica, placing on top of silica column in hexane, eluting with hexane, evapn., drying under vac.; | 66% |


| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In hexane at -30 - 20℃; for 0.166667h; Ene Reaction; Inert atmosphere; regioselective reaction; | A 31% B 63% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

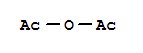


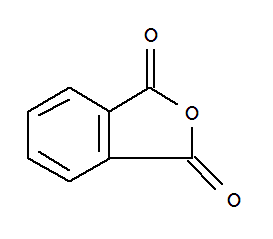


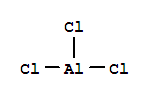








 F,
F, Xn
Xn