-
Name
2,6-Diiodo-4-nitrophenol
- EINECS 206-170-4
- CAS No. 305-85-1
- Article Data39
- CAS DataBase
- Density 2.717 g/cm3
- Solubility very sparingly soluble in water
- Melting Point 152-154 °C(lit.)
- Formula C6H3I2NO3
- Boiling Point 310.6 °C at 760 mmHg
- Molecular Weight 390.904
- Flash Point 141.6 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance yellow crystalline solid
- Safety 26-28-36/37/39-45-28A
- Risk Codes 20/21/22-36/37/38-25
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  T
T
- Synonyms 4-Nitro-2,6-diiodophenol;Ancylol;Disofen;Disophenol;Iodofen;Iodophen;Syngamix;
- PSA 66.05000
- LogP 3.03280
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogenchloride; potassium iodate; potassium iodide In methanol; water at 20℃; | 99% |
| With sodium periodate; sulfuric acid; iodine; potassium iodide; sodium sulfite In water; acetic acid at 25℃; for 4h; | 98% |
| With N-iodo-S-methyl-S-phenyl sulfoximine; acetic acid at 22℃; for 3h; Sealed tube; | 98% |

| Conditions | Yield |
|---|---|
| With iodine; silver sulfate In ethanol at 20℃; for 0.5h; | A 22% B 50% |
| With sulfuric acid; dihydrogen peroxide; potassium iodide In methanol at 60℃; for 2h; | A 18% B 42% |
| With iodine; silver nitrate In acetonitrile for 2h; Ambient temperature; | A 5% B 12% |

| Conditions | Yield |
|---|---|
| With sodium nitrite |
-
-
609-23-4
2,4,6-Triiodophenol

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol

-
B
-
20294-48-8
diiodo-4,6 nitro-2 phenol

| Conditions | Yield |
|---|---|
| With acetic acid; sodium nitrite; benzene Trennung durch fraktionierte Krystallisation der Kaliumsalze; | |
| With acetic acid; sodium nitrite; benzene |

| Conditions | Yield |
|---|---|
| With ethanol; iodine; mercury(II) oxide |

| Conditions | Yield |
|---|---|
| With iodine; mercury(II) oxide |
-
-
96-97-9
5-nitrosalicylic acid

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol

-
B
-
861565-06-2
2-hydroxy-3-iodo-5-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With ethanol; iodine; mercury(II) oxide |
-
-
28177-54-0
2,6-diiodo-phenol

-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| durch Nitrierung; |

| Conditions | Yield |
|---|---|
| With nitric acid |
-
-
16825-79-9
1-iodyl-4-nitrobenzene

-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With sodium nitrite | |
| With sodium azide |


| Conditions | Yield |
|---|---|
| durch Jodierung; |

| Conditions | Yield |
|---|---|
| bei der Jodierung; |
-
-
64-17-5
ethanol

-
-
96-97-9
5-nitrosalicylic acid

-
-
7553-56-2
iodine

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol

-
B
-
861565-06-2
2-hydroxy-3-iodo-5-nitro-benzoic acid

-
-
609-23-4
2,4,6-Triiodophenol

-
-
64-19-7
acetic acid

-
-
71-43-2
benzene

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol

-
B
-
20294-48-8
diiodo-4,6 nitro-2 phenol

| Conditions | Yield |
|---|---|
| at 12 - 15℃; |


| Conditions | Yield |
|---|---|
| With ethanol; iodine; mercury |
-
-
616-59-1
2-hydroxyl-5-nitrobenzenesulfonic acid

-
-
7553-56-2
iodine

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol


| Conditions | Yield |
|---|---|
| With iodine; mercury(II) oxide |
-
-
16825-79-9
1-iodyl-4-nitrobenzene

-
A
-
100-25-4
para-dinitrobenzene

-
B
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
16825-79-9
1-iodyl-4-nitrobenzene

-
A
-
636-98-6
p-nitrobenzene iodide

-
B
-
100-25-4
para-dinitrobenzene

-
C
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
16825-79-9
1-iodyl-4-nitrobenzene

-
A
-
1516-60-5
4-nitrophenyl azide

-
B
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
28177-54-0
2,6-diiodo-phenol

-
-
7697-37-2
nitric acid

-
-
64-19-7
acetic acid

-
A
-
305-85-1
2,6-diiodo-4-nitrophenol

-
B
-
13073-26-2
2-iodo-6-nitro-phenol

| Conditions | Yield |
|---|---|
| at -5℃; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 25 percent / AgNO3, Br2 / acetonitrile / 2 h / Ambient temperature 2: 5 percent / AgNO3, I2 / acetonitrile / 2 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: 29 percent / AgNO3, I2 / acetonitrile / 2 h / Ambient temperature 2: 5 percent / AgNO3, I2 / acetonitrile / 2 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CS2; iodine 2: durch Nitrierung View Scheme | |
| Multi-step reaction with 2 steps 1: CS2; iodine 2: nitric acid View Scheme |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 60℃; | 95% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
77-78-1
dimethyl sulfate

-
-
31106-75-9
1-methoxy-2,6-diiodido-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 0℃; for 18h; Reflux; Schlenk technique; | 92% |
| With potassium carbonate In acetone at 65℃; for 18h; Inert atmosphere; | 87% |
| With potassium carbonate In acetone at 0 - 65℃; for 18h; Inert atmosphere; | 87% |
| With alkali |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
851077-63-9
2,6-diiodo-4-nitrophenyl 4-methylbenzenesulfonate

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 20℃; | 90% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
18880-00-7
1-(bromomethyl)-4-(1,1-dimethylethyl)benzene

-
-
1423135-83-4
4-(t-butyl)benzyl 2,6-diiodo-4-nitrophenyl ether

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 24h; Reflux; | 80% |

| Conditions | Yield |
|---|---|
| With pyridine; copper(I) oxide for 2h; Reflux; | 78% |
| With pyridine; copper(I) oxide for 2h; Reflux; | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-diiodo-4-nitrophenol With copper(l) iodide; triphenylphosphine; (S)-1-Pyrrolidin-2-yl-methanol; palladium on activated charcoal In water at 25℃; for 1h; Sonogashira coupling; Stage #2: pent-1-yn-3-ol In water at 80℃; for 3h; | 75% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-diiodo-4-nitrophenol With copper(l) iodide; triphenylphosphine; (S)-1-Pyrrolidin-2-yl-methanol; palladium on activated charcoal In water at 25℃; for 1h; Sonogashira coupling; Stage #2: 2-methyl-but-3-yn-2-ol In water at 80℃; for 3h; | 75% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 16h; | 71% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
106-89-8
epichlorohydrin

-
-
95646-27-8
1-<2',6'-diiodo-4'-nitrophenoxy->-2,3-epoxy propane

| Conditions | Yield |
|---|---|
| With pyridine at 90 - 95℃; for 1h; | 70% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-diiodo-4-nitrophenol With copper(l) iodide; triphenylphosphine; (S)-1-Pyrrolidin-2-yl-methanol; palladium on activated charcoal In water at 25℃; for 1h; Sonogashira coupling; Stage #2: but-3-yn-2-ol In water at 80℃; for 3h; | 70% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
88284-48-4
2-(trimethylsilyl)phenyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile at 20℃; for 24h; | 65% |

| Conditions | Yield |
|---|---|
| With pyridine; copper(I) oxide Inert atmosphere; Reflux; | 63% |
-
-
4549-31-9
1,7-dibromoheptane

-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
1374327-23-7
1,3-dibromo-2-(7-bromoheptyloxy)-5-nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile | 52% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
106-95-6
allyl bromide

-
-
95646-25-6
2,6-diiodo-4-nitro-(2-allyl)-oxybenzene

| Conditions | Yield |
|---|---|
| With sodium carbonate In N,N-dimethyl-formamide at 80℃; for 6h; | 50% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
74-88-4
methyl iodide

-
-
31106-75-9
1-methoxy-2,6-diiodido-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 60h; | 42% |
| With potassium carbonate In acetone for 6h; Heating; |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper diacetate; diisopropylamine at 20℃; for 1h; | 40% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
89640-51-7
4-amino-2-iodophenol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium dithionite at 75℃; | 30% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate; XPhos In 1,2-dimethoxyethane; water Suzuki Coupling; Inert atmosphere; Reflux; | 24% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium carbonate In 1,2-dimethoxyethane; water for 5h; Suzuki Coupling; Inert atmosphere; Reflux; | 22% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
39546-47-9
p-chlorobenzylisocyanide

-
-
107-11-9
1-amino-2-propene

-
-
590-86-3
isovaleraldehyde

-
-
1007383-12-1
C22H24ClI2N3O3

| Conditions | Yield |
|---|---|
| With ammonium chloride In water; toluene at 100℃; for 20h; Ugi-Smiles reaction; Inert atmosphere; | 18% |

-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium carbonate In 1,2-dimethoxyethane; water at 90℃; for 14h; Suzuki Coupling; Inert atmosphere; | 18% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium carbonate In 1,2-dimethoxyethane; water Suzuki Coupling; Inert atmosphere; Reflux; | 8.7% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium carbonate In 1,2-dimethoxyethane; water for 7h; Suzuki Coupling; Inert atmosphere; Reflux; | 5.5% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium carbonate In 1,2-dimethoxyethane; water Suzuki Coupling; Inert atmosphere; Reflux; | 1.6% |
-
-
305-85-1
2,6-diiodo-4-nitrophenol

-
-
20389-01-9
2,6-diiodo-p-benzoquinone

| Conditions | Yield |
|---|---|
| With tin; acetic acid anschliessendes Behandeln mit CrO3 in wss. H2SO4; | |
| Multi-step reaction with 3 steps 1: sodium dithionite; aqueous sodium carbonate 2: ethanolic H2SO4 / Diazotization.anschliessendes Kochen 3: aqueous KOH; CrO3; water View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(ll) chloride | |
| With sodium dithionite; sodium carbonate | |
| With tin; acetic acid | |
| With hydrochloride of tin dichloride | |
| With tin(II) chloride dihdyrate In ethanol at 78℃; for 5h; | 4 g |
2,6-DIIODO-4-NITROPHENOL Chemical Properties
Empirical Formula: C6H3I2NO3 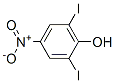
Molecular Weight: 390.9019
IUPAC Name: 2,6-Diiodo-4-nitrophenol
Synonyms: Phenol, 2,6-diiodo-4-nitro- ; Disofen ; Diisophenol ; Ancylol
Melting Point: 152-154 °C(lit.)
Flash Point: 141.6 °C
Boiling Point: 310.6 °C at 760 mmHg
Density of 2,6-Diiodo-4-nitrophenol (305-85-1): 2.717 g/cm3
Vapour Pressure: 0.000325 mmHg at 25°C
Index of Refraction: 1.782
Water Solubility: 2,6-Diiodo-4-nitrophenol (305-85-1) is very sparingly soluble
Sensitive: Light Sensitive
Stability: 2,6-Diiodo-4-nitrophenol (305-85-1) is stable.
Appearance: 2,6-Diiodo-4-nitrophenol (305-85-1) is a yellow crystalline solid
2,6-DIIODO-4-NITROPHENOL Uses
2,6-Diiodo-4-nitrophenol (305-85-1) is used against hookworm.
2,6-DIIODO-4-NITROPHENOL Toxicity Data With Reference
| 1. | orl-rat LD50:170 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 2. | ipr-rat LD50:105 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 3. | scu-rat LD50:122 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 4. | ivn-rat LD50:105 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 5. | orl-mus LD50:212 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 6. | ipr-mus LD50:107 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 7. | scu-mus LD50:110 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 8. | ivn-mus LD50:88 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 6 (1964),232. | ||
| 9. | par-uns LDLo:36 mg/kg | FAZMAE Fortschritte der Arzneimittelforschung. 17 (1973),108. |
2,6-DIIODO-4-NITROPHENOL Consensus Reports
Reported in EPA TSCA Inventory.
2,6-DIIODO-4-NITROPHENOL Safety Profile
Poison by ingestion, intraperitoneal, subcutaneous, intravenous, and parenteral routes. An anthelmintic. When heated to decomposition it emits very toxic fumes of I− and NOx. See also NITRO COMPOUNDS of AROMATIC HYDROCARBONS.
Safety: DANGER: POISON, causes cyanosis; skin, eye, lung irritation. Safety glasses, good ventilation.
Hazard Codes: Xn: Harmful ![]() ,
,
T: Toxic 
Risk Statements: 20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
25: Toxic if swallowed
Safety Statements: 26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
RIDADR UN 2811 6.1/PG 3
WGK Germany: 3
RTECS: SL0750000
HazardClass: 6.1
PackingGroup: III
2,6-DIIODO-4-NITROPHENOL Specification
2,6-Diiodo-4-nitrophenol (305-85-1) is a yellow crystalline solid used against hookworm. It is a halogenated and nitrated phenol. Reacts as a weak acid. It is stable and very sparingly soluble in water.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 305851-04-1
- 305858-63-3
- 30586-13-1
- 30586-93-7
- 3058-98-8
- 3059-67-4
- 30599-15-6
- 3059-97-0
- 306-03-6
- 3060-41-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View