-
Name
2-Phenoxyethanol
- EINECS 204-589-7
- CAS No. 122-99-6
- Article Data122
- CAS DataBase
- Density 1.102 g/cm3
- Solubility Soluble in alcohol, ether and sodium hydroxide solution. Can dissolve oil, natural resin, alkyd, cellulose acetate, ethyecellulose, nitrocellulose and polyvinyl acetate. Miscible with glycerin, propylene glycol, ethanol, ethyl ether, benzene.
- Melting Point 11-13 °C
- Formula C8H10O2
- Boiling Point 245.199 °C at 760 mmHg
- Molecular Weight 138.166
- Flash Point 105.275 °C
- Transport Information
- Appearance colorless or light yellow liquid
- Safety 26
- Risk Codes 22-36
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Sepicide LD;b-Hydroxyethyl phenyl ether;b-Phenoxyethanol;b-Phenoxyethyl alcohol;(2-Hydroxyethoxy)benzene;1-Hydroxy-2-phenoxyethane;2-Hydroxyethyl phenyl ether;2-Phenoxyethyl alcohol;Arosol;Dalpad A;Dowanol EP;Dowanol EPh;Emeressence1160;Ethylene glycol monophenyl ether;Ethylene glycol phenyl ether;Euxyl PE90120;H 4644;Hisolve EPH;NSC 1864;Newpol EFP;PHE (alcohol);PHE-G;PHE-S;Phenova;Phenoxethol;Phenoxetol;Phenoxyethanol;Phenoxyethyl alcohol;Phenyl cellosolve;Plastilit DS 3431;Rokafenol F 1;
- PSA 29.46000
- LogP 1.05770
Synthetic route

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 18h; Ambient temperature; | 100% |
-
-
22855-36-3
bis(2-phenoxyethyl)carbonate

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With methanol; sodium methylate Reflux; | 100% |
-
-
29279-13-8
S-ethyl phenoxythioacetate

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With tetrabutylammonium borohydride In chloroform for 3h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With [ReOCl2(1,2-bis(diphenylphosphino)ethane)]; hydrogen; potassium tetraphenylborate In tetrahydrofuran at 160℃; under 30003 Torr; for 24h; Autoclave; Inert atmosphere; | 99% |
| With sodium tetrahydroborate; titanium tetrachloride In 1,2-dimethoxyethane for 14h; Ambient temperature; | 95% |
| With strain of the zygomycete fungus S. racemosum MUT 770 In dimethyl sulfoxide for 72h; Enzymatic reaction; | 94% |

| Conditions | Yield |
|---|---|
| With potassium fluoride | 99% |
| With potassium iodide | 97.7% |
| With tetrabutyl ammonium fluoride In N,N-dimethyl-formamide at 170℃; for 0.583333h; Inert atmosphere; Schlenk technique; | 94% |
| With tetraethylammonium iodide |

| Conditions | Yield |
|---|---|
| With potassium carbonate; copper dichloride at 130℃; for 20h; Inert atmosphere; Schlenk technique; | 99% |

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With indium; ammonium chloride In ethanol for 18h; Debenzylation; Heating; | 98% |
| With indium; ammonium chloride In methanol for 18h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 3h; | 98% |
| Stage #1: phenoxyacetic acid ethyl ester With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 4h; Stage #2: With sodium hydroxide In tetrahydrofuran; methanol; water | 88% |
| With lithium aluminium tetrahydride In diethyl ether at 0 - 20℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| With strain of the zygomycete fungus S. racemosum MUT 770 In dimethyl sulfoxide for 72h; Enzymatic reaction; | 96% |
| With diisobutylaluminium hydride In toluene at 20℃; for 1.5h; | 73% |
-
-
143038-69-1
tert-Butyl-dimethyl-(2-phenoxy-ethoxy)-silane

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In methanol; dichloromethane at 20 - 25℃; for 0.333333h; Flow reactor; | 96% |
| With P(Me2CHNCH2CH2)3N In dimethyl sulfoxide at 80℃; for 36h; desilylation; | 95% |

| Conditions | Yield |
|---|---|
| With methanol; potassium permanganate; trimethylsulphonium iodide at 25℃; under 760.051 Torr; chemoselective reaction; | 96% |
| With methanol; potassium permanganate at 25℃; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 12h; Heating; | 95% |
| With sodium hydroxide In water at 90℃; for 12h; | 66.7% |
| With potassium carbonate In N,N-dimethyl-formamide | 47% |
-
-
75839-76-8
Phenoxy-thioacetic acid S-tert-butyl ester

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 23h; Ambient temperature; | 92% |

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| Stage #1: 1-phenoxy-2-trityloxyethane With C16H17NNa2 In tetrahydrofuran at 0℃; for 16h; Inert atmosphere; Stage #2: With water In tetrahydrofuran at 0℃; Inert atmosphere; | 92% |
-
-
139622-54-1
phenoxyethyl tetrahydro-2H-pyran-2-yl ether

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In methanol at 23℃; for 16h; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate at 130℃; for 24h; Inert atmosphere; | 91% |
-
-
16654-47-0
2-Phenoxy-aethyl-trimethylsilylaether

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With montmorillonite K-10 In methanol for 1.5h; Ambient temperature; | 89% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; urea; zinc(II) oxide at 160 - 180℃; | 89% |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonate)oxovanadium; methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate at 120℃; for 48h; | A 85% B 87% |
-
-
1334498-65-5
S-(2-oxo-2-phenylethyl) O-(2-phenoxyethyl)carbonodithioate

-
A
-
122-99-6
2-Phenoxyethanol

-
B
-
98-86-2
acetophenone

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 15h; Irradiation; Inert atmosphere; | A 84% B 86% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 4h; Heating; selective phenylation of one OH-group in var. glycols; | 85% |
| In dichloromethane for 4h; Heating; | 85% |
| In dichloromethane for 5h; Heating; | 85% |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 6h; | 82% |
| at 150℃; im geschlossenen Rohr; |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 6h; | 81% |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 7h; | 80% |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 8h; | 79% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 78℃; for 1h; | 78% |
| Stage #1: phenol With sodium hydroxide In water Stage #2: 2-chloro-ethanol In water at 80 - 95℃; for 0.833333h; | 68% |
| Stage #1: phenol With sodium hydroxide In water for 0.166667h; Glovebox; Inert atmosphere; Stage #2: 2-chloro-ethanol In water for 40h; Reflux; Inert atmosphere; Glovebox; | 53.1% |
-
-
41532-81-4
2-methoxyethyl phenyl ether

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With tetrachlorosilane; sodium iodide In dichloromethane; acetonitrile at 25℃; for 16h; | 75% |
-
-
121335-16-8
1-(5-Nitro-3-oxy-benzotriazol-1-yl)-2-phenoxy-ethanone

-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 2h; Ambient temperature; | 75% |

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate; iodine In acetonitrile for 10h; Heating; | 70% |
-
-
87841-77-8
2-(2-Phenoxy-ethylazo)-prop-2-yl-hydroperoxide

-
-
109-92-2
ethyl vinyl ether

-
A
-
493-08-3
chromane

-
B
-
122-99-6
2-Phenoxyethanol

-
C
-
622-85-5
propoxybenzene

-
D
-
19790-62-6
4-phenoxybutyraldehyde

-
E
-
87841-94-9
2-phenoxypentanal

-
F
-
103-73-1
Phenetole

| Conditions | Yield |
|---|---|
| at 50℃; Mechanism; | A n/a B 1% C 0.6% D 52% E 0.7% F 7% G n/a |


| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenoxyethanol; p-toluenesulfonyl chloride With dmap In dichloromethane at 20℃; Stage #2: With triethylamine In dichloromethane at 20℃; | 100% |
| With pyridine | 86% |
| With pyridine at 20℃; for 4h; | 24% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; oxygen; bismuth(III) nitrate; palladium on activated charcoal at 90℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With pyridine | 100% |
-
-
122-99-6
2-Phenoxyethanol

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
1579951-66-8
2-phenoxyethyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at -78 - 20℃; for 0.5h; | 100% |
| With 2,6-dimethylpyridine In dichloromethane at 0 - 20℃; for 1.5h; Inert atmosphere; | |
| With triethylamine In dichloromethane at -30℃; for 1h; Inert atmosphere; |
-
-
122-99-6
2-Phenoxyethanol

| Conditions | Yield |
|---|---|
| With C29H28F6N4OS at 20℃; for 3h; enantioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; chlorine at -20℃; for 0.5h; Reagent/catalyst; Temperature; | 99.48% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; benzoic acid | 99.4% |

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride; bis(4-chlorophenyl)sulfide; magnesium chloride at 30℃; for 0.5h; | 99.24% |

| Conditions | Yield |
|---|---|
| With dmap; oxo[hexa(trifluoroacetato)]tetrazinc In neat (no solvent) at 80℃; for 24h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 99% |
| With toluene-4-sulfonic acid; hydroquinone Entfernen des entstehenden Methanols; | |
| With sulfuric acid; hydroquinone Entfernen des entstehenden Methanols; |
-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
122-99-6
2-Phenoxyethanol

-
-
139622-54-1
phenoxyethyl tetrahydro-2H-pyran-2-yl ether

| Conditions | Yield |
|---|---|
| With sulfuric acid In dichloromethane for 0.0166667h; Ambient temperature; | 99% |
| With phosphotungstic acid In toluene at 20℃; for 1h; | 97% |
| With H6P2W18O62 In toluene at 20℃; for 2h; | 96% |
| With aminosulfonic acid at 15℃; for 6h; | 93% |
| With aluminium(III) triflate at 20℃; for 15h; | 90% |
-
-
122-99-6
2-Phenoxyethanol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
143038-69-1
tert-Butyl-dimethyl-(2-phenoxy-ethoxy)-silane

| Conditions | Yield |
|---|---|
| With P(MeNCH2CH2)3N; triethylamine In acetonitrile at 24℃; for 0.3h; | 99% |
| Stage #1: 2-Phenoxyethanol With 1H-imidazole In dichloromethane for 0.0833333h; Stage #2: tert-butyldimethylsilyl chloride In dichloromethane at 20℃; for 1h; | 97% |
-
-
122-99-6
2-Phenoxyethanol

-
-
124-63-0
methanesulfonyl chloride

-
-
141482-06-6
1-methanesulfonyloxy-2-(phenoxy)ethane

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 2h; | 99% |
| Stage #1: 2-Phenoxyethanol With triethylamine In dichloromethane at 0℃; for 0.166667h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: methanesulfonyl chloride In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; Schlenk technique; Glovebox; | 95.3% |
| With triethylamine In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenoxyethanol With sodium hydride In tetrahydrofuran at 20℃; for 0.5h; Stage #2: p-nitrophenyl isothiocyanate In tetrahydrofuran at 20℃; for 18h; Further stages.; | 99% |

| Conditions | Yield |
|---|---|
| In toluene under an inert atm.; to a soln. of TiCl4 (11.9 mmol) in toluene was added a soln. of a ligand (23.7 mmol) in toluene; the soln. was concd. under vac.; the residue was washed with petroleum spirits; the solid was dried under vac.; elem. anal.; | 99% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 100℃; for 3h; Schlenk technique; Inert atmosphere; | 99% |
| With ethanol; 1-ethyl-3-methylimidazol-3-ium ethyl sulfate at 199.84℃; under 15001.5 Torr; for 2h; Autoclave; Inert atmosphere; |
-
-
122-99-6
2-Phenoxyethanol

-
-
18296-01-0
triethylsilyl formate

-
-
16654-60-7
2-Phenoxy-aethyl-triaethylsilylaether

| Conditions | Yield |
|---|---|
| With tris(2-diphenylphosphinoethyl)phosphine; iron(II) acetate In dichloromethane Glovebox; Inert atmosphere; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With nickel oxinate In neat (no solvent) at 25℃; | 99% |

| Conditions | Yield |
|---|---|
| In diethyl ether at 5 - 8℃; for 3h; | 98.6% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenoxyethanol With sodium hydride In tetrahydrofuran at 20℃; for 0.5h; Stage #2: 4-Methoxyphenyl isothiocyanate In tetrahydrofuran at 20℃; for 18h; Further stages.; | 98% |
-
-
122-99-6
2-Phenoxyethanol

-
-
58328-36-2
N,N-diethyl-N'-benzoyl-thiourea

-
-
79564-18-4
3-Benzoyl-1,1-diethyl-2-(2-phenoxy-ethyl)-isothiourea

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran for 18h; Ambient temperature; | 97% |

| Conditions | Yield |
|---|---|
| With benzenesulfonyl chloride In 1-methyl-pyrrolidin-2-one at 20 - 100℃; for 0.5h; | 96.8% |
| With 1-methyl-pyrrolidin-2-one; benzenesulfonyl chloride In 1,2-dichloro-ethane at 80℃; for 1.5h; | 96% |
| With pyridine; thionyl chloride In toluene at 10 - 80℃; Large scale; | 94% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 20℃; for 2h; Temperature; Cooling with ice; Inert atmosphere; | 96.4% |
2-Phenoxyethanol Specification
2-Phenoxyethanol is an organic compound with the formula C8H10O2, and its systematic name is the same with the product name. With the CAS registry number 122-99-6, it is also named as 2-Hydroxyethyl phenyl ether. It belongs to the product categories of Benzhydrols, Benzyl & Special Alcohols; Ethylene Glycols & Monofunctional Ethylene Glycols; Monofunctional Ethylene Glycols. Its EINECS number is 204-589-7. In addition, the molecular weight is 138.16. Its classification codes are: (1)Anesthetics; (2)Anti-Infective Agents; (3)Anti-infective agents, local; (4)Central Nervous System Agents; (5)Central Nervous System Depressants; (6)Mutation data; (7)Skin / Eye Irritant. This chemical should be sealed and stored in a cool and dry place. It is used as a fixative for perfumes, an insect repellent, a topical antiseptic, a solvent for cellulose acetate, some dyes, inks, and resins, and it is also used in organic synthesis.
Physical properties of 2-Phenoxyethanol are: (1)ACD/LogP: 1.247; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.25; (4)ACD/LogD (pH 7.4): 1.25; (5)ACD/BCF (pH 5.5): 5.22; (6)ACD/BCF (pH 7.4): 5.22; (7)ACD/KOC (pH 5.5): 113.65; (8)ACD/KOC (pH 7.4): 113.65; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 29.46 Å2; (13)Index of Refraction: 1.526; (14)Molar Refractivity: 39.099 cm3; (15)Molar Volume: 127.449 cm3; (16)Polarizability: 15.5×10-24cm3; (17)Surface Tension: 39.97 dyne/cm; (18)Density: 1.084 g/cm3; (19)Flash Point: 105.275 °C; (20)Enthalpy of Vaporization: 50.962 kJ/mol; (21)Boiling Point: 245.199 °C at 760 mmHg; (22)Vapour Pressure: 0.016 mmHg at 25°C.
Preparation: this chemical can be prepared by tert-butyl-dimethyl-(2-phenoxy-ethoxy)-silane at the temperature of 80 °C. This reaction will need reagent P(Me2CHNCH2CH2)3N and solvent dimethylsulfoxide with the reaction time of 36 hours. The yield is about 95%.
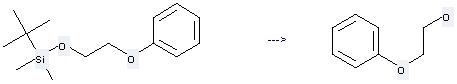
Uses of 2-Phenoxyethanol: it can be used to produce N-(2-phenoxyethoxy)phthalimide at the ambient temperature. It will need reagents diethyl azodicarboxylate, triphenylphosphine and solvent tetrahydrofuran with the reaction time of 16 hours. The yield is about 72%.

When you are using this chemical, please be cautious about it as the following:
This chemical is harmful if swallowed. It is irritating to eyes. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: c1ccc(cc1)OCCO
(2)Std. InChI: InChI=1S/C8H10O2/c9-6-7-10-8-4-2-1-3-5-8/h1-5,9H,6-7H2
(3)Std. InChIKey: QCDWFXQBSFUVSP-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 490mg/kg (490mg/kg) | BRAIN AND COVERINGS: OTHER DEGENERATIVE CHANGES | Toksikologicheskii Vestnik. Vol. (3), Pg. 37, 1996. |
| mouse | LD50 | oral | 933mg/kg (933mg/kg) | BRAIN AND COVERINGS: OTHER DEGENERATIVE CHANGES | Toksikologicheskii Vestnik. Vol. (3), Pg. 37, 1996. |
| rabbit | LD50 | skin | 5mL/kg (5mL/kg) | Union Carbide Data Sheet. Vol. 6/24/1958, | |
| rat | LD50 | intraperitoneal | 554mg/kg (554mg/kg) | BRAIN AND COVERINGS: OTHER DEGENERATIVE CHANGES | Toksikologicheskii Vestnik. Vol. (3), Pg. 37, 1996. |
| rat | LD50 | oral | 1260mg/kg (1260mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC GASTROINTESTINAL: OTHER CHANGES KIDNEY, URETER, AND BLADDER: OTHER CHANGES | Journal of Industrial Hygiene and Toxicology. Vol. 23, Pg. 259, 1941. |
| rat | LD50 | skin | 14422mg/kg (14422mg/kg) | LUNGS, THORAX, OR RESPIRATION: ACUTE PULMONARY EDEMA | Journal of the American College of Toxicology. Vol. 9, Pg. 259, 1990. |
Related Products
- 2-Phenoxyethanol
- 122996-47-8
- 123000-02-2
- 123-00-2
- 123005-57-2
- 123-01-3
- 123024-70-4
- 123-03-5
- 123036-51-1
- 123040-16-4
- 123040-69-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View