-
Name
3-Hydroxypropanenitrile
- EINECS 203-704-8
- CAS No. 109-78-4
- Article Data71
- CAS DataBase
- Density 1.04 g/cm3
- Solubility >=10 g/100 mL at 20 °C in water
- Melting Point -46 °C
- Formula C3H5NO
- Boiling Point 229.6 °C at 760 mmHg
- Molecular Weight 71.0788
- Flash Point 92.7 °C
- Transport Information 2810
- Appearance Colourless or straw-coloured liquid
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Hydracrylonitrile(8CI);1-Cyano-2-hydroxyethane;2-Cyanoethanol;2-Cyanoethyl alcohol;2-Hydroxycyanoethane;2-Hydroxyethyl cyanide;3-Hydroxypropanenitrile;Ethylene cyanhydrine;Ethylene cyanohydrin;Glycolcyanohydrin;NSC 2598;b-Cyanoethanol;b-Hydroxyethyl cyanide;b-Hydroxypropionitrile;
- PSA 44.02000
- LogP -0.10762
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium sulfide; benzyltri(n-butyl)ammonium chloride In water for 0.166667h; | A 98% B n/a |

| Conditions | Yield |
|---|---|
| With aluminum oxide; boron trifluoride diethyl etherate In propylene glycol at -10 - 10℃; for 28h; Reagent/catalyst; Temperature; | 97.3% |

| Conditions | Yield |
|---|---|
| With PEG400 for 6h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 50℃; for 0.2h; | 92% |
| With sodium hydroxide In water at 50℃; for 4h; | 65% |
| With water; copper(II) oxide |
-
-
90087-76-6, 89923-32-0
3-((tetrahydro-2H-pyran-2-yl)oxy)propanenitrile

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With silica-supported NaHSO4 In methanol at 20℃; for 0.25h; | 92% |
| With zinc(II) trifluoromethanesulfonate In methanol at 20℃; for 0.666667h; | 81% |

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With acetic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium nitrite at 20℃; for 18h; Green chemistry; chemoselective reaction; | 84% |

| Conditions | Yield |
|---|---|
| With sodium iodide In ethanol for 5h; Heating; | 80% |
| at 100℃; | |
| With ethanol |
-
-
131379-56-1
3-(1,1,1,3,3,3-hexamethyl-2-(trimethylsilyl)trisilan-2-yl)propanenitrile

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With 2,3-dimethyl-1,4-benzoquinone; tetrabutyl ammonium fluoride; oxygen In tetrahydrofuran for 0.833333h; Ambient temperature; | 71% |
-
-
107-13-1
acrylonitrile

-
-
108-95-2
phenol

-
A
-
3055-86-5
3-phenoxypropionitrile

-
B
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With tetramethyl ammoniumhydroxide In water at 60℃; for 0.5h; Temperature; Reagent/catalyst; Time; Michael Addition; Microwave irradiation; | A 68% B n/a |


| Conditions | Yield |
|---|---|
| With lithium hypochlorite In water Ambient temperature; | A 9.8% B 60% |
| With lithium hypochlorite In water Product distribution; Mechanism; Ambient temperature; other solvent and additives; var. cyclopropanamines; | A 9.8% B 60% |

| Conditions | Yield |
|---|---|
| With lithium aluminium hydride on silica gel In diethyl ether for 3h; Heating; | 54% |
-
-
2417-90-5
bromopropionitrile

-
A
-
64-19-7
acetic acid

-
B
-
107-13-1
acrylonitrile

-
C
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With oxygen; tetraethylammonium perchlorate In N,N-dimethyl-formamide at 20℃; electroreduction at -1.1 V; | A 40 % Chromat. B 10 % Chromat. C 36% |


| Conditions | Yield |
|---|---|
| With lithium hypochlorite In water Ambient temperature; | A 15% B 30 mg |

-
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

-
-
107-13-1
acrylonitrile

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| In water | 1.3% |


| Conditions | Yield |
|---|---|
| (i) NaN3, H2O, (ii) Pd, 2,5-dimethyl-hex-3-yne-2,5-diol, benzene; Multistep reaction; |
-
-
72784-43-1
methyl 1-aminocyclopropane-1-carboxylate

-
A
-
74-85-1
ethene

-
B
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With lithium hypochlorite In water at 5 - 10℃; for 0.333333h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Pt(Ppy3)4; water at 80℃; for 1h; Product distribution; var. platinum(0) catalysts; | |
| at 70℃; for 8h; Product distribution; The selective hydration with several metal oxide catalysts.; | |
| manganese(IV) oxide at 70℃; for 8h; Yield given; |
-
-
107-13-1
acrylonitrile

-
A
-
1656-48-0
2-cyanoethyl ether

-
B
-
79-06-1
2-propenamide

-
C
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| trans- at 80℃; Mechanism; Rate constant; Product distribution; in the absence or in the presence of different quantity of water; | |
| trans- at 25℃; Rate constant; Product distribution; also in the presence of different quantity of acetamide; | |
| sodium hydroxide In water at 80℃; Product distribution; |


| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 16h; |
-
-
62984-38-7
N-nitro-β-aminopropionitrile

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With sulfuric acid at 25 - 85℃; Kinetics; Mechanism; Thermodynamic data; ΔH(excit.), -ΔS(excit.); |
-
-
98-01-1
furfural

-
-
7732-18-5
water

-
-
100-85-6
N-benzyl-trimethylammonium hydroxide

-
-
107-13-1
acrylonitrile

-
A
-
101291-29-6
bis-(2-cyano-3ξ-[2]furyl-allyl)-ether

-
B
-
109-78-4
3-Hydroxypropionitrile


| Conditions | Yield |
|---|---|
| at 20℃; Kinetics; | |
| at 40℃; Kinetics; |

-
-
107-13-1
acrylonitrile

-
A
-
1656-48-0
2-cyanoethyl ether

-
B
-
79-06-1
2-propenamide

-
C
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With water; Ni(b),Cu(35) (X%) at 75℃; for 6h; Product distribution; Thermodynamic data; other time and catalyst composition; ΔE(excit); effect on the rate and product distribution; |


| Conditions | Yield |
|---|---|
| Reaktion von Calciumcyanid; | |
| Reaktion von Calciumcyanid; |

-
-
75-21-8
oxirane

-
-
74-90-8
hydrogen cyanide

-
-
143-33-9
sodium cyanide

-
-
7732-18-5
water

-
-
109-78-4
3-Hydroxypropionitrile

-
-
109-78-4
3-Hydroxypropionitrile

-
-
53868-56-7
3-hydroxypropanimidamide hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 3-Hydroxypropionitrile With acetyl chloride In methanol; toluene at 0 - 20℃; for 18h; Stage #2: With ammonia In methanol at 0 - 20℃; for 18h; | 100% |
| With hydrogenchloride; methanol; diethyl ether Umsetzen des Reaktionsprodukts mit NH3 in Metahnol bei 0grad; |
-
-
109-78-4
3-Hydroxypropionitrile

-
-
162611-40-7, 162611-41-8, 171081-08-6
5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl-(N,N-diisopropylamino)phosphorofluoridite

-
-
171081-12-2
O-[5'-O-(4,4'-dimethoxytrityl)thymidin-3'-yl] O-(2-cyanoethyl) phosphorofluoridite

| Conditions | Yield |
|---|---|
| With 1H-tetrazole In acetonitrile for 0.166667h; Ambient temperature; | 100% |
-
-
109-78-4
3-Hydroxypropionitrile

-
-
162611-38-3, 162611-39-4, 171081-09-7
O-(5'-O-(4,4'-dimethoxytrityl)-N6-benzoyl-2-deoxyadenosin-3'-yl) N,N-diisopropylfluorophosphoramidite

-
-
171081-13-3
Phosphorofluoridous acid (2R,3S,5R)-5-(6-benzoylamino-purin-9-yl)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-tetrahydro-furan-3-yl ester 2-cyano-ethyl ester

| Conditions | Yield |
|---|---|
| With 1H-tetrazole In acetonitrile for 0.166667h; Ambient temperature; | 100% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
109-78-4
3-Hydroxypropionitrile

-
-
30436-27-2
β-cyanoethyl chlorocarbonate

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 3-Hydroxypropionitrile In tetrahydrofuran at 0 - 20℃; Stage #2: With pyridine In tetrahydrofuran at 0 - 20℃; | 100% |
| In pyridine; toluene at 0℃; |
-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
109-78-4
3-Hydroxypropionitrile

-
-
89923-33-1
3-((tert-butyldimethylsilyl)oxy)propanenitrile

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 0 - 30℃; | 100% |
| With 1H-imidazole In tetrahydrofuran at 40℃; for 15h; Condensation; | 98% |
| With 1H-imidazole In dichloromethane at 20℃; | 94% |
-
-
13154-24-0
triisopropylsilyl chloride

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; | 100% |
-
-
474755-59-4
(1'S)-1-(1'-phenylethyl)-3,4-epoxypyrrolidine

-
-
1634-04-4
tert-butyl methyl ether

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; water | 100% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 1h; | 100% |
-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3-Hydroxypropionitrile With acetyl chloride In methanol; toluene at 0 - 20℃; for 18h; Stage #2: With ammonia In tetrahydrofuran; methanol at 0 - 20℃; for 18h; | 100% |
-
-
109-78-4
3-Hydroxypropionitrile

-
-
219553-44-3
1-<3-(N-tert-butoxycarbonylamino)propyl>-4-phenylpiperidine-4-carboxylic acid

-
-
219553-50-1
1-N-(tert-butoxycarbonyl)-3-<4-(2-cyanoethoxycarbonyl)-4-phenylpiperidin-1-yl>propylamine

| Conditions | Yield |
|---|---|
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane for 16h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With Iron(III) isopropoxide; zirconium(IV) tetraisopropoxide In n-heptane for 23h; | 99% |
| tetrachlorobis(tetrahydrofuran)hafnium(IV) In toluene for 24h; Heating; | 94% |
| zirconium(IV) oxychloride In n-heptane for 5h; Heating; |

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In tetrahydrofuran at 65℃; | 99% |
-
-
22097-10-5
5-methyl-1,2,3-thiadiazolyl-4-carboxylic acid

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In 1,2-dichloro-ethane at 0 - 20℃; for 3.5h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0℃; for 12h; Inert atmosphere; | 99% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
109-78-4
3-Hydroxypropionitrile

-
-
42202-44-8
3-trimethylsilanyloxypropionitrile

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether at 0 - 23℃; for 16.3333h; | 98% |
| Stage #1: 3-Hydroxypropionitrile With triethylamine at 0℃; Stage #2: chloro-trimethyl-silane In diethyl ether at 0℃; for 0.583333h; | 98.5% |
| With triethylamine In diethyl ether at 0 - 23℃; for 16h; Inert atmosphere; Schlenk technique; | 98% |
-
-
5394-63-8
2,2,6-trimethyl-4H-1,3-dioxin-4-one

-
-
109-78-4
3-Hydroxypropionitrile

-
-
65193-87-5
2-cyanoethylacetoacetate

| Conditions | Yield |
|---|---|
| In o-xylene at 141℃; for 2h; | 98.3% |
| In 5,5-dimethyl-1,3-cyclohexadiene at 140 - 145℃; for 1h; | 90% |
| In xylene at 140 - 145℃; for 1h; | 81.6% |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; water In isopropyl alcohol at 100℃; under 5171.62 Torr; for 0.833333h; | 98% |
| With CF3O3S(1-)*C36H35ClFeOP3Pt(1+); water; silver trifluoromethanesulfonate In tetrahydrofuran at 40℃; for 12h; Catalytic behavior; Inert atmosphere; | 92% |
| With silver tetrafluoroborate; C36H35ClFeOP3Pt(1+)*BF4(1-) In tetrahydrofuran; water at 80℃; for 12h; | 92% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; hydrogen; silver nitrate In methanol at 45 - 75℃; under 1500.15 Torr; Temperature; High pressure; Inert atmosphere; Autoclave; | 98% |
| With hydrogen; nickel In isopropyl alcohol at 60℃; under 3102.9 Torr; 20-24 h; | 88% |
| With ammonium hydroxide; hydrogen at 80℃; under 60006 Torr; for 6h; Sealed tube; | 80% |
-
-
109-78-4
3-Hydroxypropionitrile

-
-
15074-54-1
2-chlorophenyl dichlorophosphate

-
-
96404-97-6
2-chlorophenyl bis<2-cyanoethyl>phosphate

| Conditions | Yield |
|---|---|
| With pyridine In 1,4-dioxane for 4.5h; Ambient temperature; | 98% |
-
-
3898-66-6
2-(carboxymethyl)-5-nitrobenzoic acid

-
-
109-78-4
3-Hydroxypropionitrile

-
-
623172-50-9
2-(2-cyano-ethoxycarbonylmethyl)-5-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 70℃; for 0.116667h; | 98% |
-
-
10314-98-4
1-benzyloxycarbonylpiperidine-4-carboxylic acid

-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 5h; Ambient temperature; | 97% |
-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
109-78-4
3-Hydroxypropionitrile

-
-
42202-44-8
3-trimethylsilanyloxypropionitrile

| Conditions | Yield |
|---|---|
| With ammonium thiocyanate In dichloromethane at 0℃; for 0.166667h; | 97% |
| at 135℃; for 2h; | 167 g |
| toluene-4-sulfonic acid at 100℃; for 2.5h; | |
| toluene-4-sulfonic acid at 60 - 80℃; for 2.5 - 4.5h; |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 0℃; for 2h; | 97% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane; N,N-dimethyl-formamide Ambient temperature; | 88% |
-
-
109-78-4
3-Hydroxypropionitrile

-
-
475107-22-3
O-(2-thiono-1,3,2-oxathiaphospholanyl)-N-(tert-butoxycarbonyl)serine methyl ester

-
-
475107-34-7
2-tert-butoxycarbonylamino-3-[(2-cyano-ethoxy)-mercapto-phosphoryloxy]-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; for 3h; | 97% |
-
-
64-17-5
ethanol

-
-
109-78-4
3-Hydroxypropionitrile

-
-
20914-88-9
Ethyl 3-hydroxypropionimidate hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether at 20℃; for 3h; Pinner Imino Ether Synthesis; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| dirhodium tetraacetate In chloroform | 96% |
-
-
109-78-4
3-Hydroxypropionitrile

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20℃; for 3h; | 96% |
3-Hydroxypropionitrile Chemical Properties
Hydracrylonitrile(109-78-4)'s molecular formula is C3H5NO and its formula weight is 71.08.
The density of hydracrylonitrile(109-78-4) is 1.04 and it has a melting point of -46°C. The Boiling point is 228°C. The refractive index is about 1.4246-1.4266. Its flash point is 128°C. The water solubility should be equal to or greater than 10 g/100 mL at 20°C.
Hydracrylonitrile(109-78-4) has the property of being dissolved in water, ethanol, acetone and methyl ethyl ketone(MEK). It is not soluble in benzene, light petroleum, carbon bisulfide and Carbon tetrachloride.
The molecular structure of hydracrylonitrile(109-78-4) :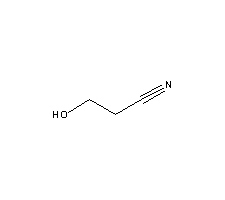
3-Hydroxypropionitrile Uses
3-Hydroxypropionitrile Toxicity Data With Reference
| 1. | skn-rbt 10 mg/24H open | JIHTAB Journal of Industrial Hygiene and Toxicology. 26 (1944),269. | ||
| 2. | skn-rbt 520 mg open MLD | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 8/18 ,1967. | ||
| 3. | eye-rbt 500 mg | AJOPAA American Journal of Ophthalmology. 29 (1946),1363. | ||
| 4. | orl-rat LD50:3200 mg/kg | 38MKAJ Pattys Industrial Hygiene and Toxicology. 2C (1982),4875. | ||
| 5. | orl-mus LD50:1800 mg/kg | AMIHBC AMA Archives of Industrial Hygiene and Occupational Medicine. 8 (1953),371. | ||
| 6. | ihl-mus LC33:300 mg/m3/2H | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow, USSR.: Centre of International Projects, GKNT,1982,66. | ||
| 7. | ipr-mus LD50:500 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 8. | orl-rbt LDLo:900 mg/kg | AMIHBC AMA Archives of Industrial Hygiene and Occupational Medicine. 8 (1953),371. | ||
| 9. | skn-rbt LD50:5000 mg/kg | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide |
3-Hydroxypropionitrile Consensus Reports
3-Hydroxypropionitrile Safety Profile
Related Products
- 3-Hydroxypropionitrile
- 109792-60-1
- 109794-97-0
- 109-79-5
- 109806-71-5
- 109-80-8
- 1098102-93-2
- 109814-50-8
- 109-81-9
- 109-82-0
- 109825-55-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View