-
Name
4'-Hydroxy-3'-methylacetophenone
- EINECS 212-880-5
- CAS No. 876-02-8
- Article Data36
- CAS DataBase
- Density 1.106 g/cm3
- Solubility
- Melting Point 107-109 °C(lit.)
- Formula C9H10O2
- Boiling Point 301.6 °C at 760 mmHg
- Molecular Weight 150.177
- Flash Point 127.3 °C
- Transport Information
- Appearance white to light beige crystalline powder
- Safety 36/37/39-26-22-37/39
- Risk Codes 36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Acetophenone,4'-hydroxy-3'-methyl- (7CI,8CI);1-(3-Methyl-4-hydroxyphenyl)ethanone;1-(4-Hydroxy-3-methylphenyl)ethanone;2-Methyl-4-acetylphenol;3'-Methyl-4'-hydroxyacetophenone;4-Acetyl-2-methylphenol;Ethanone,1-(4-hydroxy-3-methylphenyl)-;NSC 63365;4-Hydroxy-3-methylacetophenone;
- PSA 37.30000
- LogP 1.90320
Synthetic route

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide 1.) RT, 1 h, 2.) reflux, 12 h; | 96% |
| With sublimated iron chloride |
-
-
64-19-7
acetic acid

-
-
95-48-7
ortho-cresol

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

| Conditions | Yield |
|---|---|
| With aluminum oxide; methanesulfonic acid at 140℃; for 5h; | A 92% B 8% |
| With aluminum oxide; methanesulfonic acid at 140℃; for 0.0833333h; | A 92% B 8% |

-
-
533-18-6
2-methylphenyl acetate

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

| Conditions | Yield |
|---|---|
| With aluminum oxide; methanesulfonic acid at 160℃; for 0.166667h; | A 85% B 15% |
| With potassium carbonate In hexane at 25℃; for 12h; Irradiation; | A 12% B 74% |
| With zirconium(IV) chloride In dichloromethane for 8h; Ambient temperature; Irradiation; | A 32% B 47% |
| With aluminum (III) chloride at 120 - 180℃; for 2h; Fries Phenol Ester Rearrangement; |

| Conditions | Yield |
|---|---|
| Stage #1: acetic acid With trifluoromethylsulfonic anhydride at 20 - 60℃; Stage #2: ortho-cresol at 60℃; for 0.166667h; regiospecific reaction; | 83% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In nitromethane at 50℃; Fries rearrangement; | 76% |
| With N,N-dimethyl-formamide; zinc at 68℃; for 5h; Fries rearrangement; microwave irradiation; | 74% |
| With aluminum (III) chloride at 25 - 165℃; for 2h; | 28% |
-
-
43230-11-1
4'-amino-3'-methyl-acetophenone

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite durch Erhitzen der Diazoniumsalzloesung; |
-
-
103262-79-9
1-ethynyl-4-methyl-cyclohexa-2,5-diene-1,4-diol

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
95-48-7
ortho-cresol

-
-
75-36-5
acetyl chloride

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; nitrobenzene at 30℃; |


| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid 1.) room temperature, 1.5-3.5 h, 2.) 14 d; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| titanium tetrachloride | |
| Multi-step reaction with 2 steps 1: sulfuric acid / 0.5 h / 20 °C 2: aluminum (III) chloride / 2 h / 120 - 180 °C View Scheme |
-
-
10024-90-5
4-methoxy-3-methylacetophenone

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| With pyridine hydrochloride |
-
-
7446-70-0
aluminium trichloride

-
-
95-48-7
ortho-cresol

-
-
98-95-3
nitrobenzene

-
-
75-36-5
acetyl chloride

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

| Conditions | Yield |
|---|---|
| at 60℃; Product distribution; |

-
-
7550-45-0
titanium tetrachloride

-
-
95-48-7
ortho-cresol

-
-
98-95-3
nitrobenzene

-
-
75-36-5
acetyl chloride

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

| Conditions | Yield |
|---|---|
| at 60℃; Product distribution; |

-
-
7446-70-0
aluminium trichloride

-
-
533-18-6
2-methylphenyl acetate

-
-
98-95-3
nitrobenzene

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| at 20℃; |

-
-
7446-70-0
aluminium trichloride

-
-
533-18-6
2-methylphenyl acetate

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 160 - 180℃; Product distribution; |

-
-
533-18-6
2-methylphenyl acetate

-
-
7550-45-0
titanium tetrachloride

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 165℃; Product distribution; |

-
-
7446-70-0
aluminium trichloride

-
-
533-18-6
2-methylphenyl acetate

-
-
98-95-3
nitrobenzene

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 25℃; Product distribution; | |
| at 60℃; Product distribution; |

-
-
533-18-6
2-methylphenyl acetate

-
-
7550-45-0
titanium tetrachloride

-
-
98-95-3
nitrobenzene

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 30 - 60℃; Product distribution; |

-
-
533-18-6
2-methylphenyl acetate

-
-
7550-45-0
titanium tetrachloride

-
-
108-90-7
chlorobenzene

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 40℃; Product distribution; |

-
-
533-18-6
2-methylphenyl acetate

-
-
7550-45-0
titanium tetrachloride

-
-
79-34-5
1,1,2,2-tetrachloroethane

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 40℃; Product distribution; |

-
-
533-18-6
2-methylphenyl acetate

-
-
7550-45-0
titanium tetrachloride

-
-
71-43-2
benzene

-
A
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
B
-
699-91-2
2-acetyl-6-methylphenol

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| at 40℃; Product distribution; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 99 percent / triethylamine / CH2Cl2 / 20 °C 2: 76 percent / AlCl3 / nitromethane / 50 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: zinc chloride / man zersetzt das Reaktionsprodukt durch Kochen mit konz. Salzsaeure 2: NaNO2; diluted hydrochloric acid / durch Erhitzen der Diazoniumsalzloesung View Scheme |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride at 160℃; for 8h; |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
252561-07-2
4-acetyl-2-methylphenyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine at -40℃; for 0.25h; | 100% |
| With triethylamine In dichloromethane at 18 - 25℃; for 16h; | 85% |
| With triethylamine In dichloromethane at 20℃; for 16h; | 85% |
| With triethylamine In dichloromethane at 20℃; for 16h; | 85% |
| With 2,6-dimethylpyridine; dmap In dichloromethane at -78℃; |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
6315-52-2
1,2-bis-tosyloxyethane

-
-
94757-23-0
1,2-Bis-<4-acetyl-2-methyl-phenoxy>-aethan

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 100% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
100-39-0
benzyl bromide

-
-
56443-69-7
1-(4-(benzyloxy)-3-methylphenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 0℃; for 1h; Reflux; Inert atmosphere; | 100% |
| With sodium hydroxide; tetrabutylammomium bromide In dichloromethane at 20℃; | 99% |
| With potassium carbonate In acetone for 5h; Heating; | 97% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
105-36-2
ethyl bromoacetate

-
-
42018-09-7
ethyl 2-(4-acetyl-2-methylphenoxy)acetate

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 50℃; for 2h; | 100% |
| With potassium carbonate In acetonitrile at 20℃; for 8h; | |
| With caesium carbonate In acetonitrile at 20℃; | |
| With potassium carbonate In acetonitrile at 20℃; for 8h; | |
| With caesium carbonate In acetonitrile at 20℃; |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
96-32-2
bromoacetic acid methyl ester

-
-
166953-80-6
(4-acetyl-2-methylphenoxy)acetic acid methyl ester

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 20℃; for 16h; Williamson reaction; | 99% |
| With potassium carbonate In butanone for 1h; Heating / reflux; | 95% |
| With potassium carbonate In butanone for 1h; Reflux; | 95% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
456-41-7
1-(Bromomethyl)-3-fluorobenzene

-
-
758706-54-6
1-{4-[(3-fluorobenzyl)oxy]-3-methylphenyl}ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 80℃; for 1h; | 98% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
42018-09-7
ethyl 2-(4-acetyl-2-methylphenoxy)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 18h; | 98% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy-3-methylphenyl methyl ketone With potassium fluoride; potassium carbonate; N,N`-sulfuryldiimidazole; trifluoroacetic acid In water; acetonitrile Stage #2: In water; acetonitrile at 20℃; for 18h; | 97% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
74-88-4
methyl iodide

-
-
10024-90-5
4-methoxy-3-methylacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 5h; Heating; | 96% |
| With potassium carbonate In acetone | 82.4% |
| In water; N,N-dimethyl-formamide | 71% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
109-70-6
1,3-chlorobromopropane

-
-
111627-47-5
1-[4-(3-chloropropoxy)-3-methylphenyl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 24h; Heating; | 95% |
| With potassium carbonate In acetone | 35.8 g (95%) |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
85257-88-1
3-methyl-4-(tosyloxy)acetophenone

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 95% |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium(II) hydroxide/carbon In acetic acid at 25℃; under 3102.97 Torr; for 16h; | 93% |
| With 20 % Pd(OH)2/C; hydrogen In acetic acid under 3102.97 Torr; for 16h; | 93% |
| With molybdenum (IV) sulfide at 270℃; Hydrogenation; |

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
593-56-6
N-methoxylamine hydrochloride

-
-
147501-21-1
2-methyl-4-(1-methoxyimino-ethyl)phenol

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane | 93% |

-
-
1076-38-6
4-hydroxy[1]benzopyran-2-one

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
10111-08-7
2-imidazolecarbaldehyde

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 3.5h; Irradiation; | 92% |

-
-
4548-45-2
2-chloro-5-nitropyridine

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
758706-59-1
1-{3-methyl-4-[(5-nitropyridin-2-yl)oxy]phenyl}ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 89% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
32247-96-4
3,5-bis(trifluoromethyl)benzyl bromide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; acetonitrile for 5h; Heating; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy-3-methylphenyl methyl ketone With sodium hydride In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: rac-3-bromocyclohexene In N,N-dimethyl-d6-formamide at 20℃; | 88% |
| Stage #1: 4-hydroxy-3-methylphenyl methyl ketone With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.5h; Stage #2: rac-3-bromocyclohexene In N,N-dimethyl-formamide; mineral oil at 20℃; | 88% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
2910-75-0
1-(4-hydroxy-3-methylphenyl)-3-(4-hydroxyphenyl)-2-propen-1-one

| Conditions | Yield |
|---|---|
| With thionyl chloride In methanol for 2h; Claisen-Schmidt Condensation; | 88% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
62072-59-7
(rac)-4-(1-hydroxyethyl)-2-methylphenol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; cerium(III) chloride heptahydrate In methanol at 20℃; for 0.383333h; | 87% |
| With sodium hydroxide; ethanol; palladium Hydrogenation; |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| With 2,2'-azinobis(3-ethylbenzthiazolinesulfonate); laccase; potassium iodide In dimethyl sulfoxide at 20℃; for 15h; pH=5; Enzymatic reaction; | 87% |
| Stage #1: 4-hydroxy-3-methylphenyl methyl ketone With iodine; sodium acetate In methanol for 1h; Reflux; Stage #2: With sodium hydroxide In methanol; water for 1h; Reflux; | 86.8% |
| With iodine; iodic acid In ethanol; water at 35 - 40℃; for 1.5h; |
-
-
82294-70-0
4-methyl-1,3-thiazole-5-carbaldehyde

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
1086627-08-8
(E)-1-(4-hydroxy-3-methyIphenyI)-3-(4-methylthiazoI-5-yl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-methyl-1,3-thiazole-5-carbaldehyde; 4-hydroxy-3-methylphenyl methyl ketone With sodium hydroxide In ethanol at 0 - 20℃; for 18h; Stage #2: With hydrogenchloride; water In ethanol pH=4 - 5; | 87% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
619-66-9
4-Carboxybenzaldehyde

-
-
934826-21-8
(E)-4-(3-(3-(4-hydroxy-3-methylphenyl)-3-oxoprop-1-enyl)phenoxy)acetic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy-3-methylphenyl methyl ketone; 4-Carboxybenzaldehyde With potassium hydroxide In methanol; water at 0℃; Stage #2: With hydrogenchloride In methanol; water pH=4 - 5; | 87% |
| With potassium hydroxide In methanol; water Cooling with ice; |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

| Conditions | Yield |
|---|---|
| 86% |
-
-
50-00-0
formaldehyd

-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
506-59-2
N,N-dimethylammonium chloride

-
-
1076690-73-7
3-dimethylamino-1-(4-hydroxy-3-methyl-phenyl)-propan-1-one hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol at 95℃; for 18h; | 84% |


| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In toluene at 60℃; for 2h; Schlenk technique; Inert atmosphere; | 83% |
-
-
876-02-8
4-hydroxy-3-methylphenyl methyl ketone

-
-
584-08-7
potassium carbonate

-
-
74-88-4
methyl iodide

-
-
10024-90-5
4-methoxy-3-methylacetophenone

| Conditions | Yield |
|---|---|
| In acetone | 82.4% |
| In acetone | 82.4% |
| In acetone | 82.4% |
| In acetone | 82.4% |

4'-Hydroxy-3'-methylacetophenone Chemical Properties
Molecular Structure of 4-Hydroxy-3-methylacetophenone (CAS NO.876-02-8):
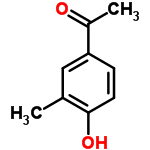
Molecular formula: C9H10O2
Molecular Weight: 150.17
EINECS: 212-880-5
Melting point: 107-109 °C(lit.)
Boiling Point: 301.6 °C at 760 mmHg
Flash Point: 127.3 °C
Index of Refraction: 1.546
Molar Refractivity: 42.98 cm3
Molar Volume: 135.6 cm3
Polarizability: 17.04 10-24 cm3
Surface Tension: 41.8 dyne/cm
Density: 1.106 g/cm3
Enthalpy of Vaporization: 56.34 kJ/mol
Vapour Pressure: 0.000585 mmHg at 25°C
BRN: 2041839
Physical Appearance: white to light beige crystalline powder
Product Categories: Aromatic Acetophenones & Derivatives (substituted);Benzene series;Benzene derivatives;C9;Carbonyl Compounds;Ketones
IUPAC Name: 1-(4-hydroxy-3-methylphenyl)ethanone
Synonyms: Ethanone, 1-(4-hydroxy-3-methylphenyl)-;4-ACETYL-2-METHYLPHENOL;4-HYDROXY-3-METHYLACETOPHENONE;1-(4-HYDROXY-3-METHYLPHENYL)ETHANONE;3'-Methyl-4'-Hydroxy Acetophenone;4'-Hydroxy-3'-methylacetophenone, 99.5+%;1-(4-HYDROXY-3-METHYLPHENYL)ETHAN-1-ONE;4'-HYDROXY-3'-METHYLACETOPHENONE;
4'-Hydroxy-3'-methylacetophenone Safety Profile
Hazard Codes:  Xi,
Xi, Xn
Xn
Risk Statements: 36/37/38-22
R36/37/38:Irritating to eyes, respiratory system and skin.
R22:Harmful if swallowed.
Safety Statements: 36/37/39-26-22-37/39
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S22:Do not breathe dust.
S37/39:Wear suitable gloves and eye/face protection.
WGK Germany: 3
Hazard Note: Irritant
HazardClass: IRRITANT
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View