-
Name
4-Bromobenzoic acid
- EINECS 209-581-7
- CAS No. 586-76-5
- Article Data548
- CAS DataBase
- Density 1.701 g/cm3
- Solubility soluble in hot water
- Melting Point 252-254 °C(lit.)
- Formula C7H5BrO2
- Boiling Point 299.8 °C at 760 mmHg
- Molecular Weight 201.019
- Flash Point 135.1 °C
- Transport Information
- Appearance White to light yellow crystal powder
- Safety 26-36-24/25-22-37/39
- Risk Codes 22-36/37/38-36-37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms p-Carboxybromobenzene;4-Bromobenzene carboxylic acid;p-Bromobenzoicacid;p-Bromobenzenecarboxylic acid;Benzoic acid, 4-bromo-;4-09-00-01017 (Beilstein Handbook Reference);Benzoic acid, p-bromo-;p-Bromobenzoic acid;
- PSA 37.30000
- LogP 2.14730
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium chromate(VI); sulfuric acid | 100% |
| With tert.-butylhydroperoxide; water at 20℃; for 14h; Inert atmosphere; | 99% |
| With sulfuric acid; ozone; acetic acid; manganese(II) acetate at 16℃; for 1.25h; Product distribution / selectivity; | 98% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; potassium hydroxide In toluene for 22h; Product distribution; Ambient temperature; | 100% |
| Stage #1: 4-methoxycarbonylphenyl bromide With sodium hydroxide In tetrahydrofuran; water at 20℃; Stage #2: With hydrogenchloride In water at 0℃; pH=2; | 100% |
| With iodine; aluminium In acetonitrile at 80℃; for 18h; | 99% |
-
-
113445-97-9
1-isocyanato-1-phenyl-2,2,2-trifluoroethyl 4-bromobenzoate

-
-
106-54-7
p-Chlorothiophenol

-
A
-
586-76-5
4-Bromobenzoic acid

-
B
-
118364-61-7
1-phenyl-1-(4-chlorophenylthio)-2,2,2-trifluoroethyl isocyanate

| Conditions | Yield |
|---|---|
| In benzene 1.) 20 deg C, 12 h, 2.) reflux, 3 h; | A 100% B 87% |


| Conditions | Yield |
|---|---|
| With amphiphilic resin-supported phosphine-palladium; water; potassium carbonate at 25℃; under 760 Torr; for 12h; hydroxycarbonylation; | 100% |
| With water; potassium carbonate In acetonitrile at 100℃; under 3750.38 Torr; for 0.0161111h; | 93% |
| With water; palladium diacetate; potassium carbonate at 20℃; under 760.051 Torr; for 6h; | 91% |
-
-
99-90-1
para-bromoacetophenone

-
-
98-80-6
phenylboronic acid

-
A
-
92-91-1
biphenyl-4-acetaldehyde

-
B
-
586-76-5
4-Bromobenzoic acid

-
C
-
92-92-2
biphenyl-4-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: para-bromoacetophenone; phenylboronic acid; immobilized 4-bromobenzoic acid With potassium carbonate In water; N,N-dimethyl-formamide Stage #2: With potassium hydroxide In ethanol; water | A 100% B 19% C 81% |


| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran at -78℃; for 0.5h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; di-tert-butyl peroxide; silica-supported selenamide In water; tert-butyl alcohol at 75℃; for 7h; | 99% |
| With 4H3N*4H(1+)*CuMo6O18(OH)6(4-); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 12h; | 99% |
| With sheep hemoglobin; oxygen In water at 20℃; under 760.051 Torr; for 150h; pH=7.0; Catalytic behavior; Darkness; Green chemistry; Enzymatic reaction; chemoselective reaction; | 99% |
-
-
24523-45-3, 41097-38-5
bis-(4-bromo-benzylidene)-hydrazine

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With poly(diselanediyl-1,2-phenylene); dihydrogen peroxide In tetrahydrofuran for 20h; Heating; | 99% |
| With [bis(acetoxy)iodo]benzene In tetrahydrofuran at 20℃; for 4h; Green chemistry; | 80% |
-
-
19350-68-6
p-bromobenzaldehyde tosylhydrazone

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With silica-supported selenamide; dihydrogen peroxide In tetrahydrofuran at 55℃; for 42h; | 99% |
| With poly(diselanediyl-1,2-phenylene); dihydrogen peroxide In tetrahydrofuran for 10h; Heating; | 90% |
-
-
124-38-9
carbon dioxide

-
-
183677-71-6
2-(4-bromophenyl)-5,5-dimethyl-1,3,2-dioxaborinane

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; copper(l) chloride; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride In tetrahydrofuran at 70℃; under 760.051 Torr; for 24h; | 99% |
| With (1,3-bis(2,6-diisopropyl-4-(morpholinomethyl)phenyl)imidazolidin-2-ylidene)copper(I) bromide; potassium tert-butylate In tetrahydrofuran under 760.051 Torr; for 24h; Inert atmosphere; Schlenk technique; Reflux; Green chemistry; | 94% |
| Stage #1: carbon dioxide; 2-(4-bromophenyl)-5,5-dimethyl-1,3,2-dioxaborinane With copper(l) iodide; 5,5-bis(4,5-dihydrooxazol-2-yl)nonane; cesium fluoride In N,N-dimethyl-formamide at 90℃; under 760.051 Torr; for 10h; Stage #2: With hydrogenchloride; water In N,N-dimethyl-formamide | 86% |
| Stage #1: carbon dioxide; 2-(4-bromophenyl)-5,5-dimethyl-1,3,2-dioxaborinane With potassium tert-butylate; silver(I) acetate; triphenylphosphine In 1,4-dioxane at 100℃; under 15201 Torr; for 8h; Inert atmosphere; Autoclave; Stage #2: With hydrogenchloride In tetrahydrofuran; water Inert atmosphere; | 71% |

| Conditions | Yield |
|---|---|
| With silica-supported Jones reagent In dichloromethane for 0.035h; | 98.3% |
| With C30H24AgBr4N8(1+)*AgBr2(1-); potassium hydroxide In 1,2-dimethoxyethane at 60℃; for 24h; Molecular sieve; Schlenk technique; | 98% |
| With diethylene glycol dimethyl ether at 70℃; for 0.5h; Sonication; | 98% |
-
-
25062-46-8, 40979-16-6, 34158-73-1
4-bromobenzaldehyde oxime

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With poly(diselanediyl-1,2-phenylene); dihydrogen peroxide In tetrahydrofuran for 28h; Heating; | 98% |
| With silica-supported selenamide; dihydrogen peroxide In tetrahydrofuran at 55℃; for 37h; | 96% |
| With [hydroxy(tosyloxy)iodo]benzene; water In dimethyl sulfoxide at 20℃; for 24h; | 89% |

| Conditions | Yield |
|---|---|
| With Oxone In water; acetonitrile for 15h; Reflux; | 98% |
| With sodium periodate; sulfuric acid In water at 95℃; for 12h; | 81% |
| With tert.-butylhydroperoxide at 100℃; for 5h; | 75% |
| With tert.-butylhydroperoxide In water at 80℃; for 12h; Green chemistry; |

| Conditions | Yield |
|---|---|
| Stage #1: carbon dioxide; (4-bromophenyl)zinc iodide With lithium chloride In N,N-dimethyl-formamide at 50℃; under 760.051 Torr; for 48h; Stage #2: With hydrogenchloride; water In N,N-dimethyl-formamide | 98% |

| Conditions | Yield |
|---|---|
| With 40% potassium fluoride/alumina for 0.0666667h; Microwave irradiation; Neat (no solvent); | A 97% B 91% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-4-ethenyl-benzene With tert.-butylhydroperoxide; iron(III) chloride hexahydrate; sodium hydroxide In water at 80℃; for 10h; Stage #2: With hydrogenchloride In water at 20℃; | 96% |
| With Oxone In water; acetonitrile for 12h; Reflux; | 90% |
| With sodium periodate; 2C16H13N2O10Ru2(1-)*Ca(2+) In water at 45℃; for 16h; Green chemistry; | 90% |
-
-
92093-23-7
1-(4-bromophenyl)ethane-1,2-diol

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; potassium tert-butylate In water at 80℃; for 8h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: para-bromoacetophenone With tert.-butylhydroperoxide; sodium hydroxide; tungsten(VI) oxide In water at 80℃; for 8h; Stage #2: With hydrogenchloride In water | 95% |
| Stage #1: para-bromoacetophenone With iodine; dimethyl sulfoxide In chlorobenzene at 130℃; for 3h; Stage #2: With tert.-butylhydroperoxide In chlorobenzene at 20 - 130℃; for 5h; | 89% |
| With Oxone; trifluoroacetic acid In 1,4-dioxane for 10h; Reflux; Green chemistry; | 85% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(ll) bromide In acetic anhydride | A 3% B 95% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; oxygen In 1,2-dimethoxyethane at 70℃; for 3h; | 95% |
| Multi-step reaction with 3 steps 1: alcohol / Sonnenlicht 2: acetyl chloride / 100 °C 3: alcoholic KOH-solution View Scheme |
-
-
21757-86-8
1-(4-bromophenyl)-2,2,2-trichloroethan-1-ol

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dihydrogen peroxide In methanol for 10h; | 95% |

| Conditions | Yield |
|---|---|
| With acetic acid In titanium | A n/a B 95% |

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium hydrogen sulfate; water at 60 - 65℃; for 2.2h; Green chemistry; | 95% |
-
-
1616506-43-4
(Z)-3-chloro-3-(4-bromophenyl)acrylic acid

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 80℃; for 0.5h; | 94% |

| Conditions | Yield |
|---|---|
| With Amberlyst A-26 (OH-); dihydrogen peroxide In 1,4-dioxane at 20℃; for 10h; | 93% |
| With Oxone In water; acetonitrile for 24h; Reflux; | 86% |
-
-
589-87-7
1,4-bromoiodobenzene

-
-
18414-58-9
diphenylmethylsilanecarboxylic acid

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With potassium trimethylsilonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; bis(dibenzylideneacetone)-palladium(0) In toluene at 40℃; for 0.333333h; | 93% |
-
-
67-56-1
methanol

-
-
1122-91-4
4-bromo-benzaldehyde

-
A
-
586-76-5
4-Bromobenzoic acid

-
B
-
619-42-1
4-methoxycarbonylphenyl bromide

| Conditions | Yield |
|---|---|
| With C12H22N6(2+)*2C2F6NO4S2(1-); 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 55℃; for 16h; Reagent/catalyst; Molecular sieve; | A 7% B 93% |
| With dihydrogen peroxide; calcium chloride at 65℃; for 48h; Reagent/catalyst; | A n/a B 81% |
| With 3,3'-dimethyl-1,1'-(hexane-1,6-diyl)diimidazolium glutarate; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 55℃; for 16h; Reagent/catalyst; Temperature; Molecular sieve; | A 11% B 78% |
| With dihydrogen peroxide at 20℃; for 8h; |


| Conditions | Yield |
|---|---|
| With aluminium trichloride; dimethylsulfide for 16h; Ambient temperature; | 92.5% |
| Stage #1: Ethyl 4-bromobenzoate With iron(III) chloride In tetrachloromethane at 85℃; for 24h; Inert atmosphere; Stage #2: With hydrogenchloride; water In tetrachloromethane Inert atmosphere; | 92% |
| Stage #1: Ethyl 4-bromobenzoate With sodium hydroxide In water at 20℃; Stage #2: With hydrogenchloride In water |

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-benzaldehyde With potassium hydroxide for 0.0833333h; Cannizzaro Reaction; Milling; Inert atmosphere; Sealed tube; Green chemistry; Stage #2: With hydrogenchloride In water Green chemistry; | A 92% B 92% |
| With sodium hydroxide In water at 15℃; for 3h; Cannizzaro Reaction; | A 90% B n/a |
| With [(hydrotris(3,5-diphenylpyrazole-1-yl)borate)-FeII(benzilate)]; oxygen In benzene at 20℃; Inert atmosphere; | A 85% B 40% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1-ethynylbenzene With tert.-butylhydroperoxide; iron(III) chloride hexahydrate; sodium hydroxide In water at 80℃; for 10h; Stage #2: With hydrogenchloride In water at 20℃; | 92% |
| With Oxone; trifluoroacetic acid In 1,4-dioxane for 10h; Reflux; Green chemistry; | 89% |
| With methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate; nickel dibromide at 120℃; under 760.051 Torr; for 48h; | 79% |

| Conditions | Yield |
|---|---|
| With pyridine; sodium hydrogencarbonate; 4-chloro-3-nitro-chromen-2-one In N,N-dimethyl-formamide at 18 - 60℃; for 0.3h; | 100% |
| With potassium carbonate In dichloromethane at 20℃; for 0.416667h; | 80% |
| Stage #1: 4-Bromobenzoic acid With Bromotrichloromethane; triphenylphosphine In dichloromethane for 5h; Reflux; Stage #2: With triethylamine In dichloromethane for 8h; Reflux; | 79% |

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane for 18h; Heating; | 100% |
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane for 1h; Product distribution / selectivity; | 100% |
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 2h; Reflux; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

-
-
16419-60-6
2-Methylphenylboronic acid

-
-
5748-43-6
4-(2-methylphenyl)benzoic acid

| Conditions | Yield |
|---|---|
| With disodium hydrogenphosphate; disodium bis(2-aminopyrimidine-4,6-diolate)palladium acetate In water at 50 - 120℃; for 0.166667h; Microwave irradiation; | 100% |
| With disodium hydrogenphosphate; Pd(OAc)2(2-amino-4,6-dihydroxypyrimidine disodium salt)2 In water at 120℃; for 0.166667h; Suzuki-Miyaura coupling; Microwave irradiation; | 99% |
| With 4C3H6N6*Pd(2+)*2C2H3O2(1-); sodium carbonate In water at 80℃; for 2h; Suzuki-Miyaura Coupling; Green chemistry; | 97% |
-
-
75-84-3
2,2-dimethyl-propanol-1

-
-
586-76-5
4-Bromobenzoic acid

-
-
104110-51-2
4-Bromo-benzoic acid 2,2-dimethyl-propyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In toluene | 100% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; Pd-complex on fluorous reversed silica gel In water at 80℃; for 16h; Suzuki reaction; | 100% |
| With potassium carbonate; carbapalladacycle complex*periodic mesoporous organosilica for 24h; Suzuki coupling; Heating; | 100% |
| With sodium phosphate; palladium on activated charcoal In water; isopropyl alcohol at 20℃; for 3.5h; Suzuki-Miyaura coupling reaction; | 100% |

| Conditions | Yield |
|---|---|
| (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In 1,4-dioxane for 2h; Heating; | 100% |
-
-
889893-47-4
(R)-4-hydroxymethyl-2-phenyl-4,5-dihydrooxazol-4-ylmethyl acetate

-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 1h; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

-
-
17933-03-8
m-tolylboronic acid

-
-
5728-33-6
3'‐methyl‐(1,1'‐biphenyl)‐4‐carboxylic acid

| Conditions | Yield |
|---|---|
| With disodium hydrogenphosphate; disodium bis(2-aminopyrimidine-4,6-diolate)palladium acetate In water at 50 - 120℃; for 0.166667h; Microwave irradiation; | 100% |
| With 4C3H6N6*Pd(2+)*2C2H3O2(1-); sodium carbonate In water at 80℃; for 2h; Suzuki-Miyaura Coupling; Green chemistry; | 99% |
| Stage #1: 4-Bromobenzoic acid; m-tolylboronic acid; palladium diacetate In water at 20℃; for 0.0833333h; Suzuki-Miyaura coupling; Stage #2: With potassium hydroxide In water at 20 - 75℃; for 2h; | 90% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,4-dioxane; water at 105℃; for 5h; Suzuki Coupling; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 20h; | 100% |
| Stage #1: 4-Bromobenzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; for 0.166667h; Stage #2: morpholine In N,N-dimethyl-formamide at 20℃; for 18h; | 99% |
| Stage #1: 4-Bromobenzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 0.5h; Stage #2: morpholine With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 2h; | |
| Stage #1: 4-Bromobenzoic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 0 - 25℃; for 1h; Stage #2: morpholine With triethylamine In dichloromethane at 0 - 25℃; for 16h; |
-
-
586-76-5
4-Bromobenzoic acid

-
-
146651-75-4
tert–butyl (2–aminophenyl)carbamate

-
-
617703-02-3
N-(2-t-butoxycarbonylaminophenyl)-4-bromobenzamide

| Conditions | Yield |
|---|---|
| With 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride In N,N-dimethyl-formamide at 20℃; for 20h; | 100% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol for 16h; Reflux; | 100% |
| With sulfuric acid In methanol | 97% |
| With sulfuric acid; sodium carbonate | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-Bromobenzoic acid With 4-(Methylamino)pyridine; diisopropyl-carbodiimide In dichloromethane at 20℃; Stage #2: With hydroxylamine In dichloromethane; water at 20℃; Further stages.; | 100% |
| Stage #1: 4-Bromobenzoic acid With 1,1'-carbonyldiimidazole In tetrahydrofuran at 23℃; for 1h; Inert atmosphere; Stage #2: In tetrahydrofuran at 23℃; for 12h; Inert atmosphere; | 85% |
| Stage #1: 4-Bromobenzoic acid With 4-methyl-morpholine; 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide In ethyl acetate; acetonitrile at 0℃; for 0.25h; Stage #2: With hydroxylamine hydrochloride In ethyl acetate; acetonitrile Ultrasonication; | 83% |

| Conditions | Yield |
|---|---|
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane at 20℃; for 36h; | 100% |
-
-
1154061-23-0
N1-tert-butyl-5-((tert-butyldimethylsilyloxy)methyl)benzene-1,2-disulfonamide

-
-
586-76-5
4-Bromobenzoic acid

-
-
1154061-22-9
4-bromo-N-(4-((tert-butyldimethylsilyloxy)methyl)-2-(N-tert-butylsulfamoyl)phenylsulfonyl)benzamide

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

-
-
1154061-22-9
4-bromo-N-(4-((tert-butyldimethylsilyloxy)methyl)-2-(N-tert-butylsulfamoyl)phenylsulfonyl)benzamide

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

-
-
867333-30-0
4-(4-bromobenzoyl)piperazine-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; for 18h; | 100% |
| With N-ethyl-N,N-diisopropylamine; HATU In N,N-dimethyl-formamide at 20℃; for 15h; | 94% |
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 18h; | |
| Stage #1: 4-Bromobenzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: 1-t-Butoxycarbonylpiperazine In N,N-dimethyl-formamide at 20℃; |
-
-
586-76-5
4-Bromobenzoic acid

-
-
177911-87-4
tert-butyl 2-(aminomethyl)-pyrrolidine-1-carboxylate

-
-
1008505-56-3
2-(4-bromobenzoylaminomethyl)pyrrolidine-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; HATU In N,N-dimethyl-formamide for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In tetrahydrofuran at 60℃; Inert atmosphere; | 100% |
| With 1-ethyl-3-(3-dimethyl-aminopropyl) carbodiimide; benzotriazol-1-ol; triethylamine In dichloromethane at 20℃; | 83% |
| Stage #1: 4-Bromobenzoic acid With chloroformic acid ethyl ester; triethylamine In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: 1-amino-2-propene In dichloromethane Inert atmosphere; | 38% |
| Stage #1: 4-Bromobenzoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.166667h; Inert atmosphere; Stage #2: 1-amino-2-propene In dichloromethane at 0 - 20℃; Inert atmosphere; |
-
-
38041-19-9
4-aminotetrahydropyran

-
-
586-76-5
4-Bromobenzoic acid

-
-
1153383-64-2
4-bromo-N-(tetrahydropyran-4-yl)benzamide

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide at 20℃; for 16h; Cooling with ice; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; HATU In N,N-dimethyl-formamide at 20℃; for 48h; | 100% |
-
-
586-76-5
4-Bromobenzoic acid

-
-
57260-73-8
N-BOC-1,2-diaminoethane

-
-
1008505-59-6
tert-butyl (2-(4-bromobenzamido)ethyl)carbamate

| Conditions | Yield |
|---|---|
| With oxalyl dichloride In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 99.6% |
| With sulfuric acid for 12h; Heating; | 98% |
| With sulfuric acid for 4h; Fischer esterification; Reflux; | 98% |

| Conditions | Yield |
|---|---|
| With fluorosulfonyl fluoride; N-ethyl-N,N-diisopropylamine In 1,2-dichloro-ethane at 20℃; for 5h; | 99% |
| With sulfuric acid Reflux; | 98% |
| With sulfuric acid for 3h; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; tetra(n-butyl)ammonium hydroxide; 1,10-phenanthroline-4,7-diol In water at 110℃; for 24h; Inert atmosphere; Schlenk technique; Sealed tube; Green chemistry; | 99% |
| Stage #1: 4-Bromobenzoic acid With copper(l) iodide; cesium hydroxide; 5-bromo-2-(1H-imidazol-2-yl)pyridine In water; dimethyl sulfoxide at 100℃; for 36h; Inert atmosphere; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide pH=1 - 2; Inert atmosphere; | 91% |
| With copper(I) oxide; 2-pyridinealdoxime; cesium hydroxide; tetrabutylammomium bromide In water at 110℃; for 48h; Inert atmosphere; | 87% |
4-BROMOBENZOIC ACID Chemical Properties
MF: C7H5BrO2
MW: 201.02
EINECS: 209-581-7
4-BROMOBENZOIC ACID,its melting point is about 252-254 °C(lit.);density is 1,894 g/cm3 ,soluble in hot water.Appearance is white to light yellow crystal powder.4-BROMOBENZOIC ACID is soluble in ethanol, ethyl ether, acetone and hot water.
Following is the molecular structure of 4-BROMOBENZOIC ACID:
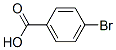
4-BROMOBENZOIC ACID Uses
4-BROMOBENZOIC ACID Toxicity Data With Reference
| 1. | orl-mus LD50:1059 mg/kg | GISAAA Gigiena i Sanitariya. 44 (12)(1979),19. | ||
| 2. | ipr-mus LD50:536 mg/kg | GISAAA Gigiena i Sanitariya. 44 (12)(1979),19. |
RTECS : DG4448050
4-BROMOBENZOIC ACID Consensus Reports
4-BROMOBENZOIC ACID Safety Profile
Hazard Codes : Xn,Xi (Harmful,Irritant)
Risk Statements :
R22:Harmful if swallowed
R36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements :
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S36:Wear suitable protective clothing
S24/25: Avoid contact with skin and eyes
S22:Do not breathe dust
S37/39 :Wear suitable gloves and eye/face protection
4-BROMOBENZOIC ACID Specification
HANDLING and STORAGE about 4-BROMOBENZOIC ACID:
Handling:
Wash thoroughly after handling.
Remove contaminated clothing and wash before reuse.
Minimize dust generation and accumulation.
Avoid ingestion and inhalation.
Avoid contact with eyes, skin, and clothing.
Keep container tightly closed.Use with adequate ventilation.
Storage:Store in a tightly closed container. Store in a cool, dry,well-ventilated area away from incompatible substances.
Related Products
- 4-BROMOBENZOIC ACID
- 586-77-6
- 586-78-7
- 5868-05-3
- 58682-75-0
- 58684-54-1
- 5868-54-2
- 586-89-0
- 58690-81-6
- 586-91-4
- 58691-88-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View