-
Name
4-NITROBENZENEDIAZONIUM TETRAFLUOROBORATE
- EINECS 207-261-1
- CAS No. 456-27-9
- Article Data70
- CAS DataBase
- Density g/cm3
-
Solubility
Stability
- Stable. Incompatible with strong oxidizing agents, moisture, strong bases.Avoid mechanical shock.
Toxicology
- Harmful if swallowed, inhaled or absorbed through the skin.Chronic exposure may caus
-
Melting Point
144-148 °C (dec.)
- Formula C6H4N3O2•BF4
- Boiling Point °Cat760mmHg
- Molecular Weight 236.921
- Flash Point °C
- Transport Information
- Appearance solid
- Safety Mutation data reported. When heated to decomposition it emits toxic vapors of boron, NOx, and F−.
- Risk Codes 20/21/22-36/37/38-34
-
Molecular Structure
- Hazard Symbols Xn,Xi,C
- Synonyms 4-Nitrobenzenediazonium Tetrafluoroborate 98%;p-NITRO-PHENYLDIAZONIUM FLUOBORATE;BENZENEDIAZONIUM, p-NITRO-, TETRAFLUORO-BORATE(1-);BENZENEDIAZONIUM, 4-NITRO-, TETRA-FLUOROBORATE(1-) (9CI);4-Nitrobenzenediazonium tetrafluoroborate;100-05-0;154960-09-5;Benzenediazonium, p-nitro-, tetrafluoroborate(1-);120600-85-3;14368-49-1;15003-70-0;Benzenediazonium, 4-nitro-;p-Nitrophenyldiazonium fluoborate;p-Nitrobenzenediazonium tetrafluoroborate;Benzenediazonium, 4-nitro-, tetrafluoroborate(1-);4-nitrobenzenediazonium; tetrafluoroboron;p-Nitrophenyl diazonium fluoroborate;4-Nitrobenzenediazonium tetrafluoroborate(1-);p-Nitrobenzenediazonium fluoborate;154960-10-8;4-Nitrophenyldiazonium tetrafluoroborate;Benzenediazonium, 4-nitro-, tetrafluoroborate (1-);4-Nitrobenzenediazonium fluoroborate;198975-25-6;Benzenediazonium, p-nitro-, tetrafluoroborate (1-);p-Nitrobenzenediazonium fluoroborate;p-Nitrophenyldiazonium tetrafluoroborate;15151-17-4;
- PSA 73.97000
- LogP 3.90258
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-aniline With hydrogenchloride; sodium nitrite In water at 0℃; Stage #2: With sodium tetrafluoroborate In water | 97% |
| With tetrafluoroboric acid; sodium nitrite In water Diazotization; | 96% |
| Stage #1: 4-nitro-aniline With boron trifluoride diethyl etherate In acetonitrile Cooling with ice; Stage #2: With isopentyl nitrite In acetonitrile at 0℃; for 0.5h; | 93% |

-
-
540-80-7
tert.-butylnitrite

-
-
100-01-6
4-nitro-aniline

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In ethanol; water at 0 - 20℃; for 1h; | 94% |
| In ethanol at 0 - 20℃; for 1h; |


| Conditions | Yield |
|---|---|
| With sodium nitrite In water at 0℃; for 0.666667h; | 87% |
| Stage #1: tetrafluoroboric acid; 4-nitro-aniline In water at 20℃; for 0.0333333h; Stage #2: With sodium nitrite In water at 0℃; for 0.25h; | 79% |
| With tert.-butylnitrite In ethanol; water at 0 - 20℃; for 1h; | 71% |

-
-
100-01-6
4-nitro-aniline

-
-
7632-00-0
sodium nitrite

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In water at 0 - 5℃; for 0.5h; | 80% |
| In water at 0℃; for 0.5h; | 72% |
| In water at 0℃; for 0.75h; | 66% |


-
-
100-01-6
4-nitro-aniline

-
-
7632-00-0
sodium nitrite

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-aniline With hydrogenchloride In water for 0.25h; Cooling with ice; Stage #2: sodium nitrite In water for 0.5h; Cooling with ice; Stage #3: lithium tetrafluoroborate In water | 71% |

-
-
74317-27-4
18-crown-6/p-nitrobenzenediazonium tetrafluoroborate complex

-
A
-
17455-13-9
18-crown-6 ether

-
B
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In methanol at 25℃; Equilibrium constant; |

-
A
-
17455-13-9
18-crown-6 ether

-
B
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In CHCl2F at -80.1℃; Thermodynamic data; rate constant; ΔGc(excit.); |

-
A
-
32743-85-4
1-hydroxy-4-sulfonatonaphthalene

-
B
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With aq. buffer at 20℃; Equilibrium constant; pH 2-4; |

-
A
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
B
-
68384-00-9
2-hydroxy-6,8-disulfonatonaphthalene

| Conditions | Yield |
|---|---|
| With aq. buffer at 20℃; Equilibrium constant; pH 2-4; |

-
A
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
B
-
83277-67-2
1-hydroxy-3,6-disulfonatonaphthalene

| Conditions | Yield |
|---|---|
| With aq. buffer at 20℃; Equilibrium constant; pH 2-4; |

-
A
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at -30 - 25℃; |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite at -15 - 5℃; for 1h; |

| Conditions | Yield |
|---|---|
| With sodium nitrite In water at 0℃; for 0.666667h; |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In tetrahydrofuran |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
100-01-6
4-nitro-aniline

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In methanol at -20 - 20℃; Inert atmosphere; | |
| With tert.-butylnitrite In methanol at -20 - 20℃; Schlenk technique; Inert atmosphere; |
-
-
67969-82-8
tetrafluoroboric acid diethyl ether

-
-
16020-14-7
(E)-1-methoxy-2-(4-nitrophenyl)diazene

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at 20℃; for 0.1h; | 100 %Spectr. |
-
-
32352-96-8
4-nitro-benzenediazohydroxide, sodium salt

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: water 2: diethyl ether / 1 h / 0 °C 3: [D3]acetonitrile / 0.1 h / 20 °C View Scheme |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diethyl ether / 1 h / 0 °C 2: [D3]acetonitrile / 0.1 h / 20 °C View Scheme |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
135-19-3
β-naphthol

-
-
93449-57-1
1,2-Naphthochinon-1-<(4-nitrophenyl)hydrazon>

| Conditions | Yield |
|---|---|
| for 48h; ultrasound irradiation; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
370839-59-1
(E)-tert-butyl 3-(4-nitrophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,6,11-triazacyclopentadeca-3,8,12-triene Pd In ethanol at 20℃; for 2.75h; Heck reaction; | 100% |
| With 3-benzyl-1-(2-hydroxy-2-phenylethyl)imidazolium chloride; palladium diacetate In ethanol at 36℃; for 3h; Heck-Matsuda reaction; | 96% |
| With dichloro bis(acetonitrile) palladium(II) In water at 0 - 25℃; for 1.5h; | 96% |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
140-88-5
ethyl acrylate

-
-
24393-61-1
ethyl (E)-3-(4-nitrophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| palladium diacetate In ethanol; water at 20℃; for 0.5h; Heck arylation; | 100% |
| With 3-benzyl-1-(2-hydroxy-2-phenylethyl)imidazolium chloride; palladium diacetate In ethanol at 36℃; for 3h; Heck-Matsuda reaction; | 99% |
| With 1,6,11-triazacyclopentadeca-3,8,12-triene Pd In ethanol at 20℃; for 0.75h; Heck reaction; | 95% |
| With palladium(II) carboxymethylcellulose In water at 20℃; for 8h; | 76% |

| Conditions | Yield |
|---|---|
| Stage #1: C12H15NO; 4-nitrobenzenediazonium tetrafluoroborate With sulfuric acid In methanol; water Stage #2: With ammonia In methanol; water | 100% |
-
-
37171-41-8
4-ethyl-3,4-dihydro-2H-benzo[b][1,4]oxazin-6-ol

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
1132773-52-4
4-ethyl-7-((4-nitrophenyl)diazenyl)-3,4-dihydro-2H-benzo[b][1,4]oxazin-6-ol

| Conditions | Yield |
|---|---|
| Stage #1: 4-ethyl-3,4-dihydro-2H-benzo[b][1,4]oxazin-6-ol; 4-nitrobenzenediazonium tetrafluoroborate With sulfuric acid In methanol; water Stage #2: With ammonia In methanol; water | 100% |
| With sulfuric acid In methanol; water at 20℃; for 0.5h; | 82% |
-
-
1415743-31-5
3-(7-methoxy-3,4-dihydro-2H-quinolin-1-yl)propane-1-sulfonic acid 2,2,2-trifluoro-1-p-tolyl-ethyl ester

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
1415743-33-7
3-[7-methoxy-6-(4-nitrophenylazo)-3,4-dihydro-2H-quinolin-1-yl]propane-1-sulfonic acid 2,2,2-trifluoro-1-p-tolyl-ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol; water at 20℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With palladium diacetate In methanol at 20℃; for 12h; Inert atmosphere; | 100% |
-
-
55023-87-5
2-tert-Butyl-2H-isoindol

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
73286-85-8
2-tert-Butyl-1-(4-nitrophenylhydrazono)-1H-isoindolium-tetrafluoroborat

| Conditions | Yield |
|---|---|
| In acetone at 0℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| With iodine; dibenzyl hantzsch ester In dimethyl sulfoxide at 20℃; | 99% |
| With trimethylsilyl iodide; methyl iodide In N,N-dimethyl-formamide for 1h; Ambient temperature; | 91% |
| With iodine; potassium iodide In dimethyl sulfoxide at 15℃; |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
50-81-7
ascorbic acid

-
-
121654-33-9
3-O-p-nitrobenzenediazoascorbic acid

| Conditions | Yield |
|---|---|
| In water; acetonitrile for 0.5h; | 99% |

| Conditions | Yield |
|---|---|
| With dibenzo-18-crown-6; potassium bromide; copper(ll) bromide; 1,10-Phenanthroline; copper(I) bromide In acetonitrile at 20℃; for 0.333333h; Sandmeyer bromination; | 99% |
| With N-Bromosuccinimide; tetrabutylammonium perchlorate In methanol; N,N-dimethyl-formamide at 20℃; for 1h; Sandmeyer Reaction; Electrochemical reaction; | 84% |
| With 1,3-bis-n-butylimidazolium bromide at 80℃; for 0.5h; | 71% |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With Pd2(dba)4; sodium acetate In acetonitrile at 20℃; for 12h; Inert atmosphere; | 99% |
| With sodium acetate; bis(dibenzylideneacetone)-palladium(0) In acetonitrile for 12h; Inert atmosphere; | 99% |
-
-
55991-65-6
[1-(4-methoxyphenyl)vinyloxy]trimethylsilane

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
20765-22-4
1-(4-methoxyphenyl)-2-(4-nitrophenyl)ethanone

| Conditions | Yield |
|---|---|
| With dibenzyl hantzsch ester In dimethyl sulfoxide | 99% |

| Conditions | Yield |
|---|---|
| With trimethylamine at 20℃; under 375.038 Torr; for 12h; | 98% |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
98-80-6
phenylboronic acid

-
-
92-93-3
1-phenyl-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With aluminium hydroxide-supported palladium nanoparticles In methanol at 20℃; for 6h; Suzuki-Miyaura cross-coupling; | 97% |
| polystyrene-supported N-heterocyclic carbene-Pd complex In ethanol at 20℃; for 8h; Suzuki-Miyaura cross-coupling; | 96% |
| With polystyrene-supported-diiodobis(theophylline)palladium(II) complex In methanol at 25℃; for 2h; Suzuki-Miyaura Coupling; | 96% |
-
-
292638-84-7
styrene

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
1694-20-8
trans-4-nitrostilbene

| Conditions | Yield |
|---|---|
| [(1,3-dimesitylimidazol-2-ylidene)(naphthoquinone)Pd(0)]2 In methanol at 65℃; for 2h; Heck reaction; | 97% |
| With 9-N-methylamino-1-oxophenalene; potassium tert-butylate In dimethyl sulfoxide at 25℃; for 24h; Meerwein Arylation; Inert atmosphere; Glovebox; | 45% |
-
-
2396-80-7
methyl 5-hexenoate

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
A
-
344556-61-2
(E)-methyl-6-(4-nitrophenyl)hex-5-enoate

-
B
-
1310810-23-1
C19H18N2O6

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0) In N,N-dimethyl acetamide at 20℃; for 0.333333h; Heck reaction; optical yield given as %de; stereoselective reaction; | A 97% B n/a |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
140-88-5
ethyl acrylate

-
-
953-26-4
ethyl 4-nitrocinnamate

| Conditions | Yield |
|---|---|
| With polystyrene resin supported theophylline carbene PdI2 complex In ethanol at 25℃; for 0.66h; Reagent/catalyst; Green chemistry; | 97% |
| In water at 40℃; for 4h; | 87% |
-
-
1075-49-6
4-ethenylbenzoic acid

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With tris(2,2'-bipyridyl)ruthenium dichloride; water at 20℃; for 4h; Meerwein Arylation; Irradiation; | 97% |

-
-
141-32-2
acrylic acid n-butyl ester

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
131061-15-9, 86622-84-6
3-(4-nitrophenyl)acrylic acid n-butyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 130℃; for 6h; Heck Reaction; | 97% |
| With titanium dioxide cellulose composite supported palladium nanoparticle In water at 20℃; for 3h; | 92% |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran Ambient temperature; | 96% |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
35629-70-0
2-amino-4-methyloxazole

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 0 - 10℃; for 0.25h; | 96% |
-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
100-61-8
N-methylaniline

-
A
-
53464-31-6
1-(4-nitrophenyl)-3,3'-methylphenyltriazene

-
B
-
31464-31-0
N-methyl-4-nitro-4'-aminoazobenzene

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride In acetonitrile | A 96% B 4% |
| With tetrabutyl-ammonium chloride In acetonitrile | A 43% B 56% |


| Conditions | Yield |
|---|---|
| [(1,3-dimesitylimidazol-2-ylidene)(naphthoquinone)Pd(0)]2 In methanol at 50℃; for 1h; Heck reaction; | 96% |
| (IMes)Pd(NQ) In ethanol at 50℃; for 1h; Conversion of starting material; Heck Reaction; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; for 2.25h; pH=8; | 96% |
| With sodium hydroxide In water at 0℃; for 0.75h; | 96% |
-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
2143-90-0
4-methoxy-4'-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With 10 wtpercent palladium nanoparticles decorated on Montmorillonite K10 In water at 20℃; for 0.75h; Suzuki-Miyaura Coupling; | 96% |
| With Palladium Nanoparticles Supported on Soil-Derived Humic Acid Coated Iron-Oxide Nanoparticles In methanol at 30℃; for 12h; Suzuki Coupling; | 91% |
| With 5% Pd/BaCO3 In methanol at 25℃; for 12h; | 90% |
-
-
90-01-7
salicylic alcohol

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
155850-91-2
2-hydroxymethyl-4-(4'-nitrophenylazo)phenol

| Conditions | Yield |
|---|---|
| Stage #1: salicylic alcohol; 4-nitrobenzenediazonium tetrafluoroborate With sodium hydroxide In water at 0 - 20℃; for 2.25h; pH=8; Stage #2: With hydrogenchloride In water at -5℃; for 1h; | 96% |

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With 1% Pd/C In methanol at 50℃; for 0.5h; Suzuki-Miyaura reaction; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon In methanol Suzuki-Miyaura cross-coupling; Reflux; Under air; | 96% |
4-Nitrobenzenediazonium tetrafluoroborate Chemical Properties
IUPAC Name: Difluoroboranyl-fluoro-(4-nitrophenyl)iminoazanium fluoride
Synonyms: Msc-0141 ; 4-Nitrobenzenesiazonium tetrafluoroborate ; 4-Nitrobenzenediazonium tetrafluoroborate ; P-nitrobenzenediazonium fluoroborate ; P-nitrobenzenediazonium tetrafluoroborate
Product Categories: Organic-metal salt;Aromatics Compounds;B (Classes of Boron Compounds);Tetrafluoroborates;Aromatics
CAS NO: 456-27-9
Molecular Formula: C6H4BF4N3O2
Molecular Weight: 236.92
Molecular Structure: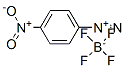
EINECS: 207-261-1
Molecular Structure:
Mol File: 456-27-9.mol
Melting point: 144-148 °C (dec.)
Storage temp: 2-8°C
Appearance: 4-Nitrobenzenediazonium fluoborate (CAS NO. 456-27-9) is pale yellow solid.
Stability: 4-Nitrobenzenediazonium fluoborate (CAS NO. 456-27-9) is stable,incompatible with strong oxidizing agents, moisture, strong bases. Avoid mechanical shock.
4-Nitrobenzenediazonium tetrafluoroborate Uses
4-Nitrobenzenediazonium fluoborate (CAS NO. 456-27-9) is used for tissue staining and determination of trace phenols.
4-Nitrobenzenediazonium tetrafluoroborate Toxicity Data With Reference
| 1. | mmo-sat 40 µg/plate | FCTOD7 Food and Chemical Toxicology. 25 (1987),669. |
4-Nitrobenzenediazonium tetrafluoroborate Consensus Reports
Reported in EPA TSCA Inventory.
4-Nitrobenzenediazonium tetrafluoroborate Safety Profile
Hazard Codes  Xi Xn,
Xi Xn, C
C
Risk Statements 20/21/22-36/37/38-34
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R34:Causes burns.
Safety Statements 22-24/25-45-36/37/39-26
S24/25:Avoid contact with skin and eyes.
S22:Do not breathe dust.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
RIDADR UN 1759 8/PG 2
WGK Germany 3
RTECS CZ1723000
F 4.3
Hazard Note Irritant
HazardClass 8
PackingGroup III
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View