-
Name
4-Phenoxyphenol
- EINECS 212-611-1
- CAS No. 831-82-3
- Article Data67
- CAS DataBase
- Density 1.175 g/cm3
- Solubility insoluble in water, solubel in benzene, diethyl ether
- Melting Point 80-84 °C(lit.)
- Formula C12H10O2
- Boiling Point 313.761 °C at 760 mmHg
- Molecular Weight 186.21
- Flash Point 141.446 °C
- Transport Information
- Appearance beige crystalline powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Phenol,p-phenoxy- (6CI,7CI,8CI);4-Hydroxydiphenyl ether;Hydroquinone monophenyl ether;NSC25027;p-Hydroxydiphenyl ether;p-Phenoxyphenol;
- PSA 29.46000
- LogP 3.18450
Synthetic route
-
-
51067-38-0
4-phenoxyphenylboronic acid

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With [Rh2(bpy)2(μ-OAc)2(OAc)2]; oxygen; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide under 760.051 Torr; for 18h; Irradiation; | 97% |
| With dihydrogen peroxide In ethanol at 20℃; for 0.166667h; | 89% |
| With oxygen; triethylamine In 2-methyltetrahydrofuran at 20℃; under 760.051 Torr; for 24h; Green chemistry; | 73% |
| With tert.-butylhydroperoxide; oxygen; trichloroacetonitrile In decane; acetonitrile at 20℃; for 12h; Irradiation; | 57% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 24h; Catalytic behavior; | 92% |
| With sodium hydroxide In neat (no solvent) at 90℃; for 9h; Green chemistry; | 60% |

| Conditions | Yield |
|---|---|
| With methanol; copper(II) ferrite at 20℃; for 24h; | 86% |

| Conditions | Yield |
|---|---|
| With 1,3-dimethyl-2-imidazolidinone; lithium diisopropyl amide In tetrahydrofuran; n-heptane; ethylbenzene at 185℃; for 12h; further reagent: NaN(SiMe3)2; | 84% |
| With hydrogen iodide | |
| With aluminium trichloride at 130 - 150℃; |

| Conditions | Yield |
|---|---|
| With zinc In N,N-dimethyl-formamide for 0.0416667h; Williamson ether synthesis; microwave irradiation; | 81% |

| Conditions | Yield |
|---|---|
| With zinc In N,N-dimethyl-formamide for 0.0583333h; Williamson ether synthesis; microwave irradiation; | 79% |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; copper(l) chloride In water at 100℃; for 16h; | 73.3% |
| Multi-step reaction with 2 steps 1: potassium methoxide, copper(I) chloride, copper bronze / 6 h / 230 °C 2: boron tribromide / CH2Cl2 / 4 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: 71 percent / K2CO3 / N,N-dimethyl-acetamide / Heating 2: 3-chloroperoxybenzoic acid / CHCl3 / Ambient temperature 3: conc. HCl / methanol / 1 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2′,4′,6′-triisopropyl-3,6-dimethoxy-[1,1′-biphenyl]-2-yl)phosphine; boric acid; palladium diacetate; caesium carbonate In 1-methyl-pyrrolidin-2-one at 80℃; for 24h; Schlenk technique; Inert atmosphere; | 67% |
| With potassium hydroxide; copper at 250℃; | |
| With alkaline solution; copper at 200℃; |

| Conditions | Yield |
|---|---|
| With trans-di(μ-acetato)bis[o-(di-o-tolyl-phosphino)benzyl]dipalladium(II); C29H45Pt; potassium carbonate In water; N,N-dimethyl-formamide at 115℃; for 0.5h; Inert atmosphere; Microwave irradiation; | 67% |

| Conditions | Yield |
|---|---|
| With hydrogen In Hexadecane at 260℃; under 7600.51 Torr; for 5h; Autoclave; | A 8.3% B 9.5% C 53.8% |


| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium tert-butylate In dimethyl sulfoxide at 100℃; for 24h; | 53% |

| Conditions | Yield |
|---|---|
| With hydrogen In Hexadecane at 260℃; under 7600.51 Torr; for 5h; Autoclave; | A 5.5% B 13.2% |

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 130℃; |

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With diethyl ether; dihydrogen peroxide |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrate | |
| With hydrogenchloride; tetrafluoroboric acid; acetic acid; sodium nitrite In water at 80℃; for 4h; Inert atmosphere; Cooling with ice; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; copper at 180 - 200℃; |
-
-
19082-52-1
4-phenoxyphenyl acetate

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol Heating; Yield given; | |
| With hydrogenchloride; methanol; water at 40℃; for 4h; |
-
-
406700-77-4
Formic acid 4-phenoxy-phenyl ester

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 1h; Ambient temperature; Yield given; |
-
-
2122-46-5, 55748-05-5
phenoxy radical

-
A
-
2417-10-9
2-Phenoxyphenol

-
B
-
1806-29-7
2,2'-dihydroxybiphenyl

-
C
-
92-88-6
4,4'-Dihydroxybiphenyl

-
D
-
611-62-1
2,4'-dihydroxybiphenyl

-
E
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| Product distribution; Irradiation; effect of abs. dose of irrad.; |


| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20 - 25℃; for 6h; Irradiation; | 0.95 g |

| Conditions | Yield |
|---|---|
| In hexane Rate constant; or in MeOH; |

-
-
108-95-2
phenol

-
A
-
2417-10-9
2-Phenoxyphenol

-
B
-
1806-29-7
2,2'-dihydroxybiphenyl

-
C
-
92-88-6
4,4'-Dihydroxybiphenyl

-
D
-
611-62-1
2,4'-dihydroxybiphenyl

-
E
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With di-tert-butyl diperoxyoxalate; di-tert-butyl peroxide In chlorobenzene Product distribution; Mechanism; a) 140 deg C or b) 25 deg C.; correlation of product distribution and stereoelectronic factors; |

-
-
108-95-2
phenol

-
A
-
1806-29-7
2,2'-dihydroxybiphenyl

-
B
-
92-88-6
4,4'-Dihydroxybiphenyl

-
C
-
611-62-1
2,4'-dihydroxybiphenyl

-
D
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide at 140℃; for 24h; Further byproducts given; | |
| With dihydrogen peroxide; horseradish peroxidase In phosphate buffer at 25℃; pH=7; Kinetics; Polymerization; |


| Conditions | Yield |
|---|---|
| With hydrogen iodide; acetic acid |

| Conditions | Yield |
|---|---|
| In water under 1E-06 Torr; Product distribution; Mechanism; Irradiation; ion-molecule reaction; |
-
-
39064-93-2
4-(4-(tert-butyl)phenoxy)phenol

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With aluminium trichloride; nitromethane |
-
-
13808-13-4
4-Phenoxy-2,4,6-tri-tert-butyl-cyclohexa-2,5-dien-1-on

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With aluminium trichloride; nitromethane |
-
-
108-95-2
phenol

-
A
-
2417-10-9
2-Phenoxyphenol

-
B
-
1806-29-7
2,2'-dihydroxybiphenyl

-
C
-
92-88-6
4,4'-Dihydroxybiphenyl

-
D
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With phosphate buffer; dihydrogen peroxide; horseradish peroxidase at 20℃; for 2h; oxidative coupling; Further byproducts given; |

-
-
108-95-2
phenol

-
A
-
2417-10-9
2-Phenoxyphenol

-
B
-
1806-29-7
2,2'-dihydroxybiphenyl

-
C
-
10181-94-9
((p-Phenoxy)-4-phenoxy)phenol

-
D
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With phosphate buffer; dihydrogen peroxide; horseradish peroxidase at 20℃; for 2h; oxidative coupling; Further byproducts given; |

-
-
831-82-3
4-Phenoxyphenol

-
-
124-63-0
methanesulfonyl chloride

-
-
23419-81-0
4-phenoxyphenyl methanesulfonate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane Inert atmosphere; Glovebox; | 100% |
-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With fluorosulfonyl fluoride; triethylamine In 1,4-dioxane at 20℃; for 24h; | 100% |
| With [(4-acetamidophenyl)(fluorosulfonyl)amino]sulfonyl fluoride; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran at 20℃; for 0.166667h; | 90% |
-
-
831-82-3
4-Phenoxyphenol

-
-
503-60-6
3,3-dimethyl-allyl chloride

-
-
42873-58-5
3-methylbut-2-en-1-yl 4-phenoxyphenyl ether

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide Ambient temperature; | 99% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
831-82-3
4-Phenoxyphenol

-
-
214690-17-2
4-(phenoxy)phenyl chloroformate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| In pyridine for 1.16667h; Cooling with ice; | 98% |
| With sodium hydroxide |

-
-
831-82-3
4-Phenoxyphenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether | 98% |

| Conditions | Yield |
|---|---|
| at 0 - 80℃; for 26280h; Product distribution / selectivity; | 98% |

| Conditions | Yield |
|---|---|
| at 0 - 50℃; for 26280h; Product distribution / selectivity; | 98% |

| Conditions | Yield |
|---|---|
| at 0 - 80℃; for 26280h; Product distribution / selectivity; | 98% |
-
-
831-82-3
4-Phenoxyphenol

-
-
54582-59-1
2-chloro-4-phenoxyphenol

| Conditions | Yield |
|---|---|
| With diisobutylamine; sulfuryl dichloride In toluene at 70℃; for 1h; | 97% |
| With N-chloro-succinimide In benzene for 48h; Heating; | 46% |
| With sodium hypochlorite | |
| With sulfuryl dichloride at 60℃; |

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 16h; | 97% |
| With pyridine for 16h; Ambient temperature; | 87% |
| With triethylamine In dichloromethane at 0 - 10℃; |
-
-
831-82-3
4-Phenoxyphenol

-
-
96-32-2
bromoacetic acid methyl ester

-
-
787575-61-5
(4-phenoxyphenoxy)acetic acid methyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; for 12h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In acetone for 14h; Reflux; | 96% |
| With potassium hydroxide In dimethyl sulfoxide Ambient temperature; | 93% |
| With potassium carbonate In acetone | |
| With potassium carbonate; potassium iodide In acetone at 60℃; for 18h; |
-
-
831-82-3
4-Phenoxyphenol

-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
16166-58-8
Trimethyl-(4-phenoxy-phenoxy)-silane

| Conditions | Yield |
|---|---|
| at 20℃; for 1.5h; Neat (no solvent); Air atmosphere; | 96% |
-
-
766-11-0
5-bromo-2-fluoropyridine

-
-
831-82-3
4-Phenoxyphenol

-
-
904961-14-4
5-Bromo-2-(4-phenoxy-phenoxy)-pyridine

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 160℃; for 0.5h; Product distribution / selectivity; Microwave irradiation; | 95.6% |
-
-
831-82-3
4-Phenoxyphenol

-
-
57057-80-4
ethyl (S)-2-[[(4-methylphenyl)sulfonyl]oxy]propanoate

-
-
100325-83-5
(R)-ethyl 2-(4-phenoxyphenoxy)propionate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 240h; Ambient temperature; | 95% |
-
-
831-82-3
4-Phenoxyphenol

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
144150-76-5
4-phenoxyphenyl 4-methylbenzenesulfonate

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; for 4h; | 95% |
| With sodium hydride In tetrahydrofuran Ambient temperature; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl acetamide for 2h; Heating; | 95% |
| With potassium carbonate In N,N-dimethyl acetamide at 170℃; for 6h; Inert atmosphere; | 73% |
-
-
831-82-3
4-Phenoxyphenol

-
-
115314-14-2
(2s)-(+)-glycidyl 3-nitrobenzenesulfonate

-
-
250778-96-2
(2 S)-2-[(4-phenoxyphenoxy)methyl]oxirane

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide; mineral oil | 95% |
| With sodium hydride In N,N-dimethyl-formamide at 20℃; |
-
-
624-28-2
2,5-dibromopyridine

-
-
831-82-3
4-Phenoxyphenol

-
-
904961-14-4
5-Bromo-2-(4-phenoxy-phenoxy)-pyridine

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 200℃; for 0.5h; Product distribution / selectivity; Microwave irradiation; | 95% |
-
-
831-82-3
4-Phenoxyphenol

-
-
127-00-4
1-Chloro-2-propanol

-
-
57650-78-9
(R,S)-2-hydroxypropyl 4-phenoxyphenyl ether

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; toluene at 85℃; for 0.166667h; Temperature; Reagent/catalyst; | 95% |
-
-
591-50-4
iodobenzene

-
-
201230-82-2
carbon monoxide

-
-
831-82-3
4-Phenoxyphenol

-
-
80202-05-7
4-phenoxyphenyl benzoate

| Conditions | Yield |
|---|---|
| With dicarbonyl(acetylacotonato)rhodium(I); triethylamine; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In toluene at 120℃; for 48h; Inert atmosphere; Autoclave; Schlenk technique; | 95% |
| With palladium diacetate; potassium carbonate at 100℃; under 5250.53 Torr; for 3h; Catalytic behavior; | 91 %Chromat. |

-
-
78632-23-2
3-ureidomethyl-3,5,5-trimethylcyclohexyl urea

-
-
831-82-3
4-Phenoxyphenol

-
-
1266555-43-4
C36H38N2O6

| Conditions | Yield |
|---|---|
| at 0 - 80℃; for 26280h; Product distribution / selectivity; | 94.1% |

| Conditions | Yield |
|---|---|
| at 0 - 80℃; for 26280h; Product distribution / selectivity; | 94.1% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 17h; | 94% |
| With potassium tert-butylate In tetrahydrofuran at 20℃; for 18h; |
-
-
882055-34-7
1-(2-bromo-1,3-benzothiazol-6-yl)ethanone

-
-
831-82-3
4-Phenoxyphenol

-
-
946661-97-8
1-[2-(4-phenoxyphenoxy)-1,3-benzothiazol-6-yl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 130℃; for 3h; | 94% |
-
-
17739-45-6, 59146-56-4
2-(2-bromoethoxy)tetrahydropyran

-
-
831-82-3
4-Phenoxyphenol

-
-
126301-65-3
2-(4-phenoxyphenoxy)-ethyl-tetrahydropyranyl-ether

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 23℃; for 16h; | 94% |
-
-
831-82-3
4-Phenoxyphenol

-
-
145279-04-5
2-nitro-4-phenoxyphenol

| Conditions | Yield |
|---|---|
| With nitric acid In tert-butyl methyl ether; water at 25℃; for 1.75h; | 93.3% |
| With ammonium nitrate; silica-gel-supported sulfuric acid In dichloromethane at 20℃; for 1.33333h; regioselective reaction; | 87% |
| With aluminum(III) nitrate nonahydrate; sulfuric acid; silica gel; silica gel In dichloromethane at 20℃; for 0.333333h; | 72% |
4-Phenoxyphenol Specification
4-Phenoxyphenol, with the CAS registry number 831-82-3, is also named as Hydroquinone monophenyl ether. It belongs to the product categories of Aromatic Phenols; Phenoles and thiophenoles; Building Blocks; C9 to C20+; Chemical Synthesis; Organic Building Blocks; Oxygen Compounds; Phenols. Its EINECS number is 212-611-1. This chemical's molecular formula is C12H10O2 and molecular weight is 186.21. What's more, its systematic name is 4-Phenoxyphenol. This chemical is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from strong oxidants. It is used as an intermediate for pesticide.
Physical properties of 4-Phenoxyphenol are: (1)ACD/LogP: 3.35; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.35; (4)ACD/LogD (pH 7.4): 3.35; (5)ACD/BCF (pH 5.5): 207.01; (6)ACD/BCF (pH 7.4): 206.57; (7)ACD/KOC (pH 5.5): 1582.66; (8)ACD/KOC (pH 7.4): 1579.33; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 29.46 Å2; (13)Index of Refraction: 1.605; (14)Molar Refractivity: 54.578 cm3; (15)Molar Volume: 158.45 cm3; (16)Polarizability: 21.636×10-24cm3; (17)Surface Tension: 46.3 dyne/cm; (18)Density: 1.175 g/cm3; (19)Flash Point: 141.446 °C; (20)Enthalpy of Vaporization: 57.698 kJ/mol; (21)Boiling Point: 313.761 °C at 760 mmHg; (22)Vapour Pressure: 0 mmHg at 25°C.
Preparation: this chemical can be prepared by 1-methoxy-4-phenoxy-benzene at the temperature of 185 °C. This reaction will need reagents lithium diisopropylamide, 1,3-dimethyl-2-imidazolidinone and solvents heptane, tetrahydrofuran, ethylbenzene with the reaction time of 12 hours. The yield is about 84%.
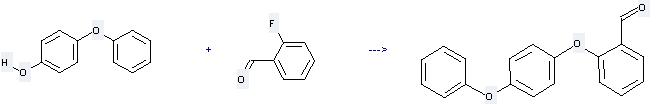
Uses of 4-Phenoxyphenol: it can be used to produce 2-(4-phenoxy-phenoxy)-benzaldehyde by heating. It will need reagent K2CO3 and solvent N,N-dimethyl-acetamide with the reaction time of 2 hours. The yield is about 95%.

When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O(c1ccccc1)c2ccc(O)cc2
(2)Std. InChI: InChI=1S/C12H10O2/c13-10-6-8-12(9-7-10)14-11-4-2-1-3-5-11/h1-9,13H
(3)Std. InChIKey: ZSBDGXGICLIJGD-UHFFFAOYSA-N
Related Products
- 4-Phenoxyphenol
- 831-91-4
- 83192-69-2
- 83199-35-3
- 83199-47-7
- 83200-09-3
- 832-01-9
- 83207-58-3
- 832080-38-3
- 832-09-7
- 832113-94-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View