-
Name
5-Hexen-1-ol
- EINECS 212-477-4
- CAS No. 821-41-0
- Article Data103
- CAS DataBase
- Density 0.833 g/cm3
- Solubility Miscible with water.
- Melting Point <-20 °C
- Formula C6H12O
- Boiling Point 149.9 °C at 760 mmHg
- Molecular Weight 100.161
- Flash Point 47.2 °C
- Transport Information UN 1987 3/PG 3
- Appearance clear colourless liquid
- Safety 16
- Risk Codes 10
-
Molecular Structure
-
Hazard Symbols
 F
F
- Synonyms 1-Hexen-6-ol;5-Hexene-1-ol;6-Hydroxy-1-hexene;5-Hexenol;
- PSA 20.23000
- LogP 1.33500
Synthetic route
-
-
43216-12-2
2-iodomethyltetrahydropyran

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With indium In methanol for 4.5h; sonication; | 95% |
| With dimethylboron bromide; tetra-(n-butyl)ammonium iodide; triethylamine In dichloromethane at 0℃; for 0.5h; | 82% |
-
-
127102-60-7
(S)-4-(but-3-enyl)-2,2-dimethyl-1,3-dioxolane

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With water; acetic acid | 95% |

| Conditions | Yield |
|---|---|
| With dimethylamine borane In ethanol at 25℃; for 0.5h; Reagent/catalyst; Time; | 95% |
| With palladium on activated charcoal; hydrogen In water at 25℃; under 760.051 Torr; for 1.5h; | 68% |
| With quinoline; hydrogen In methanol; water at 20℃; under 760.051 Torr; for 0.433333h; chemoselective reaction; | |
| With hydrogen In ethanol at 20℃; under 760.051 Torr; for 3.4h; Schlenk technique; Sealed tube; chemoselective reaction; | > 99 %Chromat. |
| With Dimethylphenylsilane; water In dimethyl sulfoxide at 70℃; for 0.333333h; Inert atmosphere; stereoselective reaction; |
-
-
54653-25-7
ethyl hex-5-enoate

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether | 93% |

| Conditions | Yield |
|---|---|
| With rhenium(VII) oxide; triphenylphosphine at 165℃; for 1h; Reagent/catalyst; Concentration; Temperature; | 91% |
| With rac-3-octanol at 170℃; for 1h; | 38% |

| Conditions | Yield |
|---|---|
| at 700℃; for 2h; | 87% |
| With tert-butoxide; 18-crown-6 ether In tetrahydrofuran for 1.85h; Heating; | 72% |
-
-
34723-82-5, 130233-15-7, 130233-16-8
2-(bromomethyl)tetrahydro-2H-pyran

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-(bromomethyl)tetrahydro-2H-pyran With sodium In diethyl ether for 2h; Metallation; Heating; Stage #2: With water Hydrolysis; | 87% |
| With sodium | 76% |
| With Na; pyridinium p-toluenesulfonate 1.) MeOH; 2.) CDCl3; Multistep reaction; |

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In dichloromethane; water for 18h; Ambient temperature; | 85% |
-
-
18420-41-2, 130233-13-5, 130233-14-6
2-(chloromethyl)tetrahydropyran

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With sodium In diethyl ether for 16h; | 83.5% |
| With diethyl ether; sodium |
-
-
77022-44-7
6-(tetrahydropyran-2-yloxy)hex-1-ene

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In methanol for 4h; Ambient temperature; | 82% |
| With ammonium chloride at 20℃; for 0.166667h; Yield given; |
-
-
1315481-26-5
2,6-diphenylphenyl hex-5-enyl ether

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-diphenylphenyl hex-5-enyl ether With sodium In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; Stage #2: With water In tetrahydrofuran at 0 - 20℃; Inert atmosphere; regioselective reaction; | 81% |
-
-
49639-25-0
phenyl(6-hexanol)sulfoxide

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| In various solvent(s) for 0.0166667h; Irradiation; | 80% |
-
-
207795-48-0
1-((hex-5-en-1-yloxy)methyl)-4-methoxybenzene

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With montmorillonite K 10 supported ammonium nitrate In neat (no solvent) for 0.05h; Irradiation; | 80% |

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With rose bengal; triethylamine In ethanol at 25℃; for 12h; Schlenk technique; Irradiation; | 78% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; copper(II) acetate monohydrate; ammonium bromide; lithium chloride In water; N,N-dimethyl-formamide at 0℃; for 12h; Electrochemical reaction; | 73% |

| Conditions | Yield |
|---|---|
| With 1-hexadecylcarboxylic acid at 330 - 350℃; under 760 Torr; for 24h; | 71% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride | 71% |
| With hydrogen In aq. phosphate buffer at 50℃; under 1500.15 Torr; for 48h; pH=7; Autoclave; | |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; |
-
-
63974-05-0
(E)-hex-2-ene-1,6-diol

-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane for 120h; Heating; | 70% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; copper(II) acetate monohydrate; ammonium bromide; lithium chloride In water; N,N-dimethyl-formamide at 0℃; for 12h; Electrochemical reaction; | 70% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -100 - 20℃; | 69% |
-
-
86692-87-7
hex-5-enyl hydroperoxide

-
A
-
821-41-0
5-Hexen-1-ol

-
B
-
6032-29-7
(+/-)-2-pentanol

-
C
-
764-59-0
hex-5-en-1-al

| Conditions | Yield |
|---|---|
| In pentane for 48h; Irradiation; | A 60% B 10% C 30% |

-
-
86692-87-7
hex-5-enyl hydroperoxide

-
A
-
821-41-0
5-Hexen-1-ol

-
B
-
764-59-0
hex-5-en-1-al

-
C
-
5048-26-0
5-hexenyl acetate

| Conditions | Yield |
|---|---|
| With titanium(III) chloride; acetic acid In water for 2h; | A 56% B 27% C 17% |


| Conditions | Yield |
|---|---|
| With sodium In tetrahydrofuran; ammonia at -78 - -33℃; Cyclization; Bouveault-Blanc reduction; | A 36% B 54% |

| Conditions | Yield |
|---|---|
| With ethanol; [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](borohydride)(carbonyl)(hydride)iron(II) at 100℃; for 24h; Inert atmosphere; Sealed tube; Darkness; | 53% |
-
-
629-03-8
1 ,6-dibromohexane

-
A
-
592-41-6
1-hexene

-
B
-
821-41-0
5-Hexen-1-ol

-
C
-
110-54-3
hexane

-
D
-
592-42-7
1,5-Hexadien

| Conditions | Yield |
|---|---|
| With tetramethylammonium perchlorate In water-d2; N,N-dimethyl-formamide Solvent; Electrolysis; Overall yield = 90 %; | A 22% B 9% C 50% D 16% |
| With tetramethylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; | A 22% B 7% C 39% D 18% |

-
-
27925-22-0
6,8-dioxa-bicyclo[3.2.1]oct-2-ene

-
A
-
821-41-0
5-Hexen-1-ol

-
B
-
63974-05-0
(E)-hex-2-ene-1,6-diol

-
C
-
6126-50-7, 928-91-6, 928-92-7
hex-4-en-1-ol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane for 150h; Heating; | A 23% B 5% C 46% |
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane for 150h; Product distribution; Heating; also with various other allylic ethers and allylic alcohols; | A 23% B 5% C 46% |

-
-
86692-87-7
hex-5-enyl hydroperoxide

-
A
-
821-41-0
5-Hexen-1-ol

-
B
-
764-59-0
hex-5-en-1-al

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| In benzene for 48h; Irradiation; | A 44% B 43% C 13% |


| Conditions | Yield |
|---|---|
| With tetrahydrofuran; alkaline aqueous hydrogen peroxide; diborane | |
| With sodium hydroxide; 9-borabicyclo[3.3.1]nonane dimer; dihydrogen peroxide 1) 2h, THF; 2) 0 deg C; 3) 50 deg C; Yield given. Multistep reaction; | |
| With sodium tetrahydroborate; 18-crown-6 ether; dihydrogen peroxide; sodium methylate; titanium(III) chloride 1.) THF, 30 deg C, 5 h, 2.) THF, methanol, 40 deg C; Yield given. Multistep reaction; | |
| Multi-step reaction with 3 steps 1: hydrogen bromide 2: acetic acid 3: ethanolic KOH-solution View Scheme |

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| Saponification; |
-
-
109-99-9
tetrahydrofuran

-
-
107671-69-2
Peroxyde de t-butyle et d'hex-5-enyle

-
A
-
3208-31-9
2-pentyl-tetrahydro-furan

-
B
-
821-41-0
5-Hexen-1-ol

-
C
-
134141-96-1
(tetrahydrofuryl-2 methyl)-2 tetrahydropyranne

| Conditions | Yield |
|---|---|
| at 140℃; for 10h; Mechanism; Product distribution; | A 6 % Chromat. B 5 % Chromat. C 20 % Chromat. D 15 % Chromat. E 12 % Chromat. F 8 % Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: 5-Hexen-1-ol With oxalyl dichloride; dimethyl sulfoxide In dichloromethane at -78℃; for 1h; Swern oxidation; Inert atmosphere; Stage #2: With triethylamine In dichloromethane at -78 - 0℃; Swern oxidation; Inert atmosphere; | 100% |
| Stage #1: 5-Hexen-1-ol With oxalyl dichloride; dimethyl sulfoxide at -78℃; for 1h; Stage #2: With triethylamine | 100% |
| With (NH4)4[CuMo6O18(OH)6]·5H2O; oxygen; sodium sulfite In water; acetonitrile at 60℃; under 760.051 Torr; for 6h; | 95% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 20℃; | 100% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; Inert atmosphere; | 98% |
| With triethylamine In tetrahydrofuran at 20℃; | 93% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 1h; | 100% |
| With triethylamine In dichloromethane at -15℃; for 1h; Green chemistry; | 100% |
| With triethylamine In dichloromethane at -15℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; | 100% |
| With zinc(II) chloride In dichloromethane at 20℃; | 100% |
| With silver(l) oxide In dichloromethane at 20℃; | 100% |
-
-
821-41-0
5-Hexen-1-ol

-
-
133708-80-2
benzeneselenenyl m-nitrobenzenesulfonate

-
-
75526-73-7
(2-(phenylselanylmethyl)oxane)

| Conditions | Yield |
|---|---|
| In acetonitrile at 0℃; for 2h; | 100% |
-
-
821-41-0
5-Hexen-1-ol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
85807-84-7
tert-butyl(5-hexenyloxy)dimethylsilane

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide | 100% |
| With 1H-imidazole In tetrahydrofuran at 20℃; | 100% |
| With 1H-imidazole In dichloromethane at 20℃; for 19h; | 100% |
-
-
821-41-0
5-Hexen-1-ol

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
18922-06-0
hex-5-enyl 4-methylbenzenesulfonate

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 5℃; for 3h; | 100% |
| With triethylamine In dichloromethane at 0 - 20℃; for 12h; | 100% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 13h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With trimethylaluminum In dichloromethane for 10h; Heating; | 100% |
| With diethylzinc In 1,2-dichloro-ethane; toluene at 20℃; | 40% |
| Stage #1: diiodomethane With diethylzinc; trifluoroacetic acid In n-heptane; dichloromethane at -10 - 10℃; for 1h; Stage #2: 5-Hexen-1-ol In n-heptane; dichloromethane at 0 - 20℃; for 1.5h; | |
| With diethylzinc; trifluoroacetic acid In hexane; dichloromethane at 0 - 20℃; for 24h; Inert atmosphere; | |
| With trimethylaluminum In hexane; dichloromethane at 20 - 40℃; |
-
-
821-41-0
5-Hexen-1-ol

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
185999-07-9
tert-butyl(hex-5-enyloxy)diphenylsilane

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 0 - 20℃; for 5h; | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; | 99% |
| With 1H-imidazole In dichloromethane at 23℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; | 100% |
| With zinc(II) chloride In dichloromethane at 20℃; | 100% |
| With silver(l) oxide In dichloromethane at 20℃; | 98% |
| With pyridine In dichloromethane at 20℃; Product distribution; Further Variations:; Reagents; Reaction partners; | 90% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at -20℃; for 0.166667h; Inert atmosphere; | 100% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 23℃; Mitsunobu reaction; Inert atmosphere; | 95% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; for 24h; Cooling with ice; Inert atmosphere; | 84% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; |
-
-
821-41-0
5-Hexen-1-ol

-
-
65118-17-4
N-allyl-o-nitrobenzenesulfonamide

-
-
1018389-28-0
N-allyl-N-(hex-5-enyl)-2'-nitrobenzenesulfonamide

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 0 - 20℃; for 1h; | 100% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 1h; Fukuyama-Mitsunobu reaction; | 98% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 20℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In dichloromethane for 2h; Reflux; Inert atmosphere; | 100% |
-
-
821-41-0
5-Hexen-1-ol

-
-
1950-78-3
p-toluenesulfonyl iodide

-
-
141246-59-5
5-iodo-6-(para-toluenesulfonyl)hexan-1-ol

| Conditions | Yield |
|---|---|
| In acetonitrile Ambient temperature; | 99% |
| In acetonitrile Mechanism; Ambient temperature; other alkenols; |
-
-
821-41-0
5-Hexen-1-ol

-
-
21915-57-1
4-(oxiran-2-yl)butyl acetate

| Conditions | Yield |
|---|---|
| With [2-percarboxyethyl] functionalized silica In dichloromethane at 20℃; for 1h; | 99% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0 - 20℃; | 86% |
| With 3-chloro-benzenecarboperoxoic acid In water for 2h; pH=7.0; | 85% |
-
-
821-41-0
5-Hexen-1-ol

-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
71138-63-1
(5-hexenyloxy)trimethylsilane

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In neat (no solvent) at 50℃; for 1.5h; | 99% |
| at 20℃; immediate distillation; Yield given; |

| Conditions | Yield |
|---|---|
| With C30H26N6Ru(2+)*2Cl(1-); sodium L-ascorbate In methanol; acetonitrile for 0.5h; Inert atmosphere; Irradiation; | 99% |
| With 2,6-dimethylpyridine In acetonitrile at 20℃; for 12h; Catalytic behavior; Reagent/catalyst; Wavelength; Irradiation; Inert atmosphere; Sealed tube; | 93% |
| With copper diacetate; Piperonyl butoxide In methanol for 48h; Ambient temperature; | 90% |
-
-
821-41-0
5-Hexen-1-ol

-
-
246540-39-6
allyl bromomethanesulfonamide

-
-
246540-42-1
N-allyl-C-bromo-N-hex-5-enyl-methanesulfonamide

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran; toluene for 0.5h; Alkylation; Mitsunobu reaction; | 99% |
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran; toluene Alkylation; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
3619-91-8
1,3-bisphosphinopropane

-
-
288145-88-0
1,3-bis[di(6-hydroxyhexyl)phosphino]propane

| Conditions | Yield |
|---|---|
| at 20℃; for 10h; Addition; Irradiation; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
6522-02-7
3-(4-(allyloxy)phenyl)propanoic acid

-
-
515835-01-5
3-(4-allyloxy-phenyl)-propionic acid hex-5-enyl ester

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; | 99% |

| Conditions | Yield |
|---|---|
| With 9-borabicyclo[3.3.1]nonane dimer In tetrahydrofuran at 20℃; for 3h; | 99% |
-
-
821-41-0
5-Hexen-1-ol

| Conditions | Yield |
|---|---|
| cyclopentadienylruthenium(II) trisacetonitrile hexafluorophosphate In acetone at 20℃; for 4h; Alder-ene reaction; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
13154-24-0
triisopropylsilyl chloride

-
-
173470-74-1
(hex-5-en-1-yloxy)triisopropylsilane

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 20℃; for 1h; | 99% |
| With 1H-imidazole In dichloromethane at 0 - 20℃; Inert atmosphere; | 94% |
| With dmap; triethylamine In tetrahydrofuran at 20℃; | 90% |
| Stage #1: 5-Hexen-1-ol With 1H-imidazole; dmap In dichloromethane at 0℃; for 0.0833333h; Inert atmosphere; Stage #2: triisopropylsilyl chloride In dichloromethane at 0 - 20℃; for 3h; Inert atmosphere; | 87% |
| With dmap; triethylamine for 72h; | 83% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 21h; | 99% |
| With triethylamine In dichloromethane at 0 - 20℃; for 21h; | 99% |
| With triethylamine In dichloromethane at 0 - 20℃; for 21h; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
1873-77-4
tris-(trimethylsilyl)silane

-
-
1001067-36-2
6-[tris(trimethylsilyl)]-1-hexanol

| Conditions | Yield |
|---|---|
| With 1,1'-azobis(1-cyanocyclohexanenitrile) In water at 100℃; for 4h; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
146176-60-5
2,2,4,4-tetramethyl-1,3-oxazolidine-3-carbonyl chloride

-
-
152857-60-8
hex-5-enyl 2,2,4,4-tetramethyl-1,3-oxazolidine-3-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: 5-Hexen-1-ol With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 0.5h; Stage #2: 2,2,4,4-tetramethyl-1,3-oxazolidine-3-carbonyl chloride In tetrahydrofuran; mineral oil at 80℃; | 99% |
-
-
821-41-0
5-Hexen-1-ol

-
-
401-55-8
ethyl bromofluoroacetate

-
-
1277181-09-5
ethyl 4-bromo-2-fluoro-8-hydroxyoctanoate

| Conditions | Yield |
|---|---|
| With Ir[(dF(CF3)ppy)2(dtbbpy)]PF6; lithium bromide In water; N,N-dimethyl-formamide for 24h; Visible-light irradiation; Inert atmosphere; | 99% |
| With C30H26N6Ru(2+)*2Cl(1-); lithium bromide In dimethyl sulfoxide for 24h; Inert atmosphere; Irradiation; | 99% |
| With 2,6-diiodo-4,4-difluoro-1,3,5,7-tetramethyl-8-phenyl-4-bora-3a,4a-diaza-s-indacene; sodium L-ascorbate; lithium bromide In water; N,N-dimethyl-formamide at 20℃; for 20h; Inert atmosphere; Schlenk technique; Irradiation; | 89% |
| With 7-(diethylamino)-3-(thiophen-2-yl)-2H-chromen-2-one In ethanol; water at 20℃; for 36h; Schlenk technique; Inert atmosphere; Irradiation; | 65% |
| With 2,6-dimethylpyridine; 4-methoxy-benzaldehyde In acetonitrile at 25℃; for 48h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; | 60% |
-
-
821-41-0
5-Hexen-1-ol

-
-
685-87-0
Diethyl 2-bromomalonate

-
-
1277180-98-9
diethyl 2-(2-bromo-6-hydroxyhexyl)malonate

| Conditions | Yield |
|---|---|
| With Ir[(dF(CF3)ppy)2(dtbbpy)]PF6; lithium bromide In water; N,N-dimethyl-formamide for 24h; Visible-light irradiation; Inert atmosphere; | 99% |
| With [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; lithium bromide In water; N,N-dimethyl-formamide Inert atmosphere; Irradiation; | 99% |
| With 2,6-dimethylpyridine; 4-methoxy-benzaldehyde In acetonitrile at 25℃; for 26h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 18h; | 99% |
5-Hexen-1-ol Specification
The CAS register number of 5-Hexen-1-ol is 821-41-0. It also can be called as Hex-5-en-1-ol and the IUPAC name about this chemical is hex-5-en-1-ol. It belongs to the following product categories, such as omega-Functional Alkanols, Carboxylic Acids, Amines & Halides, omega-Unsaturated Alkanols and so on. This chemical is flammable. When you are using it, please keep away from sources of ignition.
Physical properties about 5-Hexen-1-ol are: (1)ACD/LogP: 1.50; (2)ACD/LogD (pH 5.5): 1.5; (3)ACD/LogD (pH 7.4): 1.5; (4)ACD/BCF (pH 5.5): 8.08; (5)ACD/BCF (pH 7.4): 8.08; (6)ACD/KOC (pH 5.5): 155.3; (7)ACD/KOC (pH 7.4): 155.3; (8)#H bond acceptors: 1; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 5; (11)Polar Surface Area: 9.23Å2; (12)Index of Refraction: 1.43; (13)Molar Refractivity: 31.1 cm3; (14)Molar Volume: 120.1 cm3; (15)Polarizability: 12.33x10-24cm3; (16)Surface Tension: 28 dyne/cm; (17)Enthalpy of Vaporization: 45.06 kJ/mol; (18)Vapour Pressure: 1.5 mmHg at 25°C.
Preparation: this chemical can be prepared by 2-chloromethyl-tetrahydro-pyran. This reaction will need reagent Na and solvent diethyl ether. The reaction time is 16 hour(s). The yield is about 83.5%.
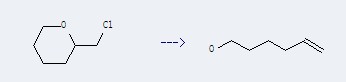
Uses of hex-5-en-1-ol: it can be used to produce 6-bromo-hex-1-ene at Heating. This reaction will need reagent phosphorus tribromide, pyridine and solvent diethyl ether with reaction time of 3 hours. The yield is about 75%.

You can still convert the following datas into molecular structure:
(1)SMILES: OCCCC\C=C
(2)InChI: InChI=1/C6H12O/c1-2-3-4-5-6-7/h2,7H,1,3-6H2
(3)InChIKey: UIZVMOZAXAMASY-UHFFFAOYAD
(4)Std. InChI: InChI=1S/C6H12O/c1-2-3-4-5-6-7/h2,7H,1,3-6H2
(5)Std. InChIKey: UIZVMOZAXAMASY-UHFFFAOYSA-N
Related Products
- 5-Hexen-1-ol
- 82141-73-9
- 82142-16-3
- 82144-78-3
- 82145-74-2
- 82148-19-4
- 821-48-7
- 82151-48-2
- 82152-65-6
- 82153-68-2
- 821-55-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View