-
Name
Acetone oxime
- EINECS 204-820-1
- CAS No. 127-06-0
- Article Data130
- CAS DataBase
- Density 0.91 g/cm3
- Solubility 330 g/L (20 °C) in water
- Melting Point 60-63 °C(lit.)
- Formula C3H7NO
- Boiling Point 135 °C at 760 mmHg
- Molecular Weight 73.0947
- Flash Point 45.2 °C
- Transport Information UN 1325
- Appearance white crystals
- Safety 22-24/25
-
Risk Codes
Xn:Harmful;
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Acetone,oxime (6CI,8CI);Acetoxime;Dimethyl ketoxime;N-Isopropylidenehydroxylamine;NSC 7601;Propanone oxime;b-Isonitrosopropane;2-Propanone, oxime;
- PSA 32.59000
- LogP 0.85640
Synthetic route

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium carbonate; ammonium chloride In water at 60℃; for 5h; | 99.6% |
| With hydroxylamine hydrochloride; sodium carbonate In ethanol; water for 24h; Heating; | 96% |
| With dihydroxylamine phosphate; potassium hydrogencarbonate In water at 39℃; for 1.83333h; Temperature; | 96% |
-
-
79-46-9
2-nitropropane

-
-
80670-36-6
3-diazo-5-phenyl-3H-1,2,4-triazole

-
A
-
100-47-0
benzonitrile

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| at 80℃; Yields of byproduct given; | A n/a B 98% |

-
-
2421-26-3
2-chloro-2-nitrosopropane

-
-
21645-26-1
3-allyl-1,1-diethyl-thiourea

-
-
88-89-1
2,4,6-Trinitrophenol

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In hydrogenchloride; ethanol at 20℃; for 1h; Cyclization; | A 89% B n/a |

-
-
13181-10-7
2,4-dinitrophenyl ether of acetone oxime

-
-
109-73-9
N-butylamine

-
A
-
13059-86-4
2,4-dinitro-N-butylaniline

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In dichloromethane Product distribution; 0 deg C, 72 h; 20 deg C, 48 h; | A 80% B 62% |


| Conditions | Yield |
|---|---|
| With hydroxylamine; oxygen In ethanol under 760.051 Torr; for 2h; Catalytic behavior; Reflux; Green chemistry; | 65% |
-
-
109-57-9
Allylthiourea

-
-
2421-26-3
2-chloro-2-nitrosopropane

-
-
88-89-1
2,4,6-Trinitrophenol

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In hydrogenchloride; ethanol at 20℃; for 1h; Cyclization; | A 64% B n/a |

-
-
2421-26-3
2-chloro-2-nitrosopropane

-
-
7341-63-1
1-allyl-3-phenyl-thiourea

-
-
88-89-1
2,4,6-Trinitrophenol

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In hydrogenchloride; ethanol at 20℃; for 1h; Cyclization; | A 60% B n/a |

-
-
3717-21-3, 3717-22-4, 3235-04-9, 140461-28-5, 140461-29-6
(E)-4-methoxybenzaldoxime

-
-
66-99-9
β-naphthaldehyde

-
A
-
123-11-5
4-methoxy-benzaldehyde

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With perchloric acid In dichloromethane; water at 23℃; for 24h; Inert atmosphere; | A 38% B 33% |

-
-
14384-45-3
N,N'-dihydroxy-2,3-dimethylbutane-2,3-diamine

-
A
-
34493-89-5
3,4-Dihydro-3,3,4,4-tetramethyl-1,2-diazete 1,2-dioxide

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With tert-butylhypochlorite In dichloromethane at 20℃; for 2h; | A n/a B 31% |

| Conditions | Yield |
|---|---|
| In dichloromethane Product distribution; 0 deg C, 72 h; 20 deg C, 48 h; | 29% |
-
-
932-90-1, 622-31-1, 140461-24-1, 140461-25-2, 622-32-2
syn-benzaldehyde oxime

-
-
67-64-1
acetone

-
A
-
100-52-7
benzaldehyde

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With perchloric acid In water; water-d2 at 23℃; for 24h; Inert atmosphere; | A n/a B 29% |

-
-
16939-49-4, 24652-60-6, 66046-34-2
(E)-4-(trifluoromethyl)benzaldoxime

-
-
66-99-9
β-naphthaldehyde

-
A
-
455-19-6
4-Trifluoromethylbenzaldehyde

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With perchloric acid In dichloromethane; water at 23℃; for 24h; Inert atmosphere; | A 15% B 9% |

-
-
60-29-7
diethyl ether

-
-
2421-26-3
2-chloro-2-nitrosopropane

-
-
544-97-8
dimethyl zinc(II)

-
A
-
34557-54-5
methane

-
B
-
593-77-1
N-Methylhydroxylamine

-
C
-
67-64-1
acetone

-
D
-
127-06-0
acetone oxime

-
-
60-29-7
diethyl ether

-
-
7119-91-7
2-bromo-2-nitroso-propane

-
-
557-20-0
diethylzinc

-
A
-
67-64-1
acetone

-
B
-
598-31-2
1-bromoacetone

-
C
-
624-81-7
ethylhydroxylamine

-
D
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| Produkte sind noch Aethan und Aethylen; |

-
-
79-46-9
2-nitropropane

-
-
13880-55-2
N,N,N',N'-tetramethyl-C-phenylmethanediamine

-
A
-
611-74-5
N,N-dimethylbenzamide

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| at 120 - 130℃; |


| Conditions | Yield |
|---|---|
| With diethyl ether; alkyl magnesium halide | |
| With diethyl ether; aryl magnesium halide | |
| With diethyl ether; zinc dialkyl |

| Conditions | Yield |
|---|---|
| With diethyl ether; ammonia | |
| With diethyl ether; alkyl magnesium halide | |
| With diethyl ether; zinc dialkyl | |
| With diethyl ether; aryl magnesium halide |

| Conditions | Yield |
|---|---|
| With methanol; palladium Hydrogenation; | |
| With methanol; palladium Hydrogenation; |

| Conditions | Yield |
|---|---|
| With diethyl ether; hydroxylamine | |
| With diammonium sulfide; diethyl ether | |
| With diethyl ether; potassium hydrosulfide | |
| With diethyl ether; aluminium amalgam |

| Conditions | Yield |
|---|---|
| With ethanol; hydroxylamine hydrochloride |

| Conditions | Yield |
|---|---|
| With water; hydroxylamine |
-
-
536-57-2
p-toluene sulfinic acid

-
-
70-55-3
toluene-4-sulfonamide

-
-
57267-72-8
toluene-4-sulfinic acid ; ammonium salt

-
A
-
932-90-1, 622-31-1, 140461-24-1, 140461-25-2, 622-32-2
syn-benzaldehyde oxime

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| Verschmelzenden; |


| Conditions | Yield |
|---|---|
| With caro's acid; magnesium oxide | |
| With sodium tungstate; dihydrogen peroxide |
-
-
109-95-5
ethyl nitrite

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
-
611-70-1
phenyl isopropyl ketone

-
A
-
532-32-1
sodium benzoate

-
B
-
93-89-0
benzoic acid ethyl ester

-
C
-
127-06-0
acetone oxime

-
-
79-46-9
2-nitropropane

-
-
93-04-9
2-Methoxynaphthalene

-
A
-
2421-26-3
2-chloro-2-nitrosopropane

-
B
-
13101-92-3
1-chloro-2-methoxynaphthalene

-
C
-
67-64-1
acetone

-
D
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With pyridine hydrochloride Product distribution; Heating; |


| Conditions | Yield |
|---|---|
| With Trimethyl(methylthio)silane; potassium hydride 1) THF, rt, 1 h, 2) rt, 48 h; Yield given. Multistep reaction; |
-
-
61558-18-7
2-hydroxyamino-propan-2-ol

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In methanol; water at 25℃; Rate constant; |
-
-
14384-45-3
N,N'-dihydroxy-2,3-dimethylbutane-2,3-diamine

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With air at 20 - 25℃; for 168h; | 0.97 g |
-
-
68385-34-2
N-(2-hydroxy-1,1-dimethyl-2-phenylethyl)hydroxylamine

-
A
-
100-52-7
benzaldehyde

-
B
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In water at 25℃; Product distribution; Mechanism; buffer of pH 8.8, controlled potential electrolysis; further N-alkylhydroxylamines with a β-hydroxy group; |

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid at 50℃; for 1h; Beckmann rearrangement; | 100% |
| With bismuth(III) chloride for 0.166667h; Beckmann rearrangement; microwave irradiation; | 90% |
| With indium(III) triflate In acetonitrile for 2h; Beckmann rearrangement; Heating; | 90% |
-
-
95092-10-7
N-methoxy-N-methyl-2-phenylacetamide

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| Stage #1: N-methoxy-N-methyl-2-phenylacetamide; acetone oxime With n-butyllithium In tetrahydrofuran at 0℃; Stage #2: With sulfuric acid In tetrahydrofuran; water for 1h; Heating; | 100% |
| With n-butyllithium; sulfuric acid 1.) THF, hexane, 0 deg C, 30 min, 2.) THF, reflux, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With pyridine; di-tert-butyl dicarbonate In tetrahydrofuran at 20℃; for 20h; | 100% |
| With dicyclohexyl-carbodiimide |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In diethyl ether at 20℃; for 0.5h; | 100% |

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In toluene at 60℃; for 2h; | 100% |
-
-
56-35-9
bis(tri-n-butyltin)oxide

-
-
127-06-0
acetone oxime

-
-
10218-32-3
Isopropylidenaminooxy-tributyl-stannan

| Conditions | Yield |
|---|---|
| In toluene 1:2; azeotropic dehydration; 2 h; | 100% |
| In benzene 1:2; azeotropic dehydration; 2 h; | 100% |
| In benzene 1:2; azeotropic dehydration; 2 h; | 100% |
-
-
914935-38-9
1-{5-({[tert-butyl(diphenyl)silyl]oxy}methyl)-4-[(2R,6S)-2,6-dimethylmorpholin-4-yl]-2,3-difluorophenyl}ethanone

-
-
127-06-0
acetone oxime

-
-
1223444-75-4
1-(5-((tert-butyldiphenylsilyloxy)methyl)-4-((2R,6S)-2,6-dimethylmorpholino)-3-fluoro-2-(propan-2-ylideneaminooxy)phenyl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.75h; Stage #2: 1-{5-({[tert-butyl(diphenyl)silyl]oxy}methyl)-4-[(2R,6S)-2,6-dimethylmorpholin-4-yl]-2,3-difluorophenyl}ethanone In tetrahydrofuran at 20℃; for 3h; | 100% |
| Stage #1: acetone oxime With potassium tert-butylate In tetrahydrofuran at 18 - 25℃; for 0.75h; Stage #2: 1-{5-({[tert-butyl(diphenyl)silyl]oxy}methyl)-4-[(2R,6S)-2,6-dimethylmorpholin-4-yl]-2,3-difluorophenyl}ethanone In tetrahydrofuran for 3h; Stage #3: With water; ammonium chloride In tetrahydrofuran |
-
-
886373-28-0
5-bromo-3-fluoropyridine-2-carbonitrile

-
-
127-06-0
acetone oxime

-
-
1448674-24-5
3,5-bis-isopropylideneaminooxypyridine-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 1h; Stage #2: 5-bromo-3-fluoropyridine-2-carbonitrile In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; | 100% |
-
-
443286-95-1
bis(N-phenylsalicylideneiminato)aluminium(III)di(μ-isopropoxo)di(isopropoxo) aluminium(III)

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In benzene byproducts: C3H7OH; under strictly unhydrous conditions; in 1:2 molar ratio (Al-complex:(CH3)2CNOH in refluxing unhydrous C6H6 for 4 h, C3H7OH fractionated azeotropically with C6H6; excess solvent stripped off under reduced pressure, recrystd. (C6H6/C6H14); elem. anal., detd. by IR, NMR; | 99.5% |

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With trifluoromethylsulfonic anhydride; triethylamine In toluene at -78℃; for 0.0833333h; Stage #2: methyl cyanoacetate sodium enolate In toluene at -78 - 20℃; for 1h; | 99% |
| Stage #1: acetone oxime With trifluoromethanesulfonic acid anhydride; triethylamine In toluene at -78℃; for 0.0833333h; Stage #2: methyl cyanoacetate sodium enolate In tetrahydrofuran; toluene at -78 - 20℃; | 99% |
-
-
443286-95-1
bis(N-phenylsalicylideneiminato)aluminium(III)di(μ-isopropoxo)di(isopropoxo) aluminium(III)

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In benzene byproducts: C3H7OH; under strictly unhydrous conditions; in 1:1 molar ratio (Al-complex:(CH3)2CNOH in refluxing unhydrous C6H6 for 4 h, C3H7OH fractionated azeotropically with C6H6; excess solvent stripped off under reduced pressure, recrystd. (C6H6/C6H14); elem. anal., detd. by IR, NMR; | 99% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium hydroxide; lithium hydroxide In N,N-dimethyl acetamide; water at 50℃; Stage #2: benzyl chloride In N,N-dimethyl acetamide; water at 62℃; for 2.25h; | 98.96% |
| With ethanol; sodium | |
| With sodium In ethanol at 25℃; Inert atmosphere; | 10.9 g |

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium hydroxide; lithium hydroxide In N,N-dimethyl acetamide; water at 50℃; Stage #2: p-bromobenzyl chloride In N,N-dimethyl acetamide; water at 65℃; for 2.25h; | 98.28% |

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium hydroxide; lithium hydroxide In N,N-dimethyl acetamide; water at 55℃; Stage #2: 2,4-Dichlorobenzyl chloride In N,N-dimethyl acetamide; water at 60℃; for 2.33333h; | 98.25% |

| Conditions | Yield |
|---|---|
| With sodium methylate at 70℃; under 760.051 Torr; for 8h; Temperature; Reagent/catalyst; Large scale; | 98.2% |

| Conditions | Yield |
|---|---|
| With sodium methylate at 130℃; under 37.5038 Torr; for 4h; Time; Large scale; | 98.2% |

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| With HCl In methanol addn. of methanolic soln. of HCl to mixt. of Mo compd. and oxime, stirred at room temp. for 7 h, pptn.; filtered off, washed (Et2O); | 98% |
-
-
33411-10-8
(η6-1-fluoro-3-methylbenzene)tricarbonylchromium(0)

-
-
14866-33-2
tetraoctyl ammonium bromide

-
-
127-06-0
acetone oxime

-
-
97932-55-3
(CO)3CrC6H4(CH3)ONC(CH3)2

| Conditions | Yield |
|---|---|
| With potassium hydroxide In benzene (N2), stirred for 1 h; solvent removed under vac., residue dissolved in Et2O, filtered on Celite, evaporated, crystd. from di-isopropyl ether; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium hydroxide; lithium hydroxide In N,N-dimethyl acetamide; water at 53℃; Stage #2: 1-chloro-2-(chloromethyl)benzene In N,N-dimethyl acetamide; water at 65℃; for 2.16667h; | 97.11% |
-
-
124-70-9
Dichloromethylvinylsilane

-
-
127-06-0
acetone oxime

-
-
73160-13-1
bis(isopropylideneaminooxy)methylvinylsilane

| Conditions | Yield |
|---|---|
| In hexane at 30 - 40℃; for 1h; | 97% |
| In toluene at 55 - 60℃; for 2h; | 61% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: acetone oxime With potassium hydroxide; lithium hydroxide In N,N-dimethyl acetamide; water at 50℃; Stage #2: 1-Chloro-4-(chloromethyl)benzene In N,N-dimethyl acetamide; water at 60℃; for 2.33333h; | 96.94% |
-
-
70935-14-7
3-hydroxyphenylglyoxal

-
-
5392-28-9
2,4,5,6-tetraaminopyrimidine sulfate

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| Stage #1: 3-hydroxyphenylglyoxal; acetone oxime With hydrogenchloride In methanol; water at 50℃; for 1h; pH=~ 2 - ~ 3; Stage #2: 2,4,5,6-tetraaminopyrimidine sulfate In water at 20℃; for 9h; Heating / reflux; Stage #3: With water; sodium hydrogencarbonate at 20℃; pH=~ 6 - ~ 7; | 96.5% |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium carbonate In methanol at 60℃; under 750.075 Torr; | 96.5% |
-
-
21160-82-7
4-nitrophenyl N2,N6-bis[(phenylmethoxy)carbonyl]-L-lysinate

-
-
127-06-0
acetone oxime

| Conditions | Yield |
|---|---|
| In chloroform Ambient temperature; | 96% |
-
-
127-06-0
acetone oxime

-
-
107-56-2
O,O-diisopropyl hydrogen phosphorodithioate

-
-
75320-62-6
S-<1-(hydroxyamino)-1-methylethyl> O,O-diisopropyl phosphorodithioate

| Conditions | Yield |
|---|---|
| In diethyl ether for 3h; | 96% |
Acetone oxime Chemical Properties
The molecular structure of Acetone oxime (CAS NO.127-06-0):
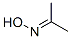
IUPAC Name: N-Propan-2-ylidenehydroxylamine
Molecular Weight: 73.09378 g/mol
Molecular Formula: C3H7NO
Density: 0.91 g/cm3
Melting Point: 60-63 °C(lit.)
Boiling Point: 135 °C at 760 mmHg
Flash Point: 45.2 °C
Index of Refraction: 1.41
Molar Refractivity: 19.9 cm3
Molar Volume: 80.2 cm3
Surface Tension: 25.6 dyne/cm
Enthalpy of Vaporization: 41.09 kJ/mol
Vapour Pressure: 4.65 mmHg at 25 °C
Water Solubility: 330 g/L (20 °C)
XLogP3: 0.1
H-Bond Donor: 1
H-Bond Acceptor: 2
Tautomer Count: 2
Exact Mass: 73.052764
MonoIsotopic Mass: 73.052764
Topological Polar Surface Area: 32.6
Heavy Atom Count: 5
Canonical SMILES: CC(=NO)C
InChI: InChI=1S/C3H7NO/c1-3(2)4-5/h5H,1-2H3
InChIKey: PXAJQJMDEXJWFB-UHFFFAOYSA-N
EINECS: 204-820-1
Product Categories: Pharmaceutical Intermediates; Oximino Silanes; Nitrogen Compounds; Organic Building Blocks; Oximes
Acetone oxime Uses
Acetone oxime (CAS NO.127-06-0) is used in organic synthesis. It is also used as an analytical reagent for the determination of cobalt.
Acetone oxime Toxicity Data With Reference
| 1. | orl-rat LD:>500 mg/kg | NCNSA6 National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. 5 (1953),26. | ||
| 2. | ipr-mus LD50:4000 mg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 119 (1957),522. |
Acetone oxime Consensus Reports
Reported in EPA TSCA Inventory.
Acetone oxime Safety Profile
Safety Statements: 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
RIDADR: UN1325
WGK Germany: 3
RTECS: AL6825000
F: 21
HazardClass: 4.1
PackingGroup: III
Moderately toxic by ingestion and intraperitoneal routes. When heated to decomposition it emits toxic fumes of NOx.
Acetone oxime Specification
Acetone oxime (CAS NO.127-06-0) is also named as 2-Propanone oxime ; 4-01-00-03202 (Beilstein Handbook Reference) ; AI3-52259 ; Acetonoxime ; Acetoxime ; BRN 1560146 ; CCRIS 5 ; NSC 7601 ; beta-Isonitrosopropane . Acetone oxime (CAS NO.127-06-0) is white crystals. It is easily soluble in water, ethanol, ether and acetone, soluble in acid and base.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View